|
Borole
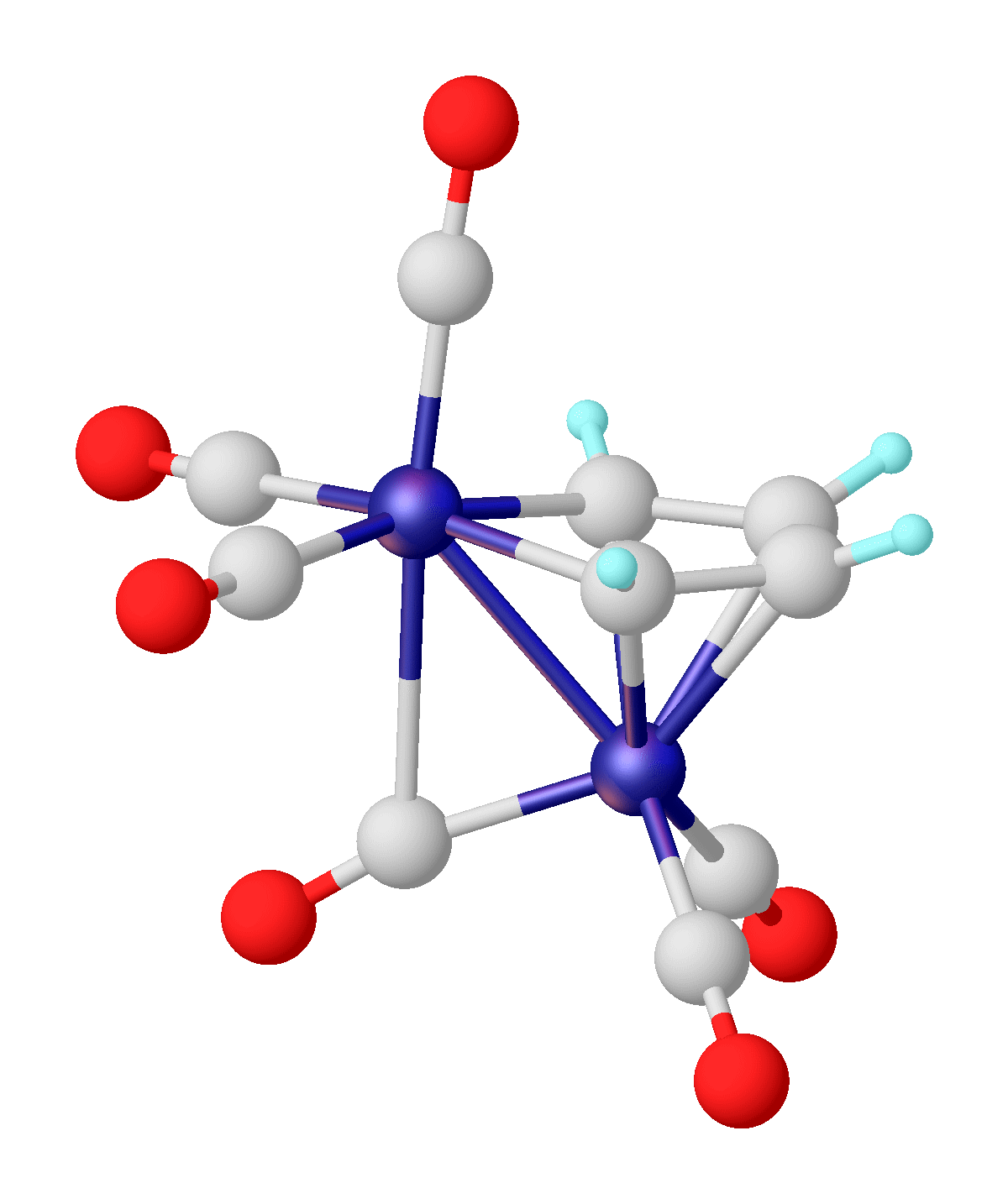
Boroles represent a class of molecules known as metalloles, which are heterocyclic 5-membered rings. As such, they can be viewed as structural analogs of cyclopentadiene, pyrrole or furan, with boron replacing a carbon, nitrogen and oxygen atom respectively. They are isoelectronic with the cyclopentadienyl cation and comprise four π electrons. Although Hückel's rule cannot be strictly applied to borole, it is considered to be antiaromatic due to having 4 π electrons. As a result, boroles exhibit unique electronic properties not found in other metalloles. The parent unsubstituted compound with the chemical formula has yet to be isolated outside a coordination sphere of transition metals. Substituted derivatives, which have been synthesized, can have various substituents at the 4 carbons and boron. The high electron deficiency leads to various reactivities such as metal free hydrogen activation and rearrangements upon cycloaddition which are unobserved in other structural analog ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Borole Homolumo
Boroles represent a class of molecules known as metalloles, which are heterocyclic 5-membered rings. As such, they can be viewed as structural analogs of cyclopentadiene, pyrrole or furan, with boron replacing a carbon, nitrogen and oxygen atom respectively. They are isoelectronic with the cyclopentadienyl cation and comprise four π electrons. Although Hückel's rule cannot be strictly applied to borole, it is considered to be antiaromatic due to having 4 π electrons. As a result, boroles exhibit unique electronic properties not found in other metalloles. The parent unsubstituted compound with the chemical formula has yet to be isolated outside a coordination sphere of transition metals. Substituted derivatives, which have been synthesized, can have various substituents at the 4 carbons and boron. The high electron deficiency leads to various reactivities such as metal free hydrogen activation and rearrangements upon cycloaddition which are unobserved in other structural analo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Borole Vs Cp+
Boroles represent a class of molecules known as metalloles, which are heterocyclic 5-membered rings. As such, they can be viewed as structural analogs of cyclopentadiene, pyrrole or furan, with boron replacing a carbon, nitrogen and oxygen atom respectively. They are isoelectronic with the cyclopentadienyl cation and comprise four π electrons. Although Hückel's rule cannot be strictly applied to borole, it is considered to be antiaromatic due to having 4 π electrons. As a result, boroles exhibit unique electronic properties not found in other metalloles. The parent unsubstituted compound with the chemical formula has yet to be isolated outside a coordination sphere of transition metals. Substituted derivatives, which have been synthesized, can have various substituents at the 4 carbons and boron. The high electron deficiency leads to various reactivities such as metal free hydrogen activation and rearrangements upon cycloaddition which are unobserved in other structural analo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Boron
Boron is a chemical element with the symbol B and atomic number 5. In its crystalline form it is a brittle, dark, lustrous metalloid; in its amorphous form it is a brown powder. As the lightest element of the ''boron group'' it has three valence electrons for forming covalent bonds, resulting in many compounds such as boric acid, the mineral borax, sodium borate, and the ultra-hard crystals of boron carbide and boron nitride. Boron is synthesized entirely by cosmic ray spallation and supernovae and not by stellar nucleosynthesis, so it is a low-abundance element in the Solar System and in the Crust (geology), Earth's crust. It constitutes about 0.001 percent by weight of Earth's crust. It is concentrated on Earth by the water-solubility of its more common naturally occurring compounds, the borate minerals. These are mined industrially as evaporites, such as borax and kernite. The largest known deposits are in Turkey, the largest producer of boron minerals. Elemental b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metallole
Metalloles are derivatives of cyclopentadiene in which the carbon atom at position 5, the saturated carbon, is replaced by a heteroatom. In contrast to its parent compound, the numbering of the metallole starts at the heteroatom. Some of these compounds are described as organometallic compounds, but in the list below quite a number of metalloids are present too. Many metalloles are fluorescent. Polymeric derivatives of pyrrole and thiophene are of interest in molecular electronics. Metalloles, which can also be viewed as structural analogs of pyrrole, include: * Arsole, a moderately-aromatic arsenic analog * Bismole, a bismuth analog * Borole, a boron analog * Furan (oxole), an oxygen analog * Gallole, a gallium analog * Germole, a germanium analog * Phosphole, a phosphorus analog * Pyrrole (azole), a nitrogen analog * Selenophene, a selenium analog * Silole, a silicon analog * Stannole, a tin analog * Stibole, an antimony analog * Tellurophene, a tellurium analog *Plumbole, a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Redshift
In physics, a redshift is an increase in the wavelength, and corresponding decrease in the frequency and photon energy, of electromagnetic radiation (such as light). The opposite change, a decrease in wavelength and simultaneous increase in frequency and energy, is known as a negative redshift, or blueshift. The terms derive from the colours red and blue which form the extremes of the visible light spectrum. In astronomy and cosmology, the three main causes of electromagnetic redshift are # The radiation travels between objects which are moving apart (" relativistic" redshift, an example of the relativistic Doppler effect) #The radiation travels towards an object in a weaker gravitational potential, i.e. towards an object in less strongly curved (flatter) spacetime (gravitational redshift) #The radiation travels through expanding space (cosmological redshift). The observation that all sufficiently distant light sources show redshift corresponding to their distance from Earth ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Synthesis Of Boroles
Synthesis or synthesize may refer to: Science Chemistry and biochemistry *Chemical synthesis, the execution of chemical reactions to form a more complex molecule from chemical precursors **Organic synthesis, the chemical synthesis of organic compounds ***Total synthesis, the complete organic synthesis of complex organic compounds, usually without the aid of biological processes ***Convergent synthesis or linear synthesis, a strategy to improve the efficiency of multi-step chemical syntheses **Dehydration synthesis, a chemical synthesis resulting in the loss of a water molecule *Biosynthesis, the creation of an organic compound in a living organism, usually aided by enzymes **Photosynthesis, a biochemical reaction using a carbon molecule to produce an organic molecule, using sunlight as a catalyst **Chemosynthesis, the synthesis of biological compounds into organic waste, using methane or an oxidized molecule as a catalyst **Amino acid synthesis, the synthesis of an amino acid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Francis Gordon Albert Stone
Francis Gordon Albert Stone CBE, FRS, FRSC (19 May 1925 – 6 April 2011), always known as Gordon, was a British chemist who was a prolific and decorated scholar. He specialized in the synthesis of main group and transition metal organometallic compounds. He was the author of more than 900 academic publications resulting in an h-index of 72 in 2011. Early life Gordon Stone was born in Exeter, Devon in 1925, the only child of Sidney Charles Stone, a civil servant, and Florence Beatrice Stone (née Coles).Bristol University ObituaryF Gordon A Stone retrieved 08/03/2012 He received his B.A. in 1948 and Ph.D. in 1951, both from Christ's College, Cambridge (Cambridge University), England, where he studied under Harry Julius Emeléus. Academic life After graduating from Christ's College, Cambridge, he was a Fulbright Scholar at the University of Southern California for two years, before being appointed as an instructor in the Chemistry Department at Harvard University, and wa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stannole
Stannole is an organotin compound with the formula ( C H)4 SnH2. It is classified as a metallole, i.e. an unsaturated five-membered ring containing a heteroatom. It is a structural analog of pyrrole, with tin replacing the nitrogen. Substituted derivatives, which have been synthesized, are also called stannoles. 1λ2-Stannole has formula C4H4Sn, with no hydrogen on the tin atom, which is in the +2 oxidation state. Examples 1,1-Dibutylstannole is a pale yellow oil prepared from 1,4-dilithio-1,3-butadiene and dibutyltin dichloride. : Reactions 1,1-Dimethyl-2,3,4,5-tetraphenyl-1''H''-stannole, for example, can be formed by the reaction of 1,4-dilithio-1,2,3,4-tetraphenyl-1,3-butadiene and dimethyltin dichloride. 1,1-Disubstituted stannoles can be formed in the +2+1cycloaddition reaction of two acetylene molecules with an organotin molecule SnR2. See also *Organotin chemistry Organotin compounds or stannanes are chemical compounds based on tin with hydrocarbon substituents. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Resonance In Borole 2-
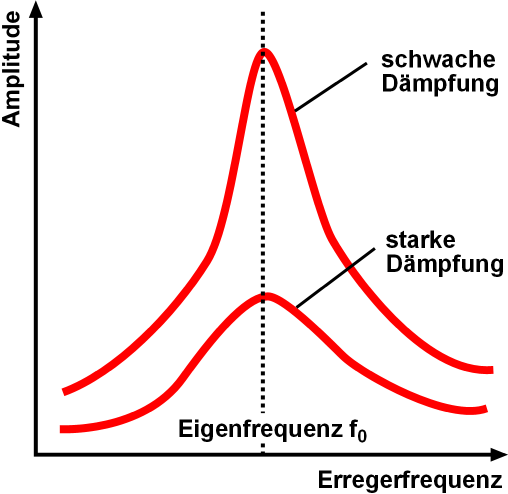
Resonance describes the phenomenon of increased amplitude that occurs when the frequency of an applied periodic force (or a Fourier component of it) is equal or close to a natural frequency of the system on which it acts. When an oscillating force is applied at a resonant frequency of a dynamic system, the system will oscillate at a higher amplitude than when the same force is applied at other, non-resonant frequencies. Frequencies at which the response amplitude is a relative maximum are also known as resonant frequencies or resonance frequencies of the system. Small periodic forces that are near a resonant frequency of the system have the ability to produce large amplitude oscillations in the system due to the storage of vibrational energy. Resonance phenomena occur with all types of vibrations or waves: there is mechanical resonance, orbital resonance, acoustic resonance, electromagnetic resonance, nuclear magnetic resonance (NMR), electron spin resonance (ESR) and reson ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Resonance (chemistry)
In chemistry, resonance, also called mesomerism, is a way of describing bonding in certain molecules or polyatomic ions by the combination of several contributing structures (or ''forms'', also variously known as ''resonance structures'' or ''canonical structures'') into a resonance hybrid (or ''hybrid structure'') in valence bond theory. It has particular value for analyzing delocalized electrons where the bonding cannot be expressed by one single Lewis structure. Overview Under the framework of valence bond theory, resonance is an extension of the idea that the bonding in a chemical species can be described by a Lewis structure. For many chemical species, a single Lewis structure, consisting of atoms obeying the octet rule, possibly bearing formal charges, and connected by bonds of positive integer order, is sufficient for describing the chemical bonding and rationalizing experimentally determined molecular properties like bond lengths, angles, and dipole moment. Howev ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aromaticity
In chemistry, aromaticity is a chemical property of cyclic ( ring-shaped), ''typically'' planar (flat) molecular structures with pi bonds in resonance (those containing delocalized electrons) that gives increased stability compared to saturated compounds having single bonds, and other geometric or connective non-cyclic arrangements with the same set of atoms. Aromatic rings are very stable and do not break apart easily. Organic compounds that are not aromatic are classified as aliphatic compounds—they might be cyclic, but only aromatic rings have enhanced stability. The term ''aromaticity'' with this meaning is historically related to the concept of having an aroma, but is a distinct property from that meaning. Since the most common aromatic compounds are derivatives of benzene (an aromatic hydrocarbon common in petroleum and its distillates), the word ''aromatic'' occasionally refers informally to benzene derivatives, and so it was first defined. Nevertheless, many non-be ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lewis Acids And Bases
A Lewis acid (named for the American physical chemist Gilbert N. Lewis) is a chemical species that contains an empty orbital which is capable of accepting an electron pair from a Lewis base to form a Lewis adduct. A Lewis base, then, is any species that has a filled orbital containing an electron pair which is not involved in bonding but may form a dative bond with a Lewis acid to form a Lewis adduct. For example, NH3 is a Lewis base, because it can donate its lone pair of electrons. Trimethylborane (Me3B) is a Lewis acid as it is capable of accepting a lone pair. In a Lewis adduct, the Lewis acid and base share an electron pair furnished by the Lewis base, forming a dative bond. In the context of a specific chemical reaction between NH3 and Me3B, a lone pair from NH3 will form a dative bond with the empty orbital of Me3B to form an adduct NH3•BMe3. The terminology refers to the contributions of Gilbert N. Lewis. From p. 142: "We are inclined to think of substances as pos ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |


.png)
