|
Annulenes
Annulenes are monocyclic hydrocarbons that contain the maximum number of non-cumulated or conjugated double bonds (' mancude'). They have the general formula CnHn (when ''n'' is an even number) or C''n''H''n''+1 (when ''n'' is an odd number). The IUPAC naming conventions are that annulenes with 7 or more carbon atoms are named as 'n''nnulene, where ''n'' is the number of carbon atoms in their ring, though sometimes the smaller annulenes are referred to using the same notation, and benzene is sometimes referred to simply as annulene. The first three even annulenes are cyclobutadiene, benzene, and cyclooctatetraene ( nnulene). Some annulenes, namely cyclobutadiene, cyclodecapentaene ( 0nnulene), cyclododecahexaene ( 2nnulene) and cyclotetradecaheptaene ( 4nnulene), are unstable, with cyclobutadiene extremely so. In the related annulynes, one double bond is replaced by a triple bond. Aromaticity Annulenes may be ''aromatic'' (benzene, nnulene and 8nnulene), ''non-aromatic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Annulenes
Annulenes are monocyclic hydrocarbons that contain the maximum number of non-cumulated or conjugated double bonds (' mancude'). They have the general formula CnHn (when ''n'' is an even number) or C''n''H''n''+1 (when ''n'' is an odd number). The IUPAC naming conventions are that annulenes with 7 or more carbon atoms are named as 'n''nnulene, where ''n'' is the number of carbon atoms in their ring, though sometimes the smaller annulenes are referred to using the same notation, and benzene is sometimes referred to simply as annulene. The first three even annulenes are cyclobutadiene, benzene, and cyclooctatetraene ( nnulene). Some annulenes, namely cyclobutadiene, cyclodecapentaene ( 0nnulene), cyclododecahexaene ( 2nnulene) and cyclotetradecaheptaene ( 4nnulene), are unstable, with cyclobutadiene extremely so. In the related annulynes, one double bond is replaced by a triple bond. Aromaticity Annulenes may be ''aromatic'' (benzene, nnulene and 8nnulene), ''non-aromatic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclododecahexaene
Cyclododecahexaene or 2nnulene () is a member of the series of annulenes with some interest in organic chemistry with regard to the study of aromaticity. Cyclododecahexaene is non-aromatic due to the lack of planarity of the structure. On the other hand the dianion with 14 electrons is a Hückel aromat and more stable. According to in silico experiments the tri-trans isomer is expected to be the most stable, followed by the 1,7-ditrans and the all cis-isomers (+1 kcal/mol) and by the 1,5-ditrans isomer (+5 kcal/mol). The first 2nnulene with sym-tri-trans configuration was synthesized in 1970 from a tricyclic precursor by photolysis at low temperatures. On heating the compound rearranges to a bicyclic .4.0isomer. Reducing the compound at low temperatures allowed analysis of the dianion by proton NMR with the inner protons resonating at -4.5 ppm relative to TMS, evidence of an aromatic diamagnetic ring current. : In one study the 1,7-ditrans isomer is generated at low t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclododecahexaene
Cyclododecahexaene or 2nnulene () is a member of the series of annulenes with some interest in organic chemistry with regard to the study of aromaticity. Cyclododecahexaene is non-aromatic due to the lack of planarity of the structure. On the other hand the dianion with 14 electrons is a Hückel aromat and more stable. According to in silico experiments the tri-trans isomer is expected to be the most stable, followed by the 1,7-ditrans and the all cis-isomers (+1 kcal/mol) and by the 1,5-ditrans isomer (+5 kcal/mol). The first 2nnulene with sym-tri-trans configuration was synthesized in 1970 from a tricyclic precursor by photolysis at low temperatures. On heating the compound rearranges to a bicyclic .4.0isomer. Reducing the compound at low temperatures allowed analysis of the dianion by proton NMR with the inner protons resonating at -4.5 ppm relative to TMS, evidence of an aromatic diamagnetic ring current. : In one study the 1,7-ditrans isomer is generated at low t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclooctatetraene
1,3,5,7-Cyclooctatetraene (COT) is an unsaturated derivative of cyclooctane, with the formula C8H8. It is also known as nnulene. This polyunsaturated hydrocarbon is a colorless to light yellow flammable liquid at room temperature. Because of its stoichiometric relationship to benzene, COT has been the subject of much research and some controversy. Unlike benzene, C6H6, cyclooctatetraene, C8H8, is not aromatic, although its dianion, (cyclooctatetraenide), is. Its reactivity is characteristic of an ordinary polyene, i.e. it undergoes addition reactions. Benzene, by contrast, characteristically undergoes substitution reactions, not additions. History 1,3,5,7-Cyclooctatetraene was initially synthesized by Richard Willstätter in Munich in 1905 using pseudopelletierine as the starting material and the Hofmann elimination as the key transformation: : Willstätter noted that the compound did not exhibit the expected aromaticity. Between 1939 and 1943, chemists throughout the US u ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclooctatetraene
1,3,5,7-Cyclooctatetraene (COT) is an unsaturated derivative of cyclooctane, with the formula C8H8. It is also known as nnulene. This polyunsaturated hydrocarbon is a colorless to light yellow flammable liquid at room temperature. Because of its stoichiometric relationship to benzene, COT has been the subject of much research and some controversy. Unlike benzene, C6H6, cyclooctatetraene, C8H8, is not aromatic, although its dianion, (cyclooctatetraenide), is. Its reactivity is characteristic of an ordinary polyene, i.e. it undergoes addition reactions. Benzene, by contrast, characteristically undergoes substitution reactions, not additions. History 1,3,5,7-Cyclooctatetraene was initially synthesized by Richard Willstätter in Munich in 1905 using pseudopelletierine as the starting material and the Hofmann elimination as the key transformation: : Willstätter noted that the compound did not exhibit the expected aromaticity. Between 1939 and 1943, chemists throughout the US u ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Annulyne
Annulynes or dehydroannulenes are conjugated monocyclic hydrocarbons with alternating single and double bonds in addition to at least one triple bond. They are related to annulenes, which only have alternating single and double bonds. The smallest member of this class is nnulyne but is never observed because the molecule carries too much angle strain. The next member is nnulyne or benzyne which is a reactive intermediate well known in organic chemistry. nnulyne is known to exist but quickly dimerizes or trimerizes; the compound has been trapped as its radical anion and observed by EPR spectroscopy. 0nnulyne, like nnulyne, only exists in theory. 2nnulyne has been observed in 2005 by Stevenson et al. in solution by NMR spectroscopy at room temperature. Reaction of 1,5-hexadiyne and potassium tert-butoxide was reported to yield two isomers 5,9-di-''trans''- 2annulyne and 3,11-di-''trans''- 2nnulyne in a 1:1 ratio. The proposed reaction sequence involved an unspecified elec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclotetradecaheptaene
Cyclotetradecaheptaene, often referred to as 4nnulene, is a hydrocarbon with molecular formula C14H14, which played an important role in the development of criteria (Hückel's rule) for aromaticity, a stabilizing property of central importance in physical organic chemistry. It forms dark-red needle-like crystals. Structure and aromaticity Although the conjugated ring of 4 nnulene contains 4''n''+2 electrons, it only exhibits limited evidence for being aromatic. It does not fully conform to Hückel's rule because none of its ''cis''/''trans'' isomers can adopt a completely planar conformation due to crowding of the interior hydrogens. There is evidence that it has two isomeric forms of comparable stability (''trans'', ''cis'', ''trans'', ''cis'', ''trans'', ''trans'', ''cis''- with four interior hydrogens (shown in the infobox) and ''trans'', ''cis'', ''trans'', ''cis'', ''trans'', ''cis'', ''cis-'' with three interior hydrogens) which rapidly interconvert at room temperature b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclodecapentaene
Cyclodecapentaene or 0nnulene is an annulene with molecular formula C10H10. This organic compound is a conjugated 10 pi electron cyclic system and according to Huckel's rule it should display aromaticity. It is not aromatic, however, because various types of ring strain destabilize an all-planar geometry. The all-cis isomer (1), a fully convex decagon, would have bond angles of 144°, which creates large amounts of angle strain relative to the ideal 120° for sp2 atomic hybridization. Instead, the all-cis isomer can adopt a planar boat-like conformation (2) to relieve the angle strain. This is still unstable because of the relative higher strain in boat shaped compared to the next planar trans, cis, trans, cis, cis isomer (3). Yet even this isomer is also unstable, suffering from steric repulsion between the two internal hydrogen atoms. The nonplanar trans, cis, cis, cis, cis isomer (4) is the most stable of all the possible isomers. The 0nnulene compound can be obtained by ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzene
Benzene is an organic chemical compound with the molecular formula C6H6. The benzene molecule is composed of six carbon atoms joined in a planar ring with one hydrogen atom attached to each. Because it contains only carbon and hydrogen atoms, benzene is classed as a hydrocarbon. Benzene is a natural constituent of petroleum and is one of the elementary petrochemicals. Due to the cyclic continuous pi bonds between the carbon atoms, benzene is classed as an aromatic hydrocarbon. Benzene is a colorless and highly flammable liquid with a sweet smell, and is partially responsible for the aroma of gasoline. It is used primarily as a precursor to the manufacture of chemicals with more complex structure, such as ethylbenzene and cumene, of which billions of kilograms are produced annually. Although benzene is a major industrial chemical, it finds limited use in consumer items because of its toxicity. History Discovery The word "''benzene''" derives from "''gum benzoin''" (benzoin res ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
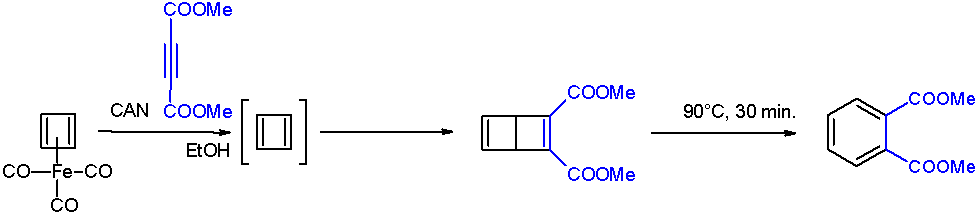
Cyclobutadiene
Cyclobutadiene is an organic compound with the formula . It is very reactive owing to its tendency to dimerize. Although the parent compound has not been isolated, some substituted derivatives are robust and a single molecule of cyclobutadiene is quite stable. Since the compound degrades by a bimolecular process, the species can be observed by matrix isolation techniques at temperatures below 35 K. It is thought to adopt a rectangular structure. Structure and reactivity The compound is the prototypical antiaromatic hydrocarbon with 4 π-electrons. It is the smallest 'n''annulene ( annulene). Its rectangular structure is the result of the Jahn–Teller effect, which distorts the molecule and lowers its symmetry, converting the triplet to a singlet ground state. The electronic states of cyclobutadiene have been explored with a variety of computational methods. The rectangular structure is consistent with the existence of two different 1,2-dideutero-1,3-cyclobutadiene valence iso ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclobutadiene
Cyclobutadiene is an organic compound with the formula . It is very reactive owing to its tendency to dimerize. Although the parent compound has not been isolated, some substituted derivatives are robust and a single molecule of cyclobutadiene is quite stable. Since the compound degrades by a bimolecular process, the species can be observed by matrix isolation techniques at temperatures below 35 K. It is thought to adopt a rectangular structure. Structure and reactivity The compound is the prototypical antiaromatic hydrocarbon with 4 π-electrons. It is the smallest 'n''annulene ( annulene). Its rectangular structure is the result of the Jahn–Teller effect, which distorts the molecule and lowers its symmetry, converting the triplet to a singlet ground state. The electronic states of cyclobutadiene have been explored with a variety of computational methods. The rectangular structure is consistent with the existence of two different 1,2-dideutero-1,3-cyclobutadiene valence iso ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclotetradecaheptaene
Cyclotetradecaheptaene, often referred to as 4nnulene, is a hydrocarbon with molecular formula C14H14, which played an important role in the development of criteria (Hückel's rule) for aromaticity, a stabilizing property of central importance in physical organic chemistry. It forms dark-red needle-like crystals. Structure and aromaticity Although the conjugated ring of 4 nnulene contains 4''n''+2 electrons, it only exhibits limited evidence for being aromatic. It does not fully conform to Hückel's rule because none of its ''cis''/''trans'' isomers can adopt a completely planar conformation due to crowding of the interior hydrogens. There is evidence that it has two isomeric forms of comparable stability (''trans'', ''cis'', ''trans'', ''cis'', ''trans'', ''trans'', ''cis''- with four interior hydrogens (shown in the infobox) and ''trans'', ''cis'', ''trans'', ''cis'', ''trans'', ''cis'', ''cis-'' with three interior hydrogens) which rapidly interconvert at room temperature b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

2.png)