|
Comins' Reagent
The Comins' reagent is a triflyl-donating reagent that is used to synthesize vinyl triflates from the corresponding ketone enolates or dienolates. It was first reported in 1992 by Daniel Comins. The vinyl triflates prepared are useful as substrates in the Suzuki reaction. See also * Bis(trifluoromethanesulfonyl)aniline Bis(trifluoromethanesulfonyl)aniline is the organic compound with the formula C6H5N(SO2CF3)2.{{cite journal, title=N-Phenyltrifluoromethanesulfonimide , last1=Zeller, first1=Wayne E., last2=Schwörer, first2=Ralf, journal=E-EROS Encyclopedia of Re ... References Reagents for organic chemistry Chloropyridines Sulfonamides Trifluoromethyl compounds Substances discovered in the 1990s {{organic-compound-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trifluoromethylsulfonyl
In organic chemistry, the triflyl group (systematic name: trifluoromethanesulfonyl group) is a functional group with the formula and structure . The triflyl group is often represented by –Tf. The related triflate group (trifluoromethanesulfonate) has the formula , and is represented by –OTf. See also * Triflyl azide, TfN3 * Trioctylmethylammonium bis(trifluoromethylsulfonyl)imide, * Comins' reagent * Bis(trifluoromethanesulfonyl)aniline * Triflic anhydride Trifluoromethanesulfonic anhydride, also known as triflic anhydride, is the chemical compound with the formula (CF3SO2)2O. It is the acid anhydride derived from triflic acid. This compound is a strong electrophile, useful for introducing the trif ... (CF3SO2)2O is a very strong triflating agent. References {{organic-chemistry-stub Triflyl compounds Functional groups ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trifluoromethanesulfonate
In organic chemistry, triflate (Preferred IUPAC name, systematic name: trifluoromethanesulfonate), is a functional group with the Chemical formula, formula and Chemical structure, structure . The triflate group is often represented by , as opposed to −Tf, which is the trifluoromethylsulfonyl, triflyl group, . For example, Butyl group, ''n''-butyl triflate can be written as . The corresponding triflate Ion, anion, , is an extremely stable polyatomic ion; this comes from the fact that triflic acid () is a superacid; i.e. it is more acidic than pure sulfuric acid, already one of the strongest acids known. Applications A triflate group is an excellent leaving group used in certain organic reactions such as nucleophilic substitution, Suzuki couplings and Heck reactions. Since alkyl triflates are extremely reactive in SN2 reaction, SN2 reactions, they must be stored in conditions free of nucleophiles (such as water). The anion owes its stability to resonance stabilization which cau ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Suzuki Reaction
The Suzuki reaction is an organic reaction, classified as a cross-coupling reaction, where the coupling partners are a boronic acid and an organohalide and the catalyst is a palladium(0) complex. It was first published in 1979 by Akira Suzuki, and he shared the 2010 Nobel Prize in Chemistry with Richard F. Heck and Ei-ichi Negishi for their contribution to the discovery and development of palladium-catalyzed cross-couplings in organic synthesis. This reaction is also known as the Suzuki–Miyaura reaction or simply as the Suzuki coupling. It is widely used to synthesize poly olefins, styrenes, and substituted biphenyls. Several reviews have been published describing advancements and the development of the Suzuki reaction. The general scheme for the Suzuki reaction is shown below, where a carbon-carbon single bond is formed by coupling a halide (R1-X) with an organoboron species (R2-BY2) using a palladium catalyst and a base. Reaction mechanism The mechanism of the Suzuki r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bis(trifluoromethanesulfonyl)aniline
Bis(trifluoromethanesulfonyl)aniline is the organic compound with the formula C6H5N(SO2CF3)2.{{cite journal, title=N-Phenyltrifluoromethanesulfonimide , last1=Zeller, first1=Wayne E., last2=Schwörer, first2=Ralf, journal=E-EROS Encyclopedia of Reagents for Organic Synthesis, year=2009, doi=10.1002/047084289X.rp142.pub2, isbn=978-0471936237 It is a white solid. The compound is used to install the triflyl group (SO2CF3). Its behavior is akin to that of triflic anhydride, but milder. See also * Comins' reagent The Comins' reagent is a triflyl-donating reagent that is used to synthesize vinyl triflates from the corresponding ketone enolates or dienolates. It was first reported in 1992 by Daniel Comins. The vinyl triflates prepared are useful as sub ..., a related triflating reagent. References Triflates Amides Reagents for organic chemistry Phenyl compounds ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reagents For Organic Chemistry
In chemistry, a reagent ( ) or analytical reagent is a substance or compound added to a system to cause a chemical reaction, or test if one occurs. The terms ''reactant'' and ''reagent'' are often used interchangeably, but reactant specifies a substance ''consumed'' in the course of a chemical reaction. ''Solvents'', though involved in the reaction mechanism, are usually not called reactants. Similarly, ''catalysts'' are not consumed by the reaction, so they are not reactants. In biochemistry, especially in connection with enzyme-catalyzed reactions, the reactants are commonly called substrates. Definitions Organic chemistry In organic chemistry, the term "reagent" denotes a chemical ingredient (a compound or mixture, typically of inorganic or small organic molecules) introduced to cause the desired transformation of an organic substance. Examples include the Collins reagent, Fenton's reagent, and Grignard reagents. Analytical chemistry In analytical chemistry, a reagent ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
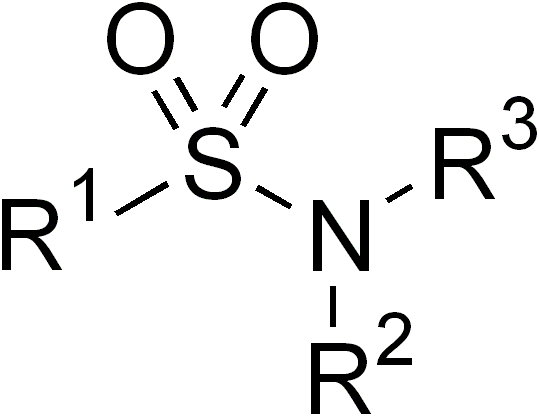
Sulfonamides
In organic chemistry, the sulfonamide functional group (also spelled sulphonamide) is an organosulfur group with the structure . It consists of a sulfonyl group () connected to an amine group (). Relatively speaking this group is unreactive. Because of the rigidity of the functional group, sulfonamides are typically crystalline; for this reason, the formation of a sulfonamide is a classic method to convert an amine into a crystalline derivative which can be identified by its melting point. Many important drugs contain the sulfonamide group. A sulfonamide (compound) is a chemical compound that contains this group. The general formula is or , where each R is some organic group; for example, "methanesulfonamide" (where R = methane, R' = R" = hydrogen) is . Any sulfonamide can be considered as derived from a sulfonic acid by replacing a hydroxyl group () with an amine group. In medicine, the term "sulfonamide" is sometimes used as a synonym for sulfa drug, a derivative or var ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
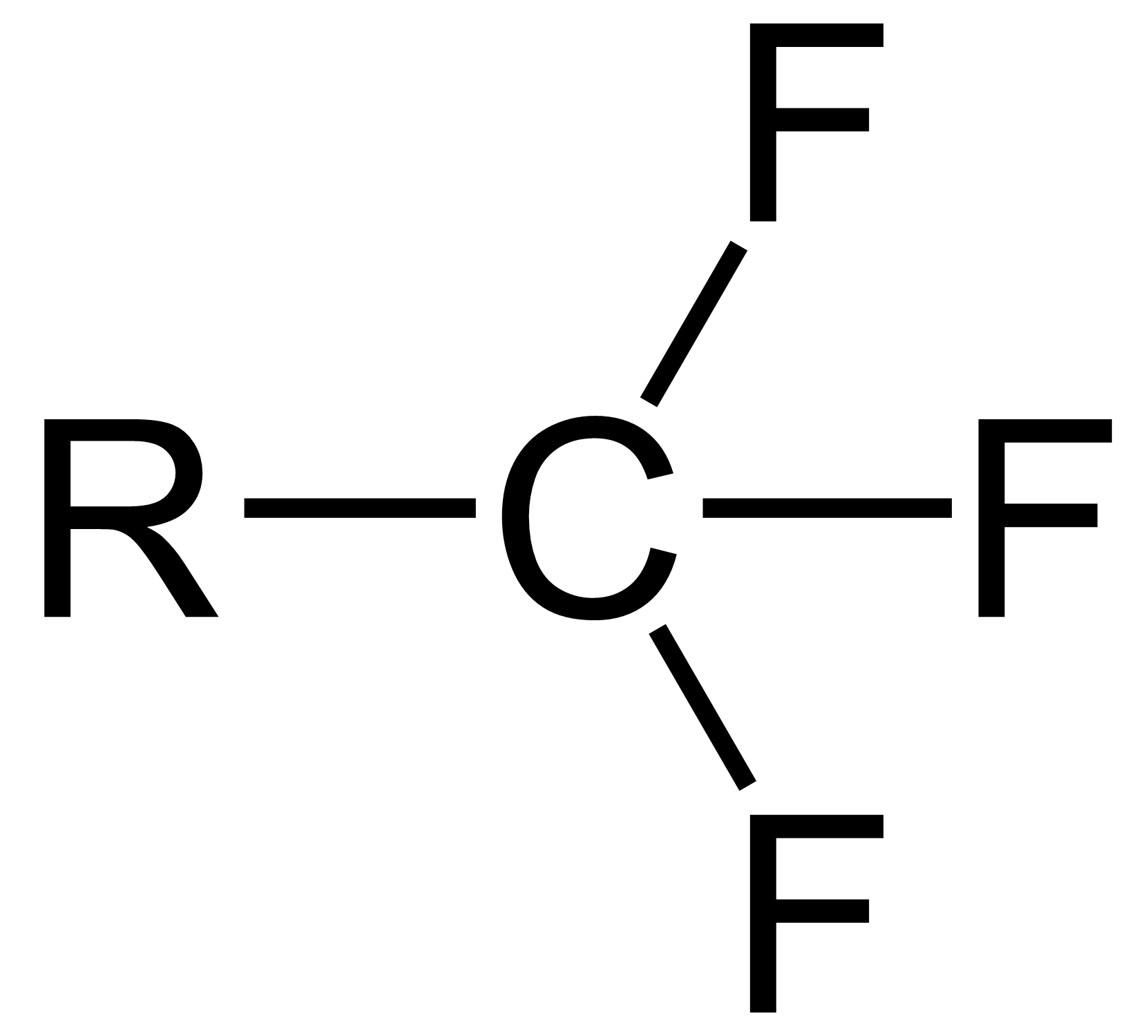
Trifluoromethyl Compounds
The trifluoromethyl group is a functional group that has the formula -CF3. The naming of is group is derived from the methyl group (which has the formula -CH3), by replacing each hydrogen atom by a fluorine atom. Some common examples are trifluoromethane H–, 1,1,1-trifluoroethane –, and hexafluoroacetone –CO–. Compounds with this group are a subclass of the organofluorines. Properties The trifluoromethyl group has a significant electronegativity that is often described as being intermediate between the electronegativities of fluorine and chlorine. For this reason, trifluoromethyl-substituted compounds are often strong acids, such as trifluoromethanesulfonic acid and trifluoroacetic acid. Conversely, the trifluoromethyl group lowers the basicity of compounds like trifluoroethanol. Uses The trifluoromethyl group occurs in certain pharmaceuticals, drugs, and abiotically synthesized natural fluorocarbon based compounds. The medicinal use of the trifloromethyl group dates from ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |