|
Copper(I) Fluoride
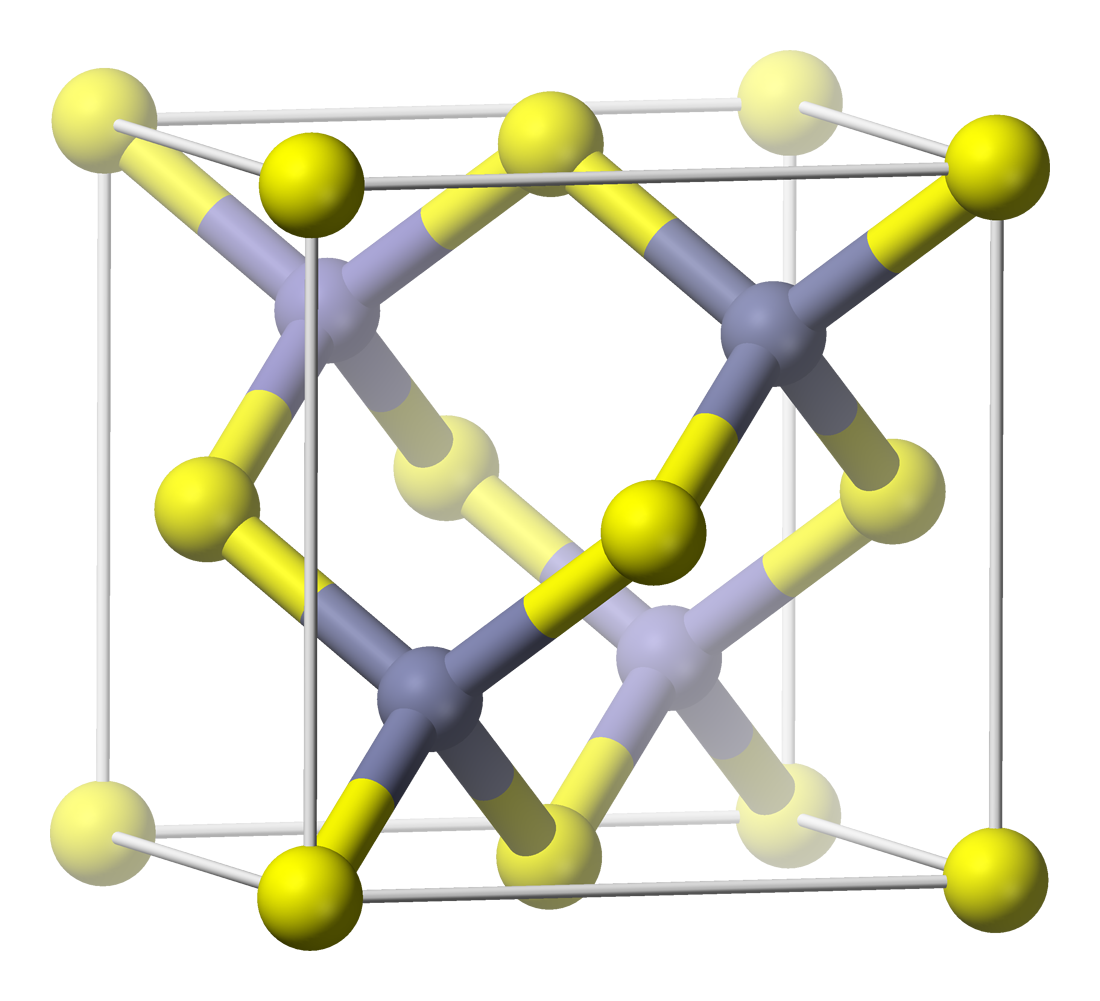
Copper(I) fluoride or cuprous fluoride is an inorganic compound with the chemical formula CuF. Its existence is uncertain. It was reported in 1933 to have a Cubic crystal system#Zincblende structure, sphalerite-type crystal structure. Modern textbooks state that CuF is not known, since fluorine is so electronegative that it will always oxidise copper to its +2 oxidation state. Coordination complex, Complexes of CuF such as [(Ph3P)3CuF] are, however, known and well characterised. Synthesis and reactivity Unlike other copper(I) halides like copper(I) chloride, copper(I) fluoride tends to disproportionate into copper(II) fluoride and copper in a one-to-one ratio at ambient conditions, unless it is stabilised through complexation as in the example of [Cu(N2)F]. :2CuF → Cu + CuF2 See also * Copper(II) fluoride, the other simple fluoride of copper References {{fluorine compounds Fluorides Metal halides Copper(I) compounds Zincblende crystal structure Hypothetical_chemica ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sphalerite
Sphalerite (sometimes spelled sphaelerite) is a sulfide mineral with the chemical formula . It is the most important ore of zinc. Sphalerite is found in a variety of deposit types, but it is primarily in Sedimentary exhalative deposits, sedimentary exhalative, Carbonate-hosted lead-zinc ore deposits, Mississippi-Valley type, and Volcanogenic massive sulfide ore deposit, volcanogenic massive sulfide deposits. It is found in association with galena, chalcopyrite, pyrite (and other sulfide mineral, sulfides), calcite, dolomite (mineral), dolomite, quartz, rhodochrosite, and fluorite. German geologist Ernst Friedrich Glocker discovered sphalerite in 1847, naming it based on the Greek word ''sphaleros'', meaning "deceiving", due to the difficulty of identifying the mineral. In addition to zinc, sphalerite is an ore of cadmium, gallium, germanium, and indium. Miners have been known to refer to sphalerite as ''zinc blende'', ''black-jack'', and ''ruby blende''. Marmatite is an opaque ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electronegative
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. The higher the associated electronegativity, the more an atom or a substituent group attracts electrons. Electronegativity serves as a simple way to quantitatively estimate the bond energy, and the sign and magnitude of a bond's chemical polarity, which characterizes a bond along the continuous scale from covalent to ionic bonding. The loosely defined term electropositivity is the opposite of electronegativity: it characterizes an element's tendency to donate valence electrons. On the most basic level, electronegativity is determined by factors like the nuclear charge (the more protons an atom has, the more "pull" it will have on electrons) and the number and location ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Copper(I) Compounds
Copper is a chemical element with the symbol Cu (from la, cuprum) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkish-orange color. Copper is used as a conductor of heat and electricity, as a building material, and as a constituent of various metal alloys, such as sterling silver used in jewelry, cupronickel used to make marine hardware and coins, and constantan used in strain gauges and thermocouples for temperature measurement. Copper is one of the few metals that can occur in nature in a directly usable metallic form (native metals). This led to very early human use in several regions, from circa 8000 BC. Thousands of years later, it was the first metal to be smelted from sulfide ores, circa 5000 BC; the first metal to be cast into a shape in a mold, c. 4000 BC; and the first metal to be purposely alloyed with another metal, tin, to create bronze, c. 350 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metal Halides
Metal halides are compounds between metals and halogens. Some, such as sodium chloride are ionic, while others are covalently bonded. A few metal halides are discrete molecules, such as uranium hexafluoride, but most adopt polymeric structures, such as palladium chloride. File:NaCl polyhedra.png, Sodium chloride crystal structure File:Uranium-hexafluoride-unit-cell-3D-balls.png, Discrete UF6 molecules File:Alpha-palladium(II)-chloride-xtal-3D-balls.png, Infinite chains of one form of palladium chloride Preparation The halogens can all react with metals to form metal halides according to the following equation: :2M + nX2 → 2MXn where M is the metal, X is the halogen, and MXn is the metal halide. In practice, this type of reaction may be very exothermic, hence impractical as a preparative technique. Additionally, many transition metals can adopt multiple oxidation states, which complicates matters. As the halogens are strong oxidizers, direct combination of the elements usua ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fluorides
Fluoride (). According to this source, is a possible pronunciation in British English. is an inorganic, monatomic anion of fluorine, with the chemical formula (also written ), whose salts are typically white or colorless. Fluoride salts typically have distinctive bitter tastes, and are odorless. Its salts and minerals are important chemical reagents and industrial chemicals, mainly used in the production of hydrogen fluoride for fluorocarbons. Fluoride is classified as a weak base since it only partially associates in solution, but concentrated fluoride is corrosive and can attack the skin. Fluoride is the simplest fluorine anion. In terms of charge and size, the fluoride ion resembles the hydroxide ion. Fluoride ions occur on Earth in several minerals, particularly fluorite, but are present only in trace quantities in bodies of water in nature. Nomenclature Fluorides include compounds that contain ionic fluoride and those in which fluoride does not dissociate. The nomenc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Disproportionate
In chemistry, disproportionation, sometimes called dismutation, is a redox reaction in which one compound of intermediate oxidation state converts to two compounds, one of higher and one of lower oxidation states. More generally, the term can be applied to any desymmetrizing reaction of the following type, regardless of whether it is a redox or some other type of process: :2A -> A' + A'' Examples *Mercury(I) chloride disproportionates upon UV-irradiation: :Hg2Cl2 → Hg + HgCl2 *Phosphorous acid disproportionates upon heating to give phosphoric acid and phosphine: :4 → 3 H3PO4 + PH3 *Desymmetrizing reactions are sometimes referred to as disproportionation, as illustrated by the thermal degradation of bicarbonate: :2 → + H2CO3 :The oxidation numbers remain constant in this acid-base reaction. This process is also called autoionization. *Another variant on disproportionation is radical disproportionation, in which two radicals form an alkene and an alkane. : Reverse ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Copper(I) Chloride
Copper(I) chloride, commonly called cuprous chloride, is the lower chloride of copper, with the formula CuCl. The substance is a white solid sparingly soluble in water, but very soluble in concentrated hydrochloric acid. Impure samples appear green due to the presence of copper(II) chloride (CuCl2). History Copper(I) chloride was first prepared by Robert Boyle in the mid-seventeenth century from mercury(II) chloride ("Venetian sublimate") and copper metal: :HgCl2 + 2 Cu → 2 CuCl + Hg In 1799, J.L. Proust characterized the two different chlorides of copper. He prepared CuCl by heating CuCl2 at red heat in the absence of air, causing it to lose half of its combined chlorine followed by removing residual CuCl2 by washing with water. An acidic solution of CuCl was formerly used for analysis of carbon monoxide content in gases, for example in Hempel's gas apparatus. This application was significant during the nineteenth and early twentieth centuries when coal gas was widely ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Inorganica Chimica Acta
''Inorganica Chimica Acta'' is a peer-reviewed scientific journal published since 1967 that covers original research and reviews of fundamental and applied aspects of inorganic chemistry. See also * List of scientific journals in chemistry A ''list'' is any set of items in a row. List or lists may also refer to: People * List (surname) Organizations * List College, an undergraduate division of the Jewish Theological Seminary of America * SC Germania List, German rugby unio ... External links * Elsevier academic journals Inorganic chemistry journals Publications established in 1967 English-language journals Journals published between 13 and 25 times per year {{chem-journal-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coordination Complex
A coordination complex consists of a central atom or ion, which is usually metallic and is called the ''coordination centre'', and a surrounding array of bound molecules or ions, that are in turn known as ''ligands'' or complexing agents. Many metal-containing compounds, especially those that include transition metals (elements like titanium that belong to the Periodic Table's d-block), are coordination complexes. Nomenclature and terminology Coordination complexes are so pervasive that their structures and reactions are described in many ways, sometimes confusingly. The atom within a ligand that is bonded to the central metal atom or ion is called the donor atom. In a typical complex, a metal ion is bonded to several donor atoms, which can be the same or different. A polydentate (multiple bonded) ligand is a molecule or ion that bonds to the central atom through several of the ligand's atoms; ligands with 2, 3, 4 or even 6 bonds to the central atom are common. These compl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxidation State
In chemistry, the oxidation state, or oxidation number, is the hypothetical charge of an atom if all of its bonds to different atoms were fully ionic. It describes the degree of oxidation (loss of electrons) of an atom in a chemical compound. Conceptually, the oxidation state may be positive, negative or zero. While fully ionic bonds are not found in nature, many bonds exhibit strong ionicity, making oxidation state a useful predictor of charge. The oxidation state of an atom does not represent the "real" formal charge on that atom, or any other actual atomic property. This is particularly true of high oxidation states, where the ionization energy required to produce a multiply positive ion is far greater than the energies available in chemical reactions. Additionally, the oxidation states of atoms in a given compound may vary depending on the choice of electronegativity scale used in their calculation. Thus, the oxidation state of an atom in a compound is purely a formalism. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zeitschrift Für Anorganische Und Allgemeine Chemie
The ''Zeitschrift für anorganische und allgemeine Chemie'' (''Journal of Inorganic and General Chemistry'') is a semimonthly peer-reviewed scientific journal covering inorganic chemistry, published by Wiley-VCH. The editors-in-chief are Thomas F. Fässler, Christian Limberg, Guodong Qian, and David Scheschkewitz. Originally the journal was published in German, but nowadays it is completely in English. Abstracting and indexing The journal is abstracted and indexed in the following databases: According to the ''Journal Citation Reports'', the journal has a 2021 impact factor The impact factor (IF) or journal impact factor (JIF) of an academic journal is a scientometric index calculated by Clarivate that reflects the yearly mean number of citations of articles published in the last two years in a given journal, as i ... of 1.414, ranking it 40th out of 46 journals in the category "Chemistry, Inorganic & Nuclear". References External links * Chemistry journals Wiley-VCH aca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Copper(I) Chloride
Copper(I) chloride, commonly called cuprous chloride, is the lower chloride of copper, with the formula CuCl. The substance is a white solid sparingly soluble in water, but very soluble in concentrated hydrochloric acid. Impure samples appear green due to the presence of copper(II) chloride (CuCl2). History Copper(I) chloride was first prepared by Robert Boyle in the mid-seventeenth century from mercury(II) chloride ("Venetian sublimate") and copper metal: :HgCl2 + 2 Cu → 2 CuCl + Hg In 1799, J.L. Proust characterized the two different chlorides of copper. He prepared CuCl by heating CuCl2 at red heat in the absence of air, causing it to lose half of its combined chlorine followed by removing residual CuCl2 by washing with water. An acidic solution of CuCl was formerly used for analysis of carbon monoxide content in gases, for example in Hempel's gas apparatus. This application was significant during the nineteenth and early twentieth centuries when coal gas was widely ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

_Chloride_Precipitate.jpg)
