|
Arsenide
In chemistry, an arsenide is a compound of arsenic with a less electronegative element or elements. Many metals form binary compounds containing arsenic, and these are called arsenides. They exist with many stoichiometries, and in this respect arsenides are similar to phosphides. Alkali metal and alkaline earth arsenides The group 1 alkali metals and the group 2, alkaline earth metals, form arsenides with isolated arsenic atoms. They form upon heating arsenic powder with excess sodium gives sodium arsenide (Na3As). The structure of Na3As is complex with unusually short Na–Na distances of 328–330 pm which are shorter than in sodium metal. This short distance indicates the complex bonding in these simple phases, i.e. they are not simply salts of As3− anion, for example. The compound LiAs, has a metallic lustre and electrical conductivity indicating some metallic bonding. These compounds are mainly of academic interest. For example, "sodium arsenide" is a structural mo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gallium Arsenide
Gallium arsenide (GaAs) is a III-V direct band gap semiconductor with a Zincblende (crystal structure), zinc blende crystal structure. Gallium arsenide is used in the manufacture of devices such as microwave frequency integrated circuits, monolithic microwave integrated circuits, infrared light-emitting diodes, laser diodes, solar cells and optical windows. GaAs is often used as a substrate material for the epitaxial growth of other III-V semiconductors, including indium gallium arsenide, aluminum gallium arsenide and others. Preparation and chemistry In the compound, gallium has a +3 oxidation state. Gallium arsenide single crystals can be prepared by three industrial processes: * The vertical gradient freeze (VGF) process. * Crystal growth using a horizontal zone furnace in the Bridgman-Stockbarger technique, in which gallium and arsenic vapors react, and free molecules deposit on a seed crystal at the cooler end of the furnace. * Liquid encapsulated Czochralski process, Czoch ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zincblende (crystal Structure)
In crystallography, the cubic (or isometric) crystal system is a crystal system where the Crystal_structure#Unit_cell, unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals. There are three main varieties of these crystals: *Primitive cubic (abbreviated ''cP'' and alternatively called simple cubic) *Body-centered cubic (abbreviated ''cI'' or bcc) *Face-centered cubic (abbreviated ''cF'' or fcc, and alternatively called Close-packing_of_equal_spheres, ''cubic close-packed'' or ccp) Each is subdivided into other variants listed below. Although the ''unit cells'' in these crystals are conventionally taken to be cubes, the primitive_cell, primitive unit cells often are not. Bravais lattices The three Bravais lattices in the cubic crystal system are: The primitive cubic lattice (cP) consists of one Lattice_(group), lattice point on each corner of the cube; this means each simple cubic unit cell has in total one latt ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
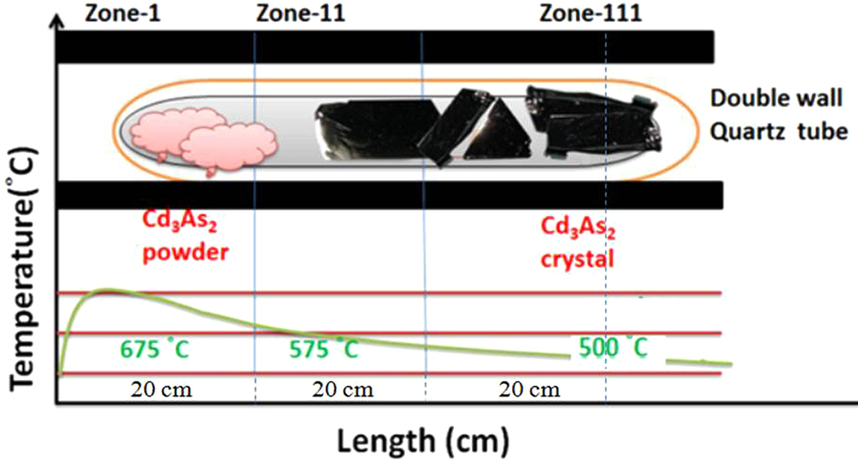
Cadmium Arsenide
Cadmium arsenide ( Cd3 As2) is an inorganic semimetal in the II-V family. It exhibits the Nernst effect. Properties Thermal Cd3As2 dissociates between 220 and 280 °C according to the reaction :2 Cd3As2(s) → 6 Cd(g) + As4(g) An energy barrier was found for the nonstoichiometric vaporization of arsenic due to the irregularity of the partial pressures with temperature. The range of the energy gap is from 0.5 to 0.6 eV. Cd3As2 melts at 716 °C and changes phase at 615 °C/ Phase transition Pure cadmium arsenide undergoes several phase transitions at high temperatures, making phases labeled α (stable), α’, α” (metastable), and β. At 593° the polymorphic transition α → β occurs. :α-Cd3As2 ↔ α’-Cd3As2 occurs at ~500 K. :α’-Cd3As2 ↔ α’’-Cd3As2 occurs at ~742 K and is a regular first order phase transition with marked hysteresis loop. :α”-Cd3As2 ↔ β-Cd3As2 occurs at 868 K. Single crystal x-ray diffraction was used to dete ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Arsenic
Arsenic is a chemical element with the symbol As and atomic number 33. Arsenic occurs in many minerals, usually in combination with sulfur and metals, but also as a pure elemental crystal. Arsenic is a metalloid. It has various allotropes, but only the gray form, which has a metallic appearance, is important to industry. The primary use of arsenic is in alloys of lead (for example, in car batteries and ammunition). Arsenic is a common n-type dopant in semiconductor electronic devices. It is also a component of the III-V compound semiconductor gallium arsenide. Arsenic and its compounds, especially the trioxide, are used in the production of pesticides, treated wood products, herbicides, and insecticides. These applications are declining with the increasing recognition of the toxicity of arsenic and its compounds. A few species of bacteria are able to use arsenic compounds as respiratory metabolites. Trace quantities of arsenic are an essential dietary element in rats, ham ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zinc Cadmium Phosphide Arsenide
Zinc cadmium phosphide arsenide ( Zn- Cd- P- As) is a quaternary system of group II (IUPAC group 12) and group V (IUPAC group 15) elements. Many of the inorganic compounds in the system are II-V semiconductor materials. The quaternary system of II3V2 compounds, (Zn1−xCdx)3(P1−yAsy)2, has been shown to allow solid solution continuously over the whole compositional range. This material system and its subsets have applications in electronics, optoelectronics, including photovoltaics, and thermoelectrics. List of all binary compounds This system of elements contains numerous binary compounds and their solid solutions. Stable at atmospheric pressure The binary compounds thermodynamically stable at atmospheric pressure are listed in the following table: Metastable or unstable at atmospheric pressure Compounds metastable or unstable at atmospheric pressure are the following: Quaternary compounds The compounds of the form II3V2 have similar crystalline structures and e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zinc Arsenide
Zinc arsenide (Zn3As2) is a binary compound of zinc with arsenic which forms gray tetragonal crystals. It is an inorganic semiconductor with a band gap of 1.0 eV. Synthesis and reactions Zinc arsenide can be prepared by the reaction of zinc with arsenic :3 Zn + 2 As → Zn3As2 Structure Zn3As2 has a room-temperature tetragonal form that converts to a different tetragonal phase at 190 °C and to a third phase at 651 °C. In the room-temperature form, the zinc atoms are tetrahedrally coordinated and the arsenic atoms are surrounded by six zinc atoms at the vertices of a distorted cube. The crystalline structure of zinc arsenide is very similar to that of cadmium arsenide (Cd3As2), zinc phosphide (Zn3P2) and cadmium phosphide (Cd3P2). These compounds of the Zn-Cd-P-As quaternary system exhibit full continuous solid-solution. Electronic structure Its lowest direct and indirect bandgaps are within 30 meV or each other. References arsenide zinc Zinc i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nickeline
Nickeline or niccolite is a mineral consisting primarily of nickel arsenide (NiAs). The naturally-occurring mineral contains roughly 43.9% nickel and 56.1% arsenic by mass, but composition of the mineral may vary slightly. Small quantities of sulfur, iron and cobalt are usually present, and sometimes the arsenic is largely replaced by antimony. This last forms an isomorphous series with breithauptite (nickel antimonide). Etymology and history Medieval miners looking for copper in the German Erzgebirge ("Ore Mountains") would sometimes find a red mineral, superficially resembling copper ore. Upon attempting extraction, no copper was produced, and subsequently, the miners would be afflicted with mysterious illness. They blamed a mischievous sprite of German mythology, Nickel (similar to ''Old Nick'') for besetting the copper (German: Kupfer). This German equivalent of "copper-nickel" was used as early as 1694 (other old German synonyms are ''Rotnickelkies'' and ''Arsennickel' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkali Metal
The alkali metals consist of the chemical elements lithium (Li), sodium (Na), potassium (K),The symbols Na and K for sodium and potassium are derived from their Latin names, ''natrium'' and ''kalium''; these are still the origins of the names for the elements in some languages, such as German and Russian. rubidium (Rb), caesium (Cs), and francium (Fr). Together with hydrogen they constitute Group (periodic table)#Group names, group 1, which lies in the s-block of the periodic table. All alkali metals have their outermost electron in an atomic orbital, s-orbital: this shared electron configuration results in their having very similar characteristic properties. Indeed, the alkali metals provide the best example of periodic trends, group trends in properties in the periodic table, with elements exhibiting well-characterised homology (chemistry), homologous behaviour. This family of elements is also known as the lithium family after its leading element. The alkali metals are all sh ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Arsenide
Sodium arsenide, also known as trisodium arsenide, is the inorganic compound of sodium and arsenic Arsenic is a chemical element with the symbol As and atomic number 33. Arsenic occurs in many minerals, usually in combination with sulfur and metals, but also as a pure elemental crystal. Arsenic is a metalloid. It has various allotropes, b ... with the formula Na3As. It is a dark colored solid that degrades upon contact with water or air. It is prepared by the reaction of the elements at 200–400 °C. The compound is mainly of interest as exhibiting an archetypal structure. The normal pressure "sodium arsenide" phase is adopted by many alkali metal pnictides. At 3.6 gigapascals, Na3As adopts the Li3Bi structure, which is another archetypal structure. Sodium arsenide is a crystalline solid used as a semiconductor and in photo optic applications. Its IUPAC name is disodioarsanylsodium. References Sodium compounds {{Inorganic-compound-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Skutterudite
Named after Skuterudåsen, a hill in Modum, Norway, skutterudite is a cobalt arsenide mineral containing variable amounts of nickel and iron substituting for cobalt with the ideal formula CoAs3. Some references give the arsenic a variable formula subscript of 2–3. High nickel varieties are referred to as nickel-skutterudite, previously chloanthite. It is a hydrothermal ore mineral found in moderate to high temperature veins with other Ni-Co minerals. Associated minerals are arsenopyrite, native silver, erythrite, annabergite, nickeline, cobaltite, silver sulfosalts, native bismuth, calcite, siderite, barite and quartz. It is mined as an ore of cobalt and nickel with a by-product of arsenic. The crystal structure of this mineral has been found to be exhibited by several compounds with important technological uses. The mineral has a bright metallic luster, and is tin white or light steel gray in color with a black streak. The specific gravity is 6.5 and the hardness is 5.5–6. I ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Arsine
Arsine (IUPAC name: arsane) is an inorganic compound with the formula As H3. This flammable, pyrophoric, and highly toxic pnictogen hydride gas is one of the simplest compounds of arsenic. Despite its lethality, it finds some applications in the semiconductor industry and for the synthesis of organoarsenic compounds. The term ''arsine'' is commonly used to describe a class of organoarsenic compounds of the formula AsH3−xRx, where R = aryl or alkyl. For example, As(C6H5)3, called triphenylarsine, is referred to as "an arsine". General properties At its standard state, arsine is a colorless, denser-than-air gas that is slightly soluble in water (20% at 20 °C) and in many organic solvents as well. Whereas arsine itself is odorless, owing to its oxidation by air, it is possible to smell a slight garlic or fish-like scent when the compound is present above 0.5 ppm. This compound is kinetically stable: at room temperature it decomposes only slowly. At temperatures of ca. 230&n ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Band Theory
In solid-state physics, the electronic band structure (or simply band structure) of a solid describes the range of energy levels that electrons may have within it, as well as the ranges of energy that they may not have (called ''band gaps'' or ''forbidden bands''). Band theory derives these bands and band gaps by examining the allowed quantum mechanical wave functions for an electron in a large, periodic lattice of atoms or molecules. Band theory has been successfully used to explain many physical properties of solids, such as electrical resistivity and optical absorption, and forms the foundation of the understanding of all solid-state devices (transistors, solar cells, etc.). Why bands and band gaps occur The electrons of a single, isolated atom occupy atomic orbitals each of which has a discrete energy level. When two or more atoms join together to form a molecule, their atomic orbitals overlap and hybridize. Similarly, if a large number ''N'' of identical atoms come ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |