|
Chain Transfer
Chain transfer is a polymerization reaction by which the activity of a growing polymer chain is transferred to another molecule. :P• + XR' → PX + R'• Chain transfer reactions reduce the average molecular weight of the final polymer. Chain transfer can be either introduced deliberately into a polymerization (by use of a ''chain transfer agent'') or it may be an unavoidable side-reaction with various components of the polymerization. Chain transfer reactions occur in most forms of addition polymerization including radical polymerization, ring-opening polymerization, coordination polymerization, and cationic polymerization, as well as anionic polymerization. Types Chain transfer reactions are usually categorized by the nature of the molecule that reacts with the growing chain. * Transfer to chain transfer agent. Chain transfer agents have at least one weak chemical bond, which therefore facilitates the chain transfer reaction. Common chain transfer agents include thiols, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polymerization
In polymer chemistry, polymerization (American English), or polymerisation (British English), is a process of reacting monomer molecules together in a chemical reaction to form polymer chains or three-dimensional networks. There are many forms of polymerization and different systems exist to categorize them. In chemical compounds, polymerization can occur via a variety of reaction mechanisms that vary in complexity due to the functional groups present in the reactants and their inherent steric effects. In more straightforward polymerizations, alkenes form polymers through relatively simple radical reactions; in contrast, reactions involving substitution at a carbonyl group require more complex synthesis due to the way in which reactants polymerize. Alkanes can also be polymerized, but only with the help of strong acids. As alkenes can polymerize in somewhat straightforward radical reactions, they form useful compounds such as polyethylene and polyvinyl chloride (PVC), w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Halocarbons
Halocarbon compounds are chemicals in which one or more carbon atoms are linked by covalent bonds with one or more halogen atoms (fluorine, chlorine, bromine or iodine – ) resulting in the formation of organofluorine compounds, organochlorine compounds, organobromine compounds, and organoiodine compounds. Chlorine halocarbons are the most common and are called organochlorides. Many synthetic organic compounds such as plastic polymers, and a few natural ones, contain halogen atoms; they are known as ''halogenated'' compounds or ''organohalogens''. Organochlorides are the most common industrially used organohalides, although the other organohalides are used commonly in organic synthesis. Except for extremely rare cases, organohalides are not produced biologically, but many pharmaceuticals are organohalides. Notably, many pharmaceuticals such as Prozac have trifluoromethyl groups. For information on inorganic halide chemistry, see halide. Chemical families Halocarbons are t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethylene
Ethylene ( IUPAC name: ethene) is a hydrocarbon which has the formula or . It is a colourless, flammable gas with a faint "sweet and musky" odour when pure. It is the simplest alkene (a hydrocarbon with carbon-carbon double bonds). Ethylene is widely used in the chemical industry, and its worldwide production (over 150 million tonnes in 2016) exceeds that of any other organic compound. Much of this production goes toward polyethylene, a widely used plastic containing polymer chains of ethylene units in various chain lengths. Ethylene is also an important natural plant hormone and is used in agriculture to force the ripening of fruits. The hydrate of ethylene is ethanol. Structure and properties This hydrocarbon has four hydrogen atoms bound to a pair of carbon atoms that are connected by a double bond. All six atoms that comprise ethylene are coplanar. The H-C-H angle is 117.4°, close to the 120° for ideal sp² hybridized carbon. The molecule is also relatively weak: ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
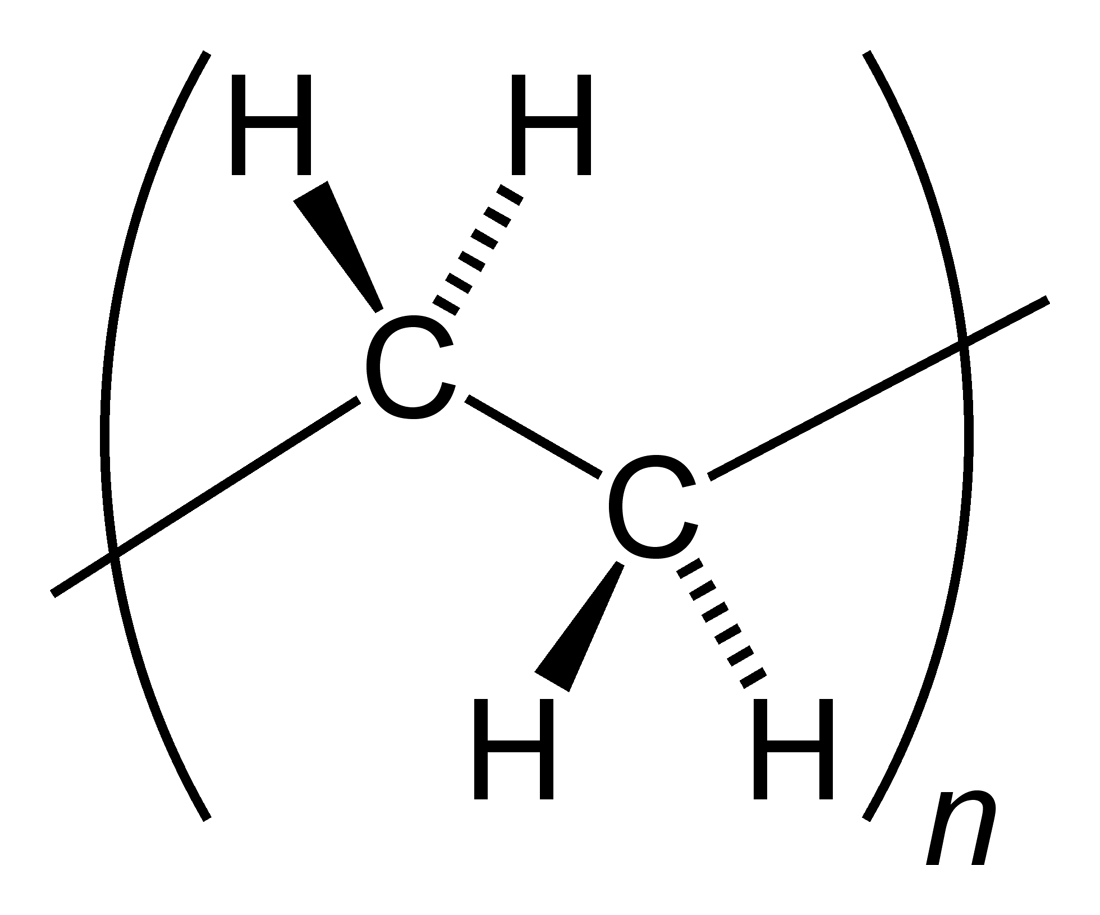
Polyethylene
Polyethylene or polythene (abbreviated PE; IUPAC name polyethene or poly(methylene)) is the most commonly produced plastic. It is a polymer, primarily used for packaging (plastic bags, plastic films, geomembranes and containers including bottles, etc.). , over 100 million tonnes of polyethylene resins are being produced annually, accounting for 34% of the total plastics market. Many kinds of polyethylene are known, with most having the chemical formula (C2H4)''n''. PE is usually a mixture of similar polymers of ethylene, with various values of ''n''. It can be ''low-density'' or ''high-density'': low-density polyethylene is extruded using high pressure () and high temperature (), while high-density polyethylene is extruded using low pressure () and low temperature (). Polyethylene is usually thermoplastic, but it can be modified to become thermosetting instead, for example, in cross-linked polyethylene. History Polyethylene was first synthesized by the German ch ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hugh Stott Taylor
Sir Hugh Stott Taylor (6 February 1890 – 17 April 1974) was an English chemist primarily interested in catalysis.Who Was Who, Published by A&C Black Limited In 1925, in a landmark contribution to catalytic theory, Taylor suggested that a catalysed chemical reaction is not catalysed over the entire solid surface of the catalyst but only at certain ' active sites' or centres. He also developed important methods for procuring heavy water during World War II and pioneered the use of stable isotopes in studying chemical reactions. Early life Taylor was born in St Helens, Lancashire, England in 1890, the son of glass technologist James and Ellen (née Stott) Taylor. He was educated at Cowley Grammar School in St Helens and then attended the University of Liverpool, where he received his BSc in 1909 and his MSc in 1910. Taylor then carried out three years of graduate work in Liverpool, after which he spent one year at the Nobel Institute in Stockholm in the laboratory of Svan ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Termination - Chain Transfer - Solvent
Termination may refer to: Science *Termination (geomorphology), the period of time of relatively rapid change from cold, glacial conditions to warm interglacial condition *Termination factor, in genetics, part of the process of transcribing RNA * Termination type, in lithic reduction, a characteristic indicating the manner in which the distal end of a lithic flake detaches from a core *Chain termination, in chemistry, a chemical reaction which halts polymerization * Termination shock, in solar studies, a feature of the heliosphere *Terminating computation, in computer science ** Termination analysis, a form of program analysis in computer science **Termination proof, a mathematical proof concerning the termination of a program **Termination (term rewriting), in particular for term rewriting systems Technology *Electrical termination, ending a wire or cable properly to prevent interference *Termination of wires to a **Crimp connection **Electrical connector ** Solder joint * Abor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oligomers
In chemistry and biochemistry, an oligomer () is a molecule that consists of a few repeating units which could be derived, actually or conceptually, from smaller molecules, monomers.Quote: ''Oligomer molecule: A molecule of intermediate relative molecular mass, the structure of which essentially comprises a small plurality of units derived, actually or conceptually, from molecules of lower relative molecular mass.'' The name is composed of Greek elements '' oligo-'', "a few" and '' -mer'', "parts". An adjective form is ''oligomeric''. The oligomer concept is contrasted to that of a polymer, which is usually understood to have a large number of units, possibly thousands or millions. However, there is no sharp distinction between these two concepts. One proposed criterion is whether the molecule's properties vary significantly with the removal of one or a few of the units. An oligomer with a specific number of units is referred to by the Greek prefix denoting that number, w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemically Inert
In chemistry, the term chemically inert is used to describe a substance that is not chemically reactive. From a thermodynamic perspective, a substance is inert, or nonlabile, if it is thermodynamically unstable (positive standard Gibbs free energy of formation) yet decomposes at a slow, or negligible rate. Most of the noble gases, which appear in the last column of the periodic table, are classified as inert (or unreactive). These elements are stable in their naturally occurring form (gaseous form) and they are called inert gases. Noble gas The noble gases ( helium, neon, argon, krypton, xenon and radon) were previously known as 'inert gases' because of their perceived lack of participation in any chemical reactions. The reason for this is that their outermost electron shells (valence shells) are completely filled, so that they have little tendency to gain or lose electrons. They are said to acquire a noble gas configuration, or a full electron configuration. It is no ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solution Polymerization
Solution polymerization is a method of industrial polymerization. In this procedure, a monomer is dissolved in a non-reactive solvent that contains a catalyst or initiator. The reaction results in a polymer which is also soluble in the chosen solvent. Heat released by the reaction is absorbed by the solvent, reducing the reaction rate. Moreover, the viscosity of the reaction mixture is reduced, preventing autoacceleration at high monomer concentrations. A decrease in viscosity of the reaction mixture by dilution also aids heat transfer, one of the major issues connected with polymer production, since most polymerizations are exothermic reactions. Once the desired conversion is reached, excess solvent must be removed to obtain the pure polymer. Accordingly, solution polymerization is primarily used in applications where the presence of a solvent is desired anyway, as is the case for varnish and adhesives. Another application of polymer solutions includes the manufacture of fibers b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Termination - Chain Transfer - Polymer
Termination may refer to: Science *Termination (geomorphology), the period of time of relatively rapid change from cold, glacial conditions to warm interglacial condition *Termination factor, in genetics, part of the process of transcribing RNA *Termination type, in lithic reduction, a characteristic indicating the manner in which the distal end of a lithic flake detaches from a core *Chain termination, in chemistry, a chemical reaction which halts polymerization *Termination shock, in solar studies, a feature of the heliosphere *Terminating computation, in computer science **Termination analysis, a form of program analysis in computer science **Termination proof, a mathematical proof concerning the termination of a program **Termination (term rewriting), in particular for term rewriting systems Technology *Electrical termination, ending a wire or cable properly to prevent interference *Termination of wires to a **Crimp connection **Electrical connector ** Solder joint * Abort ( ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Low-density Polyethylene
Low-density polyethylene (LDPE) is a thermoplastic made from the monomer ethylene. It was the first grade of polyethylene, produced in 1933 by Imperial Chemical Industries (ICI) using a high pressure process via free radical polymerization. Its manufacture employs the same method today. The EPA estimates 5.7% of LDPE ( resin identification code 4) is recycled in the United States. Despite competition from more modern polymers, LDPE continues to be an important plastic grade. In 2013 the worldwide LDPE market reached a volume of about US$33 billion. Despite its designation with the recycling symbol, it cannot be as commonly recycled as No. 1 (polyethylene terephthalate) or 2 plastics (high-density polyethylene). Properties LDPE is defined by a density range of 917–930 kg/m3. At room temperature it is not reactive, except to strong oxidizers; some solvents cause it to swell. It can withstand temperatures of continuously and for a short time. Made in translucent and opaqu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Branching (polymer Chemistry)
In polymer chemistry, branching is the regular or irregular attachment of side chains to a polymer's backbone chain. It occurs by the replacement of a substituent (e.g. a hydrogen atom) on a monomer subunit by another covalently-bonded chain of that polymer; or, in the case of a graft copolymer, by a chain of another type. Branched polymers have more compact and symmetrical molecular conformations, and exhibit intra-heterogeneous dynamical behavior with respect to the unbranched polymers. In crosslinking rubber by vulcanization, short sulfur branches link polyisoprene chains (or a synthetic variant) into a multiple-branched thermosetting elastomer. Rubber can also be so completely vulcanized that it becomes a rigid solid, so hard it can be used as the bit in a smoking pipe. Polycarbonate chains can be crosslinked to form the hardest, most impact-resistant thermosetting plastic, used in safety glasses. Branching may result from the formation of carbon-carbon or various ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |