|
Armed And Disarmed Saccharides
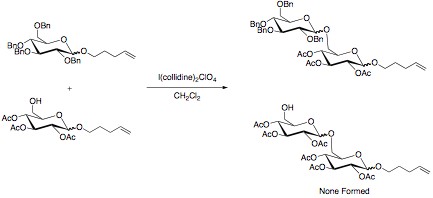
The armed/disarmed approach to glycosylation is an effective way to prevent sugar molecules from self-glycosylation when synthesizing disaccharides. This approach was first recognized when acetylated sugars only acted as glycosyl acceptors when reacted with benzylated sugars. The acetylated sugars were termed “disarmed” while the benzylated sugars were termed “armed”. Electronic Effect The selectivity in the reaction is due to the stronger electron withdrawing power of the esters compared to the ethers. A stronger electron withdrawing substituent leads to a greater destabilization of the oxocarbenium ion. This slows this reaction pathway, and allows for disaccharide formation to occur with the benzylated sugar. Other effective electron withdrawing groups that have shown selectivity are halogens and azido groups, while deoxygenation has been proven an effective tool in “arming” sugars. Torsional Effect Disarming sugars can also be accomplished by adding 1,3-dioxane ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glycosylation
Glycosylation is the reaction in which a carbohydrate (or ' glycan'), i.e. a glycosyl donor, is attached to a hydroxyl or other functional group of another molecule (a glycosyl acceptor) in order to form a glycoconjugate. In biology (but not always in chemistry), glycosylation usually refers to an enzyme-catalysed reaction, whereas glycation (also 'non-enzymatic glycation' and 'non-enzymatic glycosylation') may refer to a non-enzymatic reaction (though in practice, 'glycation' often refers more specifically to Maillard-type reactions). Glycosylation is a form of co-translational and post-translational modification. Glycans serve a variety of structural and functional roles in membrane and secreted proteins. The majority of proteins synthesized in the rough endoplasmic reticulum undergo glycosylation. Glycosylation is also present in the cytoplasm and nucleus as the ''O''-GlcNAc modification. Aglycosylation is a feature of engineered antibodies to bypass glycosylation. Five clas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Disaccharide
A disaccharide (also called a double sugar or ''biose'') is the sugar formed when two monosaccharides are joined by glycosidic linkage. Like monosaccharides, disaccharides are simple sugars soluble in water. Three common examples are sucrose, lactose, and maltose. Disaccharides are one of the four chemical groupings of carbohydrates (monosaccharides, disaccharides, oligosaccharides, and polysaccharides). The most common types of disaccharides—sucrose, lactose, and maltose—have 12 carbon atoms, with the general formula C12H22O11. The differences in these disaccharides are due to atomic arrangements within the molecule. The joining of monosaccharides into a double sugar happens by a condensation reaction, which involves the elimination of a water molecule from the functional groups only. Breaking apart a double sugar into its two monosaccharides is accomplished by hydrolysis with the help of a type of enzyme called a disaccharidase. As building the larger sugar ejects a water ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Armed Disarmed 1
Armed (May, 1941–1964) was an American Thoroughbred gelding race horse who was the American Horse of the Year in 1947 and Champion Older Male Horse in both 1946 and 1947. He was inducted into the National Museum of Racing and Hall of Fame in 1963. Background Armed was sired by the great stakes winner Bull Lea, the sire of Citation. His dam was Armful, whose sire was Belmont Stakes winner Chance Shot and whose grandsire was the great Fair Play. Besides being small for his age and very headstrong, Armed had the habits of biting and kicking hay out of his handler's pitchfork. Since he was also practically untrainable, his trainer, Ben A. Jones, sent him back to Calumet Farm to be gelded and turned out to grow up. He returned to the track late in his two-year-old season and resumed training. Racing career His first start was as a three-year-old the following February, and he won at Hialeah Park by eight lengths. He won again less than a week later but then won only on ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxocarbenium
An oxocarbenium ion (or oxacarbenium ion) is a chemical species characterized by a central sp2-hybridized carbon, an oxygen substituent, and an overall positive charge that is delocalized between the central carbon and oxygen atoms. An oxocarbenium ion is represented by two limiting resonance structures, one in the form of a carbenium ion with the positive charge on carbon and the other in the form of an oxonium species with the formal charge on oxygen. As a resonance hybrid, the true structure falls between the two. Compared to neutral carbonyl compounds like ketones or esters, the carbenium ion form is a larger contributor to the structure. They are common reactive intermediates in the hydrolysis of glycosidic bonds, and are a commonly used strategy for chemical glycosylation. These ions have since been proposed as reactive intermediates in a wide range of chemical transformations, and have been utilized in the total synthesis of several natural products. In addition, they com ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Armed Disarmed 2
Armed (May, 1941–1964) was an American Thoroughbred gelding race horse who was the American Horse of the Year in 1947 and Champion Older Male Horse in both 1946 and 1947. He was inducted into the National Museum of Racing and Hall of Fame in 1963. Background Armed was sired by the great stakes winner Bull Lea, the sire of Citation. His dam was Armful, whose sire was Belmont Stakes winner Chance Shot and whose grandsire was the great Fair Play. Besides being small for his age and very headstrong, Armed had the habits of biting and kicking hay out of his handler's pitchfork. Since he was also practically untrainable, his trainer, Ben A. Jones, sent him back to Calumet Farm to be gelded and turned out to grow up. He returned to the track late in his two-year-old season and resumed training. Racing career His first start was as a three-year-old the following February, and he won at Hialeah Park by eight lengths. He won again less than a week later but then won only on ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dioxane
1,4-Dioxane () is a heterocyclic organic compound, classified as an ether. It is a colorless liquid with a faint sweet odor similar to that of diethyl ether. The compound is often called simply dioxane because the other dioxane isomers ( 1,2- and 1,3-) are rarely encountered. Dioxane is used as a solvent for a variety of practical applications as well as in the laboratory, and also as a stabilizer for the transport of chlorinated hydrocarbons in aluminum containers.Wisconsin Department of Health Services (20131,4-Dioxane Fact Sheet Publication 00514. Accessed 2016-11-12. Synthesis Dioxane is produced by the acid-catalysed dehydration of diethylene glycol, which in turn is obtained from the hydrolysis of ethylene oxide. In 1985, the global production capacity for dioxane was between 11,000 and 14,000 tons. In 1990, the total U.S. production volume of dioxane was between 5,250 and 9,150 tons. Structure The dioxane molecule is centrosymmetric, meaning that it adopts a chai ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
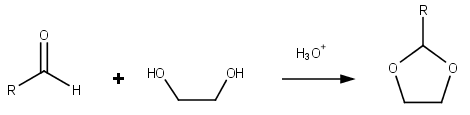
Dioxolane
Dioxolane is a heterocyclic acetal with the chemical formula (CH2)2O2CH2. It is related to tetrahydrofuran by interchange of one oxygen for a CH2 group. The corresponding saturated 6-membered C4O2 rings are called dioxanes. The isomeric 1,2-dioxolane (wherein the two oxygen centers are adjacent) is a peroxide. 1,3-dioxolane is used as a solvent and as a comonomer in polyacetals. As a class of compounds Dioxolanes are a group of organic compounds containing the dioxolane ring. Dioxolanes can be prepared by acetalization of aldehydes and ketalization of ketones with ethylene glycol. (+)-''cis''-Dioxolane is the trivial name for which is a muscarinic acetylcholine receptor agonist. Protecting groups Organic compounds containing carbonyl groups sometimes need protection so that they do not undergo reactions during transformations of other functional groups that may be present. A variety of approaches to protection and deprotection of carbonyls including as dioxolanes are known ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hyperconjugation
In organic chemistry, hyperconjugation (σ-conjugation or no-bond resonance) refers to the delocalization of electrons with the participation of bonds of primarily σ-character. Usually, hyperconjugation involves the interaction of the electrons in a sigma (σ) orbital (e.g. C–H or C–C) with an adjacent unpopulated non-bonding p or antibonding σ* or π* orbitals to give a pair of extended molecular orbitals. However, sometimes, low-lying antibonding σ* orbitals may also interact with filled orbitals of lone pair character (n) in what is termed ''negative hyperconjugation''. Increased electron delocalization associated with hyperconjugation increases the stability of the system. In particular, the new orbital with bonding character is stabilized, resulting in an overall stabilization of the molecule. Only electrons in bonds that are in the β position can have this sort of direct stabilizing effect — donating from a sigma bond on an atom to an orbital in another ato ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Inductive Effect
In chemistry, the inductive effect in a molecule is a local change in the electron density due to electron-withdrawing or electron-donating groups elsewhere in the molecule, resulting in a permanent dipole in a bond. It is present in a σ (sigma) bond, unlike the electromeric effect which is present in a π (pi) bond. The halogen atoms in an alkyl halide are electron withdrawing while the alkyl groups have electron donating tendencies. If the electronegative atom (missing an electron, thus having a positive charge) is then joined to a chain of atoms, usually carbon, the positive charge is relayed to the other atoms in the chain. This is the electron-withdrawing inductive effect, also known as the effect. In short, alkyl groups tend to donate electrons, leading to the effect. Its experimental basis is the ionization constant. It is distinct from and often opposite to the mesomeric effect. Bond polarization Covalent bonds can be polarized depending on the relative electron ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Armed Disarmed 3
Armed (May, 1941–1964) was an American Thoroughbred gelding race horse who was the American Horse of the Year in 1947 and Champion Older Male Horse in both 1946 and 1947. He was inducted into the National Museum of Racing and Hall of Fame in 1963. Background Armed was sired by the great stakes winner Bull Lea, the sire of Citation. His dam was Armful, whose sire was Belmont Stakes winner Chance Shot and whose grandsire was the great Fair Play. Besides being small for his age and very headstrong, Armed had the habits of biting and kicking hay out of his handler's pitchfork. Since he was also practically untrainable, his trainer, Ben A. Jones, sent him back to Calumet Farm to be gelded and turned out to grow up. He returned to the track late in his two-year-old season and resumed training. Racing career His first start was as a three-year-old the following February, and he won at Hialeah Park by eight lengths. He won again less than a week later but then won only on ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glycosyl Donor
A glycosyl donor is a carbohydrate mono- or oligosaccharide that will react with a suitable glycosyl acceptor to form a new glycosidic bond. By convention, the donor is the member of this pair that contains the resulting anomeric carbon of the new glycosidic bond.T. K. Lindhorst "Essentials of Carbohydrate Chemistry and Biochemistry" 2007 Wiley-VCH Verlag, Weinheim The resulting reaction is referred to as a glycosylation or chemical glycosylation. In a glycosyl donor, a leaving group is required at the anomeric position. The simplest leaving group is the OH group that is naturally present in monosaccharides, but it requires activation by acid catalysis in order to function as leaving group (in the Fischer glycosylation). More effective leaving groups are in general used in the glycosyl donors employed in chemical synthesis of glycosides. Typical leaving groups are halides, thioalkyl groups, or imidates, but acetate, phosphate, and O-pentenyl groups are also employed. Natural glycosyl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glycosyl Acceptor
A glycosyl acceptor is any suitable nucleophile-containing molecule that will react with a glycosyl donor to form a new glycosidic bond. By convention, the acceptor is the member of this pair which did not contain the resulting anomeric carbon of the new glycosidic bond. Since the nucleophilic atom of the acceptor is typically an oxygen atom, this can be remembered using the mnemonic of ''the acceptor is the alcohol.'' A glycosyl acceptor can be a mono- or oligosaccharide that contains an available nucleophile, such as an unprotected hydroxyl. Background Examples glucose to haemoglobin See also * Chemical glycosylation * Glycosyl halide * Armed and disarmed saccharides * Carbohydrate chemistry Carbohydrate chemistry is a subdiscipline of chemistry primarily concerned with the detection, synthesis, structure, and function of carbohydrates. Due to the general structure of carbohydrates, their synthesis is often preoccupied with the select ... References *{{cite journal , las ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |