|
Barium Chromate(V)
Barium chromate, named barium tetraoxochromate(VI) by the IUPAC, is a yellow sand like powder with the formula BaCrO4. It is a known oxidizing agent and produces a green flame when heated, a result of the barium ions. History The first naturally occurring barium chromate was found in the country of Jordan. The brown crystals found perched on host rocks were named hashemite in honor of the Hashemite Kingdom of Jordan. The hashemite crystals range in color from light yellowish-brown to a darker greenish-brown and are usually less than 1mm in length. The hashemite crystals are not composed of pure barium chromate but instead contain some small sulfur content as well. The different crystals contain a range of sulfur impurities ranging from the more pure dark crystals, Ba1.00(Cr0.93, S0.07)1.00O4, to the less pure light crystals, Ba1.00(Cr0.64, S0.36)1.00O4. Hashemite was found to be an isostructural chromate analog of baryte, BaSO4. Preparation and Reactions It can be synthesized by ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
IUPAC
The International Union of Pure and Applied Chemistry (IUPAC ) is an international federation of National Adhering Organizations working for the advancement of the chemical sciences, especially by developing nomenclature and terminology. It is a member of the International Science Council (ISC). IUPAC is registered in Zürich, Switzerland, and the administrative office, known as the "IUPAC Secretariat", is in Research Triangle Park, North Carolina, United States. This administrative office is headed by IUPAC's executive director, currently Lynn Soby. IUPAC was established in 1919 as the successor of the International Congress of Applied Chemistry for the advancement of chemistry. Its members, the National Adhering Organizations, can be national chemistry societies, national academies of sciences, or other bodies representing chemists. There are fifty-four National Adhering Organizations and three Associate National Adhering Organizations. IUPAC's Inter-divisional Committee on ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fumaric Acid
Fumaric acid is an organic compound with the formula HO2CCH=CHCO2H. A white solid, fumaric acid occurs widely in nature. It has a fruit-like taste and has been used as a food additive. Its E number is E297. The salts and esters are known as fumarates. Fumarate can also refer to the ion (in solution). Fumaric acid is the trans isomer of butenedioic acid, while maleic acid is the cis isomer. Biosynthesis and occurrence It is produced in eukaryotic organisms from succinate in complex 2 of the electron transport chain via the enzyme succinate dehydrogenase. It is one of two isomeric unsaturated dicarboxylic acids, the other being maleic acid. In fumaric acid the carboxylic acid groups are ''trans'' (''E'') and in maleic acid they are ''cis'' (''Z''). Fumaric acid is found in fumitory (''Fumaria officinalis''), bolete mushrooms (specifically ''Boletus fomentarius var. pseudo-igniarius''), lichen, and Iceland moss. Fumarate is an intermediate in the citric acid cycle used b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
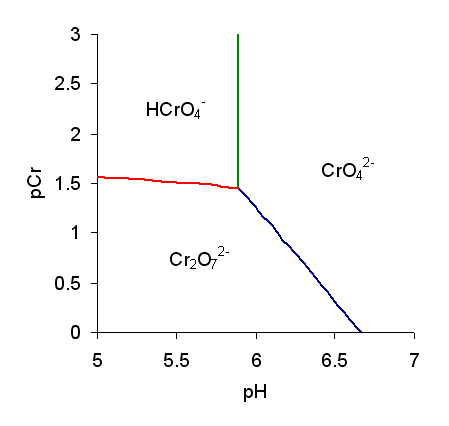
Chromates
Chromate salts contain the chromate anion, . Dichromate salts contain the dichromate anion, . They are oxyanions of chromium in the +6 oxidation state and are moderately strong oxidizing agents. In an aqueous solution, chromate and dichromate ions can be interconvertible. Chemical properties Potassium-chromate-sample.jpg, potassium chromate Potassium-dichromate-sample.jpg, potassium dichromate Chromates react with hydrogen peroxide, giving products in which peroxide, , replaces one or more oxygen atoms. In acid solution the unstable blue peroxo complex Chromium(VI) oxide peroxide, CrO(O2)2, is formed; it is an uncharged covalent molecule, which may be extracted into ether. Addition of pyridine results in the formation of the more stable complex CrO(O2)2py. Acid–base properties In aqueous solution, chromate and dichromate anions exist in a chemical equilibrium. :2 + 2 H+ + H2O The predominance diagram shows that the position of the equilibrium depends on b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Barium Compounds
Barium is a chemical element with the symbol Ba and atomic number 56. It is the fifth element in group 2 and is a soft, silvery alkaline earth metal. Because of its high chemical reactivity, barium is never found in nature as a free element. The most common minerals of barium are baryte (barium sulfate, BaSO4) and witherite (barium carbonate, BaCO3). The name ''barium'' originates from the alchemical derivative "baryta", from Greek (), meaning 'heavy'. ''Baric'' is the adjectival form of barium. Barium was identified as a new element in 1774, but not reduced to a metal until 1808 with the advent of electrolysis. Barium has few industrial applications. Historically, it was used as a getter for vacuum tubes and in oxide form as the emissive coating on indirectly heated cathodes. It is a component of YBCO (high-temperature superconductors) and electroceramics, and is added to steel and cast iron to reduce the size of carbon grains within the microstructure. Barium compounds are ad ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carcinogen
A carcinogen is any substance, radionuclide, or radiation that promotes carcinogenesis (the formation of cancer). This may be due to the ability to damage the genome or to the disruption of cellular metabolic processes. Several radioactive substances are considered carcinogens, but their carcinogenic activity is attributed to the radiation, for example gamma rays and alpha particles, which they emit. Common examples of non-radioactive carcinogens are inhaled asbestos, certain dioxins, and tobacco smoke. Although the public generally associates carcinogenicity with synthetic chemicals, it is equally likely to arise from both natural and synthetic substances. Carcinogens are not necessarily immediately toxic; thus, their effect can be insidious. Carcinogens, as mentioned, are agents in the environment capable of contributing to cancer growth. Carcinogens can be categorized into two different types: activation-dependent and activation-independent, and each nature impacts their level ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Toxicity
Toxicity is the degree to which a chemical substance or a particular mixture of substances can damage an organism. Toxicity can refer to the effect on a whole organism, such as an animal, bacterium, or plant, as well as the effect on a substructure of the organism, such as a cell ( cytotoxicity) or an organ such as the liver (hepatotoxicity). By extension, the word may be metaphorically used to describe toxic effects on larger and more complex groups, such as the family unit or society at large. Sometimes the word is more or less synonymous with poisoning in everyday usage. A central concept of toxicology is that the effects of a toxicant are dose-dependent; even water can lead to water intoxication when taken in too high a dose, whereas for even a very toxic substance such as snake venom there is a dose below which there is no detectable toxic effect. Toxicity is species-specific, making cross-species analysis problematic. Newer paradigms and metrics are evolving to bypas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cytotoxic
Cytotoxicity is the quality of being toxic to cells. Examples of toxic agents are an immune cell or some types of venom, e.g. from the puff adder (''Bitis arietans'') or brown recluse spider (''Loxosceles reclusa''). Cell physiology Treating cells with the cytotoxic compound can result in a variety of cell fates. The cells may undergo necrosis, in which they lose membrane integrity and die rapidly as a result of cell lysis. The cells can stop actively growing and dividing (a decrease in cell viability), or the cells can activate a genetic program of controlled cell death (apoptosis). Cells undergoing necrosis typically exhibit rapid swelling, lose membrane integrity, shut down metabolism, and release their contents into the environment. Cells that undergo rapid necrosis in vitro do not have sufficient time or energy to activate apoptotic machinery and will not express apoptotic markers. Apoptosis is characterized by well defined cytological and molecular events including a change i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Genotoxic
Genotoxicity is the property of chemical agents that damage the genetic information within a cell causing mutations, which may lead to cancer. While genotoxicity is often confused with Mutagen, mutagenicity, all mutagens are genotoxic, but some genotoxic substances are not mutagenic. The alteration can have direct or indirect effects on the DNA: the induction of mutations, mistimed event activation, and direct DNA damage leading to mutations. The permanent, heritable changes can affect either somatic cells of the organism or germ cells to be passed on to future generations. Cells prevent expression of the genotoxic mutation by either DNA repair or apoptosis; however, the damage may not always be fixed leading to mutagenesis. To bioassay, assay for genotoxic molecules, researchers assay for DNA damage in cells exposed to the toxic substrates. This DNA damage can be in the form of single- and double-strand breaks, loss of excision repair, cross-linking, alkali-labile sites, point mutat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zinc Chromate
Zinc chromate, Zn Cr O4, is a chemical compound containing the chromate anion, appearing as odorless yellow powder or yellow-green crystals, but, when used for coatings, pigments are often added. It is used industrially in chromate conversion coatings, having been developed by the Ford Motor Company in the 1920s. Production A process known as the Cronak process is used to create zinc chromate for use in industry. This process is done by putting zinc or a zinc plated metal in a solution of sodium dichromate and sulfuric acid for a few seconds. Zinc chromate can also be synthesized by using neutral potassium chromate (K2CrO4) and zinc sulfate (ZnSO4), which forms a precipitate. K2CrO4 + ZnSO4 → ZnCrO4 + K2SO4 Uses Zinc chromate's main use is in industrial painting as a coating over iron or aluminum materials. It was used extensively on aircraft by the US military, especially during the 1930s and 1940s. It is also used in a variety of paint coatings for the aerospace and a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Claude Monet
Oscar-Claude Monet (, , ; 14 November 1840 – 5 December 1926) was a French painter and founder of impressionist painting who is seen as a key precursor to modernism, especially in his attempts to paint nature as he perceived it. During his long career, he was the most consistent and prolific practitioner of impressionism's philosophy of expressing one's perceptions before nature, especially as applied to ''plein air'' (outdoor) landscape painting. The term "Impressionism" is derived from the title of his painting '' Impression, soleil levant'', exhibited in the 1874 ("exhibition of rejects") initiated by Monet and his associates as an alternative to the Salon. Monet was raised in Le Havre, Normandy, and became interested in the outdoors and drawing from an early age. Although his mother, Louise-Justine Aubrée Monet, supported his ambitions to be a painter, his father, Claude-Adolphe, disapproved and wanted him to pursue a career in business. He was very close to his mot ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pierre-Auguste Renoir
Pierre-Auguste Renoir (; 25 February 1841 – 3 December 1919) was a French artist who was a leading painter in the development of the Impressionist style. As a celebrator of beauty and especially feminine sensuality, it has been said that "Renoir is the final representative of a tradition which runs directly from Rubens to Watteau." He was the father of actor Pierre Renoir (1885–1952), filmmaker Jean Renoir (1894–1979) and ceramic artist Claude Renoir (1901–1969). He was the grandfather of the filmmaker Claude Renoir (1913–1993), son of Pierre. Life Youth Pierre-Auguste Renoir was born in Limoges, Haute-Vienne, France, in 1841. His father, Léonard Renoir, was a tailor of modest means, so, in 1844, Renoir's family moved to Paris in search of more favorable prospects. The location of their home, in rue d’Argenteuil in central Paris, placed Renoir in proximity to the Louvre. Although the young Renoir had a natural proclivity for drawing, he exhibited a greater t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Barium Chromate(V)
Barium chromate, named barium tetraoxochromate(VI) by the IUPAC, is a yellow sand like powder with the formula BaCrO4. It is a known oxidizing agent and produces a green flame when heated, a result of the barium ions. History The first naturally occurring barium chromate was found in the country of Jordan. The brown crystals found perched on host rocks were named hashemite in honor of the Hashemite Kingdom of Jordan. The hashemite crystals range in color from light yellowish-brown to a darker greenish-brown and are usually less than 1mm in length. The hashemite crystals are not composed of pure barium chromate but instead contain some small sulfur content as well. The different crystals contain a range of sulfur impurities ranging from the more pure dark crystals, Ba1.00(Cr0.93, S0.07)1.00O4, to the less pure light crystals, Ba1.00(Cr0.64, S0.36)1.00O4. Hashemite was found to be an isostructural chromate analog of baryte, BaSO4. Preparation and Reactions It can be synthesized by ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |