|
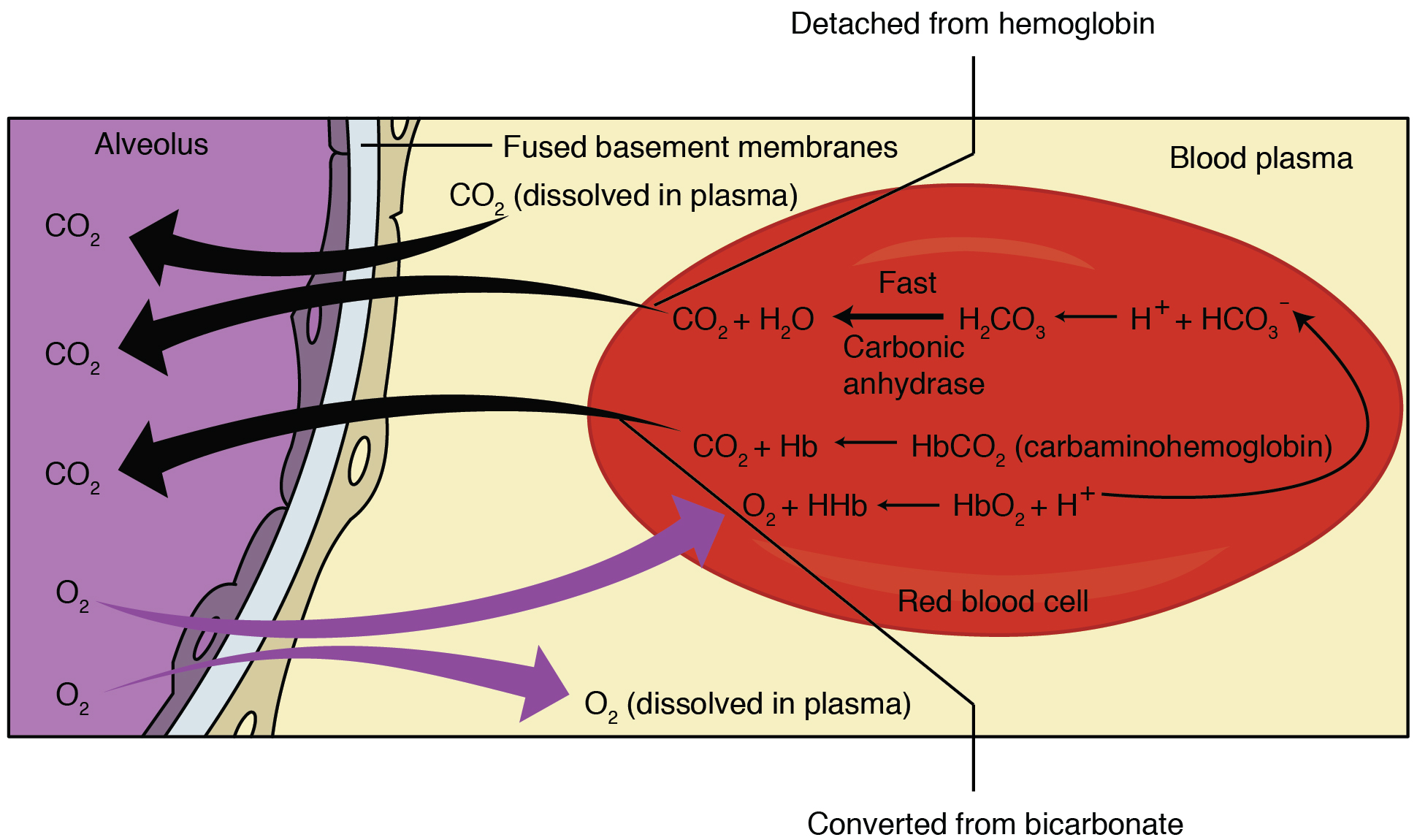
Chloride Shift
Chloride shift (also known as the Hamburger phenomenon or lineas phenomenon, named after Hartog Jakob Hamburger) is a process which occurs in a cardiovascular system and refers to the exchange of bicarbonate (HCO3−) and chloride (Cl−) across the membrane of red blood cells (RBCs). Mechanism Carbon dioxide (CO2) is produced in tissues as a byproduct of normal metabolism. It dissolves in the solution of blood plasma and into red blood cells (RBC), where carbonic anhydrase catalyzes its hydration to carbonic acid (H2CO3). Carbonic acid then spontaneously dissociates to form bicarbonate Ions (HCO3−) and a hydrogen ion (H+). In response to the decrease in intracellular pCO2, more CO2 passively diffuses into the cell. Cell membranes are generally impermeable to charged ions (i.e. H+, HCO3− ) but RBCs are able to exchange bicarbonate for chloride using the anion exchanger protein Band 3. Thus, the rise in intracellular bicarbonate leads to bicarbonate export and chloride intake ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
2319 Fig 23
__NOTOC__ Year 319 ( CCCXIX) was a common year starting on Thursday (link will display the full calendar) of the Julian calendar. At the time, it was known as the Year of the Consulship of Constantinus and Licinius (or, less frequently, year 1072 ''Ab urbe condita''). The denomination 319 for this year has been used since the early medieval period, when the Anno Domini calendar era became the prevalent method in Europe for naming years. Events By place Roman Empire * Emperor Constantine the Great prohibits the separation of the families of slaves, during a change in ownership. India * King Chandragupta I succeeds his father Ghatotkacha, as ruler of the Gupta Empire. Georgia * Christianity is introduced in Colchis, present-day Georgia. By topic Religion * Arius travels to Nicomedia at the invitation of Bishop Eusebius, after having been accused of heresy and condemned by Alexander, the Patriarch of Alexandria. This gives rise to the Arian Controversy. Births ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Ion
A hydrogen ion is created when a hydrogen atom loses or gains an electron. A positively charged hydrogen ion (or proton) can readily combine with other particles and therefore is only seen isolated when it is in a gaseous state or a nearly particle-free space. Due to its extremely high charge density of approximately 2×1010 times that of a sodium ion, the bare hydrogen ion cannot exist freely in solution as it readily hydrates, i.e., bonds quickly. The hydrogen ion is recommended by IUPAC as a general term for all ions of hydrogen and its isotopes. Depending on the charge of the ion, two different classes can be distinguished: positively charged ions and negatively charged ions. Cation (positively charged) A hydrogen atom is made up of a nucleus with charge +1, and a single electron. Therefore, the only positively charged ion possible has charge +1. It is noted H+. Depending on the isotope in question, the hydrogen cation has different names: * Hydron: general name referri ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Allosteric
In biochemistry, allosteric regulation (or allosteric control) is the regulation of an enzyme by binding an effector molecule at a site other than the enzyme's active site. The site to which the effector binds is termed the ''allosteric site'' or ''regulatory site''. Allosteric sites allow effectors to bind to the protein, often resulting in a conformational change and/or a change in protein dynamics. Effectors that enhance the protein's activity are referred to as ''allosteric activators'', whereas those that decrease the protein's activity are called ''allosteric inhibitors''. Allosteric regulations are a natural example of control loops, such as feedback from downstream products or feedforward from upstream substrates. Long-range allostery is especially important in cell signaling. Allosteric regulation is also particularly important in the cell's ability to adjust enzyme activity. The term ''allostery'' comes from the Ancient Greek ''allos'' (), "other", and ''stereos' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as well as with other compounds. Oxygen is Earth's most abundant element, and after hydrogen and helium, it is the third-most abundant element in the universe. At standard temperature and pressure, two atoms of the element bind to form dioxygen, a colorless and odorless diatomic gas with the formula . Diatomic oxygen gas currently constitutes 20.95% of the Earth's atmosphere, though this has changed considerably over long periods of time. Oxygen makes up almost half of the Earth's crust in the form of oxides.Atkins, P.; Jones, L.; Laverman, L. (2016).''Chemical Principles'', 7th edition. Freeman. Many major classes of organic molecules in living organisms contain oxygen atoms, such as proteins, nucleic acids, carbohydrates, and fats, as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hemoglobin
Hemoglobin (haemoglobin BrE) (from the Greek word αἷμα, ''haîma'' 'blood' + Latin ''globus'' 'ball, sphere' + ''-in'') (), abbreviated Hb or Hgb, is the iron-containing oxygen-transport metalloprotein present in red blood cells (erythrocytes) of almost all vertebrates (the exception being the fish family Channichthyidae) as well as the tissues of some invertebrates. Hemoglobin in blood carries oxygen from the respiratory organs (''e.g.'' lungs or gills) to the rest of the body (''i.e.'' tissues). There it releases the oxygen to permit aerobic respiration to provide energy to power functions of an organism in the process called metabolism. A healthy individual human has 12to 20grams of hemoglobin in every 100mL of blood. In mammals, the chromoprotein makes up about 96% of the red blood cells' dry content (by weight), and around 35% of the total content (including water). Hemoglobin has an oxygen-binding capacity of 1.34mL O2 per gram, which increases the total blood oxygen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Haldane Effect
The Haldane effect is a property of hemoglobin first described by John Scott Haldane, within which oxygenation of blood in the lungs displaces carbon dioxide from hemoglobin, increasing the removal of carbon dioxide. Consequently, oxygenated blood has a reduced affinity for carbon dioxide. Thus, the Haldane effect describes the ability of hemoglobin to carry increased amounts of carbon dioxide (CO2) in the deoxygenated state as opposed to the oxygenated state. A high concentration of CO2 facilitates dissociation of oxyhemoglobin. Carbaminohemoglobin Carbon dioxide travels through the blood in three different ways. One of these ways is by binding to amino groups, creating carbamino compounds. Amino groups are available for binding at the N-terminals and at side-chains of arginine and lysine residues in hemoglobin. When carbon dioxide binds to these residues carbaminohemoglobin is formed. This amount of carbaminohemoglobin formed is inversely proportional to the amount of oxygen at ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Band 3
Band 3 anion transport protein, also known as anion exchanger 1 (AE1) or band 3 or solute carrier family 4 member 1 (SLC4A1), is a protein that is encoded by the gene in humans. Band 3 anion transport protein is a phylogenetically-preserved transport protein responsible for mediating the exchange of chloride (Cl−) with bicarbonate (HCO3−) across plasma membranes. Functionally similar members of the AE clade are AE2 and AE3. Function Band 3 is present in the basolateral face of the α-intercalated cells of the collecting ducts of the nephron, which are the main acid-secreting cells of the kidney. They generate hydrogen ions and bicarbonate ions from carbon dioxide and water – a reaction catalysed by carbonic anhydrase. The hydrogen ions are pumped into the collecting duct tubule by vacuolar H+ ATPase, the apical proton pump, which thus excretes acid into the urine. kAE1 exchanges bicarbonate for chloride on the basolateral surface, essentially returning bicarbonate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
PCO2
''p''CO2, pCO2, or P_\ceis the partial pressure of carbon dioxide (CO2), often used in reference to blood but also used in meteorology, climate science, oceanography, and limnology to describe the fractional pressure of CO2 as a function of its concentration in gas or dissolved phases. The units of ''p''CO2 are mmHg, atm, torr, Pa, or any other standard unit of atmospheric pressure. The ''p''CO2 of Earth's atmosphere has risen from approximately 280 ppm (parts-per-million) to a mean 2019 value of 409.8 ppm as a result of anthropogenic release of carbon dioxide from fossil fuel burning. This is the highest atmospheric concentration to have existed on Earth for at least the last 800,000 years. Medicine In medicine, the partial pressure of carbon dioxide in arterial blood is called P_ or PaCO2. Measurement of P_ in the systemic circulation indicates the effectiveness of ventilation at the lungs' alveoli, given the diffusing capacity of the gas. It is a good indicator of res ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dissociate
Dissociation in chemistry is a general process in which molecules (or ionic compounds such as salts, or complexes) separate or split into other things such as atoms, ions, or radicals, usually in a reversible manner. For instance, when an acid dissolves in water, a covalent bond between an electronegative atom and a hydrogen atom is broken by heterolytic fission, which gives a proton (H+) and a negative ion. Dissociation is the opposite of association or recombination. Dissociation constant For reversible dissociations in a chemical equilibrium :AB A + B the dissociation constant ''K''d is the ratio of dissociated to undissociated compound :K_d = \mathrm where the brackets denote the equilibrium concentrations of the species. Dissociation degree The dissociation degree \alpha is the fraction of original solute molecules that have dissociated. It is usually indicated by the Greek symbol α. More accurately, degree of dissociation refers to the amount of solute dissociated int ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hartog Jakob Hamburger
Hartog Jakob or Hartog Jacob Hamburger (9 March 1859 – 4 January 1924) was a Dutch physiologist, born in Alkmaar. After completing the Hogere Burgerschool in Alkmaar, Hamburger studied chemistry at Utrecht University, where he received his doctorate in 1883, on the determination of urea in urine. He subsequently worked with Utrecht ophthalmologist and physiologist Franciscus Cornelis Donders for seven years, and completed a medical degree. From 1888 he lectured in physiology and pathology at the National Veterinary School, also in Utrecht. In 1896, he invented the crystalloid solution known as Hamburger's solution or normal saline. Based on plant-based experiments by botanist Hugo de Vries, he developed a salt solution that was thought to have the same osmolality as human blood and therefore did not cause haemolysis of red blood cells. It is uncertain whether the saline was ever originally intended for intravenous administration. In 1901 he joined the University of Groningen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbonic Anhydrase
The carbonic anhydrases (or carbonate dehydratases) () form a family of enzymes that catalyze the interconversion between carbon dioxide and water and the dissociated ions of carbonic acid (i.e. bicarbonate and hydrogen ions). The active site of most carbonic anhydrases contains a zinc ion. They are therefore classified as metalloenzymes. The enzyme maintains acid-base balance and helps transport carbon dioxide. Carbonic anhydrase helps maintain acid–base homeostasis, regulate pH, and fluid balance. Depending on its location, the role of the enzyme changes slightly. For example, carbonic anhydrase produces acid in the stomach lining. In the kidney, the control of bicarbonate ions influences the water content of the cell. The control of bicarbonate ions also influences the water content in the eyes. Inhibitors of carbonic anhydrase are used to treat glaucoma, the excessive build-up of water in the eyes. Blocking this enzyme shifts the fluid balance in the eyes to reduce flui ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |