|
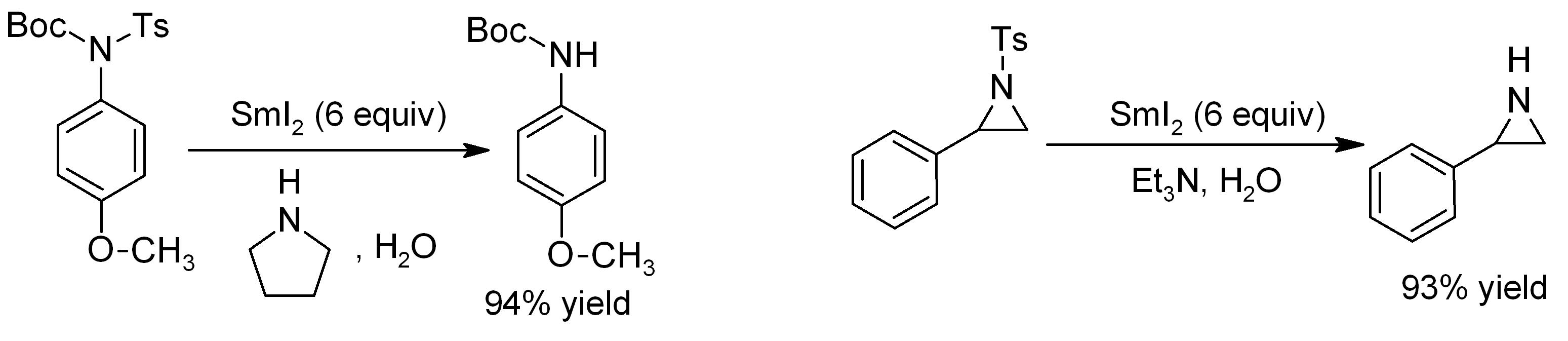
Annulation
In organic chemistry, annulation (; occasionally annelation) is a chemical reaction in which a new ring is constructed on a molecule. : Examples are the Robinson annulation, Danheiser annulation and certain cycloadditions. Annular molecules are constructed from side-on condensed cyclic segments, for example helicenes and acenes. In transannulation a bicyclic molecule is created by intramolecular carbon-carbon bond formation in a large monocyclic ring. An example is the samarium(II) iodide induced ketone - alkene cyclization of ''5-methylenecyclooctanone'' which proceeds through a ketyl intermediate: : Benzannulation The term benzannulated compounds refers to derivatives of cyclic compounds (usually aromatic) which are fused to a benzene Benzene is an Organic compound, organic chemical compound with the Chemical formula#Molecular formula, molecular formula C6H6. The benzene molecule is composed of six carbon atoms joined in a planar hexagonal Ring (chemistry), ring ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Robinson Annulation
The Robinson annulation is a chemical reaction used in organic chemistry for ring formation. It was discovered by Robert Robinson (organic chemist), Robert Robinson in 1935 as a method to create a six membered ring by forming three new carbon–carbon bonds. The method uses a ketone and a methyl vinyl ketone to form an α,β-unsaturated ketone in a cyclohexane ring by a Michael addition followed by an aldol condensation. This procedure is one of the key methods to form fused ring systems. Formation of cyclohexenone and derivatives are important in chemistry for their application to the synthesis of many natural products and other interesting organic compounds such as antibiotics and steroids. Specifically, the synthesis of cortisone is completed through the use of the Robinson annulation. The initial paper on the Robinson annulation was published by William Sage Rapson, William Rapson and Robert Robinson while Rapson studied at Oxford with professor Robinson. Before their work, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Danheiser Annulation
The Danheiser annulation or Danheiser TMS-cyclopentene annulation is an organic reaction of an α,β-unsaturated ketone and a trialkylsilylallene (e.g., trimethylsilyl- or triisopropylsilyl-) in the presence of a Lewis Acid to give a trialkylsilylcyclopentene in a regiocontrolled annulation In organic chemistry, annulation (; occasionally annelation) is a chemical reaction in which a new ring is constructed on a molecule. : Examples are the Robinson annulation, Danheiser annulation and certain cycloadditions. Annular molecules a .... References {{Organic reactions Organic reactions Name reactions ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Helicene
In organic chemistry, helicenes are aromatic ortho substituent, ortho-condensed Polycyclic compound, polycyclic Aromaticity, aromatic compounds in which Benzene, benzene rings or other aromatics are angularly annulation, annulated to give helix, helically-shaped chiral molecules. The chemistry of helicenes has attracted continuing attention because of their unique structural, Spectroscopy, spectral, and Optics, optical features. Structure and properties The systematic nomenclature, systematic naming for this class of compounds is based on the number of rings: [''n'']helicene is the structure consisting of ''n'' rings. According to IUPAC, only structures where ''n'' is at least 5 are considered helicenes. Some specific compounds also have alternate or trivial names. As the number of rings increases, starting at four, the structure becomes non-planar, but instead the planes of consecutive rings tilt to prevent steric collisions. For helicenes with six benzene units, a 360° turn is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Samarium(II) Iodide
Samarium(II) iodide is an inorganic compound with the formula SmI2. When employed as a solution for organic synthesis, it is known as Kagan's reagent. SmI2 is a green solid and forms a dark blue solution in THF. It is a strong one-electron reducing agent that is used in organic synthesis. Structure In solid samarium(II) iodide, the metal centers are seven-coordinate with a face-capped octahedral geometry. In its ether adducts, samarium remains heptacoordinate with five ether and two terminal iodide ligands. Preparation Samarium iodide is easily prepared in nearly quantitative yields from samarium metal and either diiodomethane or 1,2-diiodoethane. When prepared in this way, its solutions is most often used without purification of the inorganic reagent. Solid, solvent-free SmI2 forms by high temperature decomposition of samarium(III) iodide (SmI3).G. Jantsch, N. Skalla: "Zur Kenntnis der Halogenide der seltenen Erden. IV. – Über Samarium(II)jodid und den thermischen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Chemistry
Organic chemistry is a subdiscipline within chemistry involving the science, scientific study of the structure, properties, and reactions of organic compounds and organic matter, organic materials, i.e., matter in its various forms that contain carbon atoms.Clayden, J.; Greeves, N. and Warren, S. (2012) ''Organic Chemistry''. Oxford University Press. pp. 1–15. . Study of structure determines their structural formula. Study of properties includes Physical property, physical and Chemical property, chemical properties, and evaluation of Reactivity (chemistry), chemical reactivity to understand their behavior. The study of organic reactions includes the organic synthesis, chemical synthesis of natural products, drugs, and polymers, and study of individual organic molecules in the laboratory and via theoretical (in silico) study. The range of chemicals studied chemistry includes hydrocarbons (compounds containing only carbon and hydrogen) as well as compounds based on carbon, but a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isoquinoline
Isoquinoline is an individual chemical specimen - a heterocyclic aromatic organic compound - as well as the name of a family of many thousands of natural plant alkaloids, any one of which might be referred to as "an isoquinoline". It is a structural isomer of quinoline. Isoquinoline and quinoline are benzopyridines, which are composed of a benzene ring fused to a pyridine ring. In a broader sense, the term isoquinoline is used to make reference to isoquinoline derivatives. 1-Benzylisoquinoline is the structural backbone in many naturally occurring alkaloids such as papaverine. The isoquinoline ring in these natural compound derives from the aromatic amino acid tyrosine. Properties Isoquinoline is a colorless hygroscopic liquid at temperatures above its melting point with a penetrating, unpleasant odor. Impure samples can appear brownish, as is typical for nitrogen heterocycles. It crystallizes in form of platelets that have a low solubility in water but dissolve well in et ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyrene
Pyrene is a polycyclic aromatic hydrocarbon (PAH) consisting of four fused benzene rings, resulting in a flat aromatic system. The chemical formula is . This yellow-green solid is the smallest peri-fused PAH (one where the rings are fused through more than one face). Pyrene forms during incomplete combustion of organic compounds. Occurrence and properties Pyrene was first isolated from coal tar, where it occurs up to 2% by weight. As a peri-fused PAH, pyrene is much more resonance-stabilized than its five-member-ring containing isomer fluoranthene. Therefore, it is produced in a wide range of combustion conditions. For example, automobiles produce about 1 μg/km.Senkan, Selim and Castaldi, Marco (2003) "Combustion" in ''Ullmann's Encyclopedia of Industrial Chemistry'', Wiley-VCH, Weinheim. Reactions Oxidation with chromate affords perinaphthenone and then naphthalene-1,4,5,8-tetracarboxylic acid. Pyrene undergoes a series of hydrogenation reactions and is susceptible to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Quinoline
Quinoline is a heterocyclic aromatic organic compound with the chemical formula C9H7N. It is a colorless hygroscopic liquid with a strong odor. Aged samples, especially if exposed to light, become yellow and later brown. Quinoline is only slightly soluble in cold water but dissolves readily in hot water and most organic solvents. Quinoline itself has few applications, but many of its derivatives are useful in diverse applications. A prominent example is quinine, an alkaloid found in plants. Over 200 biologically active quinoline and quinazoline alkaloids are identified. 4-Hydroxy-2-alkylquinolines (HAQs) are involved in antibiotic resistance. Occurrence and isolation Quinoline was first extracted from coal tar in 1834 by German chemist Friedlieb Ferdinand Runge; he called quinoline ''leukol'' ("white oil" in Greek). Coal tar remains the principal source of commercial quinoline. In 1842, French chemist Charles Gerhardt obtained a compound by dry distilling quinine, st ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyridine
Pyridine is a basic (chemistry), basic heterocyclic compound, heterocyclic organic compound with the chemical formula . It is structurally related to benzene, with one methine group replaced by a nitrogen atom . It is a highly flammable, weakly alkaline, water-miscible liquid with a distinctive, unpleasant fish-like smell. Pyridine is colorless, but older or impure samples can appear yellow, due to the formation of extended, unsaturated Polymer, polymeric chains, which show significant electrical conductivity. The pyridine ring occurs in many important compounds, including agrochemicals, pharmaceuticals, and vitamins. Historically, pyridine was produced from coal tar. As of 2016, it is synthesized on the scale of about 20,000 tons per year worldwide. Properties Physical properties Pyridine is diamagnetism, diamagnetic. Its critical point (thermodynamics), critical parameters are: pressure 5.63 MPa, temperature 619 K and volume 248 cm3/mol. In the temperatur ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isochromene
4H-1-Benzopyran is an organic compound with the formula . It is one of two isomers of benzopyran, the other being 2H-1-benzopyran, which is more prevalent. It can be viewed as the fusion of a benzene ring to a heterocyclic pyran ring. Some benzopyrans have shown anticancerous activity ''in vitro''. The radical form of benzopyran is paramagnetic. The unpaired electron is delocalized over the whole benzopyran molecule, rendering it less reactive than one would expect otherwise. A similar example is the cyclopentadienyl radical. Commonly, benzopyran is encountered in the reduced state, in which it is partially saturated with one hydrogen atom, introducing a tetrahedral CH2 group in the pyran ring. Therefore, there are many structural isomers owing to the multiple possible positions of the oxygen atom and the tetrahedral carbon atom: Nomenclature According to current IUPAC nomenclature, the name chromene used in previous recommendations is retained; however, systematic ‘ben ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyran
In chemistry, pyran is a six-membered heterocyclic, non-aromatic ring, consisting of five carbon atoms and one oxygen atom and containing two double bonds. The molecular formula is C5H6O. There are two isomers of pyran that differ by the location of the double bonds. In 2''H''-pyran, the saturated carbon is at position 2, whereas, in 4''H''-pyran, the saturated carbon is at position 4. "Oxine” is not used for pyran because it has been used as a trivial name for quinolin-8-ol. 4''H''-Pyran was first isolated and characterized in 1962 via pyrolysis of 2-acetoxy-3,4-dihydro-2''H''-pyran. It was found to be unstable, particularly in the presence of air. 4''H''-pyran easily disproportionates to the corresponding dihydropyran and the pyrylium ion, which is easily hydrolyzed in aqueous medium. Although the pyrans themselves have little significance in chemistry, many of their derivatives are important biological molecules, such as the pyranoflavonoids. The term pyran is also often ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |