|
Crabbé Reaction
The Crabbé reaction (or Crabbé allene synthesis, Crabbé–Ma allene synthesis) is an organic reaction that converts a terminal alkyne and aldehyde (or, sometimes, a ketone) into an allene in the presence of a soft Lewis acid catalyst (or stoichiometric promoter) and secondary amine. Given continued developments in scope and generality, it is a convenient and increasingly important method for the preparation of allenes, a class of compounds often viewed as exotic and synthetically challenging to access. Overview and scope The transformation was discovered in 1979 by Pierre Crabbé and coworkers at the Université Scientifique et Médicale (currently merged into Université Grenoble Alpes) in Grenoble, France. As initially discovered, the reaction was a one-carbon homologation reaction (the Crabbé homologation) of a terminal alkyne into a terminal allene using formaldehyde as the carbon source, with diisopropylamine as base and copper(I) bromide as catalyst. Despite the excell ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Reaction
Organic reactions are chemical reactions involving organic compounds. The basic organic chemistry reaction types are addition reactions, elimination reactions, substitution reactions, pericyclic reactions, rearrangement reactions, Mechanistic Organic Photochemistry, photochemical reactions and organic redox reaction, redox reactions. In organic synthesis, organic reactions are used in the construction of new organic molecules. The production of many man-made chemicals such as drugs, plastics, food additives, fabrics depend on organic reactions. The oldest organic reactions are combustion of organic fuels and saponification of fats to make soap. Modern organic chemistry starts with the Wöhler synthesis in 1828. In the history of the Nobel Prize in Chemistry awards have been given for the invention of specific organic reactions such as the Grignard reaction in 1912, the Diels-Alder reaction in 1950, the Wittig reaction in 1979 and olefin metathesis in 2005. Classifications Organic c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
University Of Missouri
The University of Missouri (Mizzou, MU, or Missouri) is a public university, public Land-grant university, land-grant research university in Columbia, Missouri. It is Missouri's largest university and the flagship of the four-campus University of Missouri System. MU was founded in 1839 and was the first public university west of the Mississippi River. It has been a member of the Association of American Universities since 1908 and is classified among "R1: Doctoral Universities – Very high research activity". To date, the University of Missouri alumni, faculty, and staff include 18 Rhodes Scholars, 19 Truman Scholars, 141 Fulbright Scholars, 7 Governors of Missouri, and 6 members of the U.S. Congress. Enrolling 31,401 students in 2021, it offers more than 300 degree programs in thirteen major academic divisions. Its well-known Missouri School of Journalism was founded by Walter Williams (journalist), Walter Williams in 1908 as the world's first journalism school; It publishes ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Chemistry
Organic chemistry is a subdiscipline within chemistry involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms.Clayden, J.; Greeves, N. and Warren, S. (2012) ''Organic Chemistry''. Oxford University Press. pp. 1–15. . Study of structure determines their structural formula. Study of properties includes physical and chemical properties, and evaluation of chemical reactivity to understand their behavior. The study of organic reactions includes the chemical synthesis of natural products, drugs, and polymers, and study of individual organic molecules in the laboratory and via theoretical ( in silico) study. The range of chemicals studied in organic chemistry includes hydrocarbons (compounds containing only carbon and hydrogen) as well as compounds based on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (included in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkynylation
In organic chemistry, alkynylation is an addition reaction in which a terminal alkyne () is added to a carbonyl group () to form an Alpha and beta carbon, α-alkynyl alcohol (chemistry), alcohol (). When the acetylide is formed from acetylene (), the reaction gives an α-ethynyl alcohol. This process is often referred to as ethynylation. Such processes often involve metal acetylide intermediates. Scope The principal reaction of interest involves the addition of the acetylene () to a ketone () or aldehyde (): :RR'C=O + HC#CR'' -> RR'C(OH)C#CR'' The reaction proceeds with retention of the triple bond. For aldehydes and unsymmetrical ketones, the product is chiral, hence there is interest in asymmetric variants. These reactions invariably involve metal-acetylide intermediates. This reaction was discovered by chemist John Ulric Nef (chemist), John Ulric Nef in 1899 while experimenting with reactions of elemental sodium, phenylacetylene, and acetophenone. For this reason, the re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coupling Reaction
A coupling reaction in organic chemistry is a general term for a variety of reactions where two fragments are joined together with the aid of a metal catalyst. In one important reaction type, a main group organometallic compound of the type R-M (R = organic fragment, M = main group center) reacts with an organic halide of the type R'-X with formation of a new carbon-carbon bond in the product R-R'. The most common type of coupling reaction is the cross coupling reaction. Richard F. Heck, Ei-ichi Negishi, and Akira Suzuki were awarded the 2010 Nobel Prize in Chemistry for developing palladium-catalyzed cross coupling reactions. Broadly speaking, two types of coupling reactions are recognized: *Heterocouplings combine two different partners, such as in the Heck reaction of an alkene (RC=CH) and an alkyl halide (R'-X) to give a substituted alkene, or the Corey–House synthesis of an alkane by the reaction of a lithium diorganylcuprate (R2CuLi) with an organyl (pseudo)halide (R'X ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
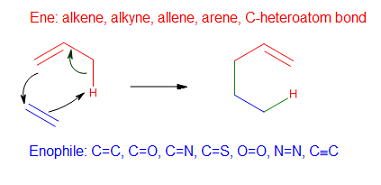
Ene Reaction
In organic chemistry, the ene reaction (also known as the Alder-ene reaction by its discoverer Kurt Alder in 1943) is a chemical reaction between an alkene with an allylic hydrogen (the ene) and a compound containing a multiple bond (the enophile), in order to form a new σ-bond with migration of the ene double bond and 1,5 hydrogen shift. The product is a substituted alkene with the double bond shifted to the allylic position. This transformation is a group transfer pericyclic reaction, and therefore, usually requires highly activated substrates and/or high temperatures. Nonetheless, the reaction is compatible with a wide variety of functional groups that can be appended to the ene and enophile moieties. Many useful Lewis acid-catalyzed ene reactions have been also developed, which can afford high yields and selectivities at significantly lower temperatures, making the ene reaction a useful C–C forming tool for the synthesis of complex molecules and natural products. Ene ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mannich Reaction
In organic chemistry, the Mannich reaction is a three-component organic reaction that involves the amino alkylation of an acidic proton next to a carbonyl () functional group by formaldehyde () and a primary or secondary amine () or ammonia (). The final product is a β-amino-carbonyl compound also known as a Mannich base. Reactions between aldimines and α-methylene carbonyls are also considered Mannich reactions because these imines form between amines and aldehydes. The reaction is named after Carl Mannich. center, 500px, Scheme 1 - Ammonia or an amine reacts with formaldehyde and an alpha acidic proton of a carbonyl compound to a beta amino carbonyl compound. The Mannich reaction starts with the nucleophilic addition of an amine to a carbonyl group followed by dehydration to the Schiff base. The Schiff base is an electrophile which reacts in a second step in an electrophilic addition with an enol formed from a carbonyl compound containing an acidic alpha-proton. The Mann ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
A3 Coupling Reaction
The A3 coupling (also known as A3 coupling reaction or the aldehyde-alkyne-amine reaction), coined by Prof. Chao-Jun Li of McGill University, is a type of multicomponent reaction involving an aldehyde, an alkyne and an amine which react to give a propargylamine. The reaction proceeds via direct dehydrative condensation and requires a metal catalyst, typically based on ruthenium/copper, gold or silver. Chiral catalyst can be used to give an enantioselective reaction, yielding a chiral amine. The solvent can be water. In the catalytic cycle the metal activates the alkyne to a metal acetylide, the amine and aldehyde combine to form an imine which then reacts with the acetylide in a nucleophilic addition. The reaction type was independently reported by three research groups in 2001 -2002; one report on a similar reaction dates back to 1953. If the amine substituents have an alpha hydrogen present and provided a suitable zinc or copper catalyst is used, the A3 coupling product may u ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cdi2crabbe
CDI, CDi, CD-i, or .cdi may refer to: Businesses and organizations Government and political organizations *Center for Defense Information, a Washington, D.C. think tank founded in 1972 *Center for Digital Inclusion, originally "Comitê para Democratização da Informática", a non-governmental organization in Brazil * Centrist Democrat International, formerly the Christian Democrat International, a political international formed in 1961 *China Development Institute, a Shenzhen, China think tank founded in 1989 * Comisión Nacional para el Desarrollo de los Pueblos Indígenas, Mexico's National Commission for the Development of Indigenous Peoples *California Department of Insurance, The State of California insurance regulation department Schools and colleges *CDI College, a for-profit college in Canada *Collège des Ingénieurs, educational institution in France, Germany and Italy *Control Data Institute, an international technical vocational school created by the American Control ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |