|
Bis(allyl)nickel
Bis(allyl)nickel is an organonickel compound with the formula Ni(η3-C3H5)2. The molecule consists of two allyl ligands bound to nickel(II). It has inversion symmetry. It is a volatile yellow liquid. Preparation and reactions It can be prepared by the reaction of allyl magnesium bromide with anhydrous nickel chloride. It was first prepared similarly by Gunther Wilke et al. The same group reported that the complex react with carbon monoxide to give nickel tetracarbonyl and 1,5-hexadiene. It catalyzes the trimerization of butadiene 1,3-Butadiene () is the organic compound with the formula (CH2=CH)2. It is a colorless gas that is easily condensed to a liquid. It is important industrially as a precursor to synthetic rubber. The molecule can be viewed as the union of two viny .... With tertiary phosphines, the complex gives the tetrakis derivative. Such reactions to proceed via the intermediacy of the 18-electron adduct. : : : References {{Reflist Organometallic chemist ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Transition Metal Allyl Complex
Transition-metal allyl complexes are coordination complexes with allyl and its derivatives as ligands. Allyl is the radical with the connectivity CH2CHCH2, although as a ligand it is usually viewed as an allyl anion CH2=CH−CH2−, which is usually described as two equivalent resonance structures. Examples The allyl ligand is commonly found in organometallic chemistry. Most commonly, allyl ligands bind to metals via all three carbon atoms, the η3-binding mode. The η3-allyl group is classified as an LX-type ligand in the Green LXZ ligand classification scheme, serving as a 3e– donor using neutral electron counting and 4e– donor using ionic electron counting. More common are complexes with allyl and other ligands. Examples include (η3-allyl)Mn(CO)4 and CpPd(allyl). Homoleptic complexes * bis(allyl)nickel * bis(allyl)palladium * bis(allyl)platinum *tris(allyl)chromium * tris(allyl)rhodium * tris(allyl)iridium Synthetic methods Allyl complexes are often generated by ox ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organonickel Compound
Organonickel chemistry is a branch of organometallic chemistry that deals with organic compounds featuring nickel-carbon bonds. They are used as a catalyst, as a building block in organic chemistry and in chemical vapor deposition. Organonickel compounds are also short-lived intermediates in organic reactions. The first organonickel compound was nickel tetracarbonyl Ni(CO)4, reported in 1890 and quickly applied in the Mond process for nickel purification. Organonickel complexes are prominent in numerous industrial processes including carbonylations, hydrocyanation, and the Shell higher olefin process. Classes of compounds : Alkyl and aryl complexes A popular reagent is Ni(CH3)2(tetramethylethylenediamine). Many alkyl and aryl complexes are known with the formula NiR(X)L2. Examples include dppf)Ni(cinnamyl)Cl) ''trans''-(PCy2Ph)2Ni(''o''-tolyl)Cl, (dppf)Ni(''o''-tolyl)Cl, (TMEDA)Ni(''o''-tolyl)Cl, and (TMEDA)NiMe2. Nickel compounds of the type NiR2 also exist with just 12 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
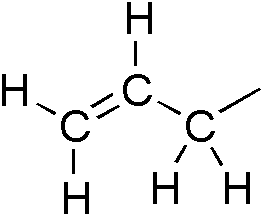
Allyl Magnesium Bromide
Allylmagnesium bromide is a Grignard reagent used for introducing the allyl group. It is commonly available as a solution in diethyl ether. It may be synthesized by treatment of magnesium with allyl bromide Allyl bromide (3-bromopropene) is an organic halide. It is an alkylating agent used in synthesis of polymers, pharmaceuticals, synthetic perfumes and other organic compounds. Physically, allyl bromide is a colorless liquid with an irritating and p ... while maintaining the reaction temperature below 0 °C to suppress formation of hexadiene. Allyl chloride can also be used in place of the bromide to give allylmagnesium chloride. These reagents are used to prepare metal allyl complexes. References Further reading * {{cite book , author = Chabot, P. , editor1=Rakita, P. E. , editor2=Silverman, G. , chapter = 7. Infrared and Raman Spectroscopy , title = Handbook of Grignard Reagents , year = 1996 , pages = 93–102 , location = New York, N.Y. , publisher = Marcel Dekke ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nickel Chloride
Nickel(II) chloride (or just nickel chloride) is the chemical compound NiCl2. The anhydrous salt is yellow, but the more familiar hydrate NiCl2·6H2O is green. Nickel(II) chloride, in various forms, is the most important source of nickel for chemical synthesis. The nickel chlorides are deliquescent, absorbing moisture from the air to form a solution. Nickel salts have been shown to be carcinogenic to the lungs and nasal passages in cases of long-term inhalation exposure. Production and syntheses The largest scale production of nickel chloride involves the extraction with hydrochloric acid of nickel matte and residues obtained from roasting refining nickel-containing ores. Nickel chloride is not usually prepared in the laboratory because it is inexpensive and has a long shelf-life. Heating the hexahydrate in the range 66–133.°C gives the yellowish dihydrate, NiCl2·2H2O. The hydrates convert to the anhydrous form upon heating in thionyl chloride or by heating under ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gunther Wilke
Gundaharius or Gundahar (died 437), better known by his legendary names Gunther ( gmh, Gunther) or Gunnar ( non, Gunnarr), was a historical king of Burgundy in the early 5th century. Gundahar is attested as ruling his people shortly after they crossed the Rhine into Roman Gaul. He was involved in the campaigns of the failed Roman usurper Jovinus before the latter's defeat, after which he was settled on the left bank of the Rhine as a Roman ally. In 436, Gundahar launched an attack from his kingdom on the Roman province of Belgica Prima. He was defeated by the Roman general Flavius Aetius, who destroyed Gundahar's kingdom with the help of Hunnish mercenaries the following year, resulting in Gundahar's death. The historical Gundahar's death became the basis for a tradition in Germanic heroic legend in which the legendary Gunther met his death at the court of Attila the Hun (Etzel/Atli). The character also became attached to other legends: most notably he is associated with Siegfr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon Monoxide
Carbon monoxide (chemical formula CO) is a colorless, poisonous, odorless, tasteless, flammable gas that is slightly less dense than air. Carbon monoxide consists of one carbon atom and one oxygen atom connected by a triple bond. It is the simplest molecule of the oxocarbon family. In coordination complexes the carbon monoxide ligand is called carbonyl. It is a key ingredient in many processes in industrial chemistry. The most common source of carbon monoxide is the partial combustion of carbon-containing compounds, when insufficient oxygen or heat is present to produce carbon dioxide. There are also numerous environmental and biological sources that generate and emit a significant amount of carbon monoxide. It is important in the production of many compounds, including drugs, fragrances, and fuels. Upon emission into the atmosphere, carbon monoxide affects several processes that contribute to climate change. Carbon monoxide has important biological roles across phylogenetic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nickel Tetracarbonyl
Nickel carbonyl (IUPAC name: tetracarbonylnickel) is a nickel(0) organometallic compound with the formula Ni(CO)4. This colorless liquid is the principal carbonyl of nickel. It is an intermediate in the Mond process for producing very high-purity nickel and a reagent in organometallic chemistry, although the Mond Process has fallen out of common usage due to the health hazards in working with the compound. Nickel carbonyl is one of the most dangerous substances yet encountered in nickel chemistry due to its very high toxicity, compounded with high volatility and rapid skin absorption. Structure and bonding In nickel tetracarbonyl, the oxidation state for nickel is assigned as zero. The formula conforms to 18-electron rule. The molecule is tetrahedral, with four carbonyl (carbon monoxide) ligands. Electron diffraction studies have been performed on this molecule, and the Ni–C and C–O distances have been calculated to be 1.838(2) and 1.141(2) angstroms respectively. Prepar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
1,5-Hexadiene
1,5-Hexadiene is the organic compound with the formula (CH)(CH=CH). It is a colorless, volatile liquid. It is used as a crosslinking agent and precursor to a variety of other compounds. Synthesis 1,5-Hexadiene is produced commercially by the ethenolysis of 1,5-cyclooctadiene: :(CHCH=CHCH) + 2 CH=CH → 2 (CH)CH=CH The catalyst is derived from ReO on alumina. A laboratory-scale preparation involves reductive coupling of allyl chloride Allyl chloride is the organic compound with the formula C H2=CHCH2 Cl. This colorless liquid is insoluble in water but soluble in common organic solvents. It is mainly converted to epichlorohydrin, used in the production of plastics. It is a ch ... using magnesium: :2 ClCHCH=CH + Mg → (CH)(CH=CH) + MgCl References Alkadienes Monomers {{Hydrocarbons ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Butadiene
1,3-Butadiene () is the organic compound with the formula (CH2=CH)2. It is a colorless gas that is easily condensed to a liquid. It is important industrially as a precursor to synthetic rubber. The molecule can be viewed as the union of two vinyl groups. It is the simplest conjugated diene. Although butadiene breaks down quickly in the atmosphere, it is nevertheless found in ambient air in urban and suburban areas as a consequence of its constant emission from motor vehicles. The name butadiene can also refer to the isomer, 1,2-butadiene, which is a cumulated diene with structure H2C=C=CH−CH3. This allene has no industrial significance. History In 1863, the French chemist E. Caventou isolated butadiene from the pyrolysis of amyl alcohol. This hydrocarbon was identified as butadiene in 1886, after Henry Edward Armstrong isolated it from among the pyrolysis products of petroleum. In 1910, the Russian chemist Sergei Lebedev polymerized butadiene and obtained a material wit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organometallic Chemistry
Organometallic chemistry is the study of organometallic compounds, chemical compounds containing at least one chemical bond between a carbon atom of an organic molecule and a metal, including alkali, alkaline earth, and transition metals, and sometimes broadened to include metalloids like boron, silicon, and selenium, as well. Aside from bonds to organyl fragments or molecules, bonds to 'inorganic' carbon, like carbon monoxide (metal carbonyls), cyanide, or carbide, are generally considered to be organometallic as well. Some related compounds such as transition metal hydrides and metal phosphine complexes are often included in discussions of organometallic compounds, though strictly speaking, they are not necessarily organometallic. The related but distinct term " metalorganic compound" refers to metal-containing compounds lacking direct metal-carbon bonds but which contain organic ligands. Metal β-diketonates, alkoxides, dialkylamides, and metal phosphine complexes are repres ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Transition Metals
In chemistry, a transition metal (or transition element) is a chemical element in the d-block of the periodic table (groups 3 to 12), though the elements of group 12 (and less often group 3) are sometimes excluded. They are the elements that can use d orbitals as valence orbitals to form chemical bonds. The lanthanide and actinide elements (the f-block) are called inner transition metals and are sometimes considered to be transition metals as well. Since they are metals, they are lustrous and have good electrical and thermal conductivity. Most (with the exception of group 11 and group 12) are hard and strong, and have high melting and boiling temperatures. They form compounds in any of two or more different oxidation states and bind to a variety of ligands to form coordination complexes that are often coloured. They form many useful alloys and are often employed as catalysts in elemental form or in compounds such as coordination complexes and oxides. Most are strongly paramagn ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Allyl Complexes
In organic chemistry, an allyl group is a substituent with the structural formula , where R is the rest of the molecule. It consists of a methylene bridge () attached to a vinyl group (). The name is derived from the scientific name for garlic, . In 1844, Theodor Wertheim isolated an allyl derivative from garlic oil and named it "". The term allyl applies to many compounds related to , some of which are of practical or of everyday importance, for example, allyl chloride. Allylation is any chemical reaction that adds an allyl group to a substrate. Nomenclature A site adjacent to the unsaturated carbon atom is called the allylic position or allylic site. A group attached at this site is sometimes described as allylic. Thus, "has an allylic hydroxyl group". Allylic C−H bonds are about 15% weaker than the C−H bonds in ordinary sp3 carbon centers and are thus more reactive. Benzylic and allylic are related in terms of structure, bond strength, and reactivity. Other reac ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |


_complexes_in_aqueous_solution.jpg)