|
AFm Phase
An AFm phase is an "alumina, ferric oxide, monosubstituted" phase, or aluminate ferrite monosubstituted, or , mono, in cement chemist notation. AFm phases are important hydration products in the hydration of hydraulic cements. They are crystalline hydrates with general, simplified formula where n may be comprised between 13 and 19 water molecules. They form ''inter alia'' when tricalcium aluminate () reacts with dissolved calcium sulfate () or calcium carbonate (). As their sulfate form is the dominant one, they are often referred to as Aluminate Ferrite monosulfate or calcium aluminate monosulfate. However, carbonate-AFm phases also exist: monocarbonate and hemicarbonate. See also * Concrete degradation#Chloride attack * Layered double hydroxides (LDH) * Friedel's salt * Ettringite (AFt) * Pitting corrosion Pitting corrosion, or pitting, is a form of extremely localized corrosion that leads to the random creation of small holes in metal. The driving power for pitting co ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cement Chemist Notation
Cement chemist notation (CCN) was developed to simplify the formulas cement chemists use on a daily basis. It is a shorthand way of writing the chemical formula of oxides of calcium, silicon, and various metals. Abbreviations of oxides The main oxides present in cement (or in glass and ceramics) are abbreviated in the following way: Conversion of hydroxides in oxide and free water For the sake of mass balance calculations, hydroxides present in hydrated phases found in hardened cement paste, such as in portlandite, Ca(OH)2, must first be converted into oxide and water. To better understand the conversion process of hydroxide anions in oxide and water, it is necessary to consider the autoprotolysis of the hydroxyl anions; it implies a proton exchange between two OH−, like in a classical acid–base reaction: : + → + or also, :2 OH− → O2− + H2O For portlandite this gives thus the following mass balance: :Ca(OH)2 → CaO + H2O Thus portlandite can be written as Ca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pitting Corrosion
Pitting corrosion, or pitting, is a form of extremely localized corrosion that leads to the random creation of small holes in metal. The driving power for pitting corrosion is the depassivation of a small area, which becomes anodic (oxidation reaction) while an unknown but potentially vast area becomes cathodic (reduction reaction), leading to very localized galvanic corrosion. The corrosion penetrates the mass of the metal, with a limited diffusion of ions. Another term arises, pitting factor, which is defined as the ratio of the depth of the deepest pit (resulting due to corrosion) to the average penetration, which can be calculated based on the weight loss. Development and kinetics of pitting According to Frankel (1998) who performed a review on pitting corrosion, it develops in three successive steps: (or nucleation) by breakdown of the passive film protecting the metal surface from oxidation, (2) growth of metastable pits (growing up to the micron scale and then repassiva ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
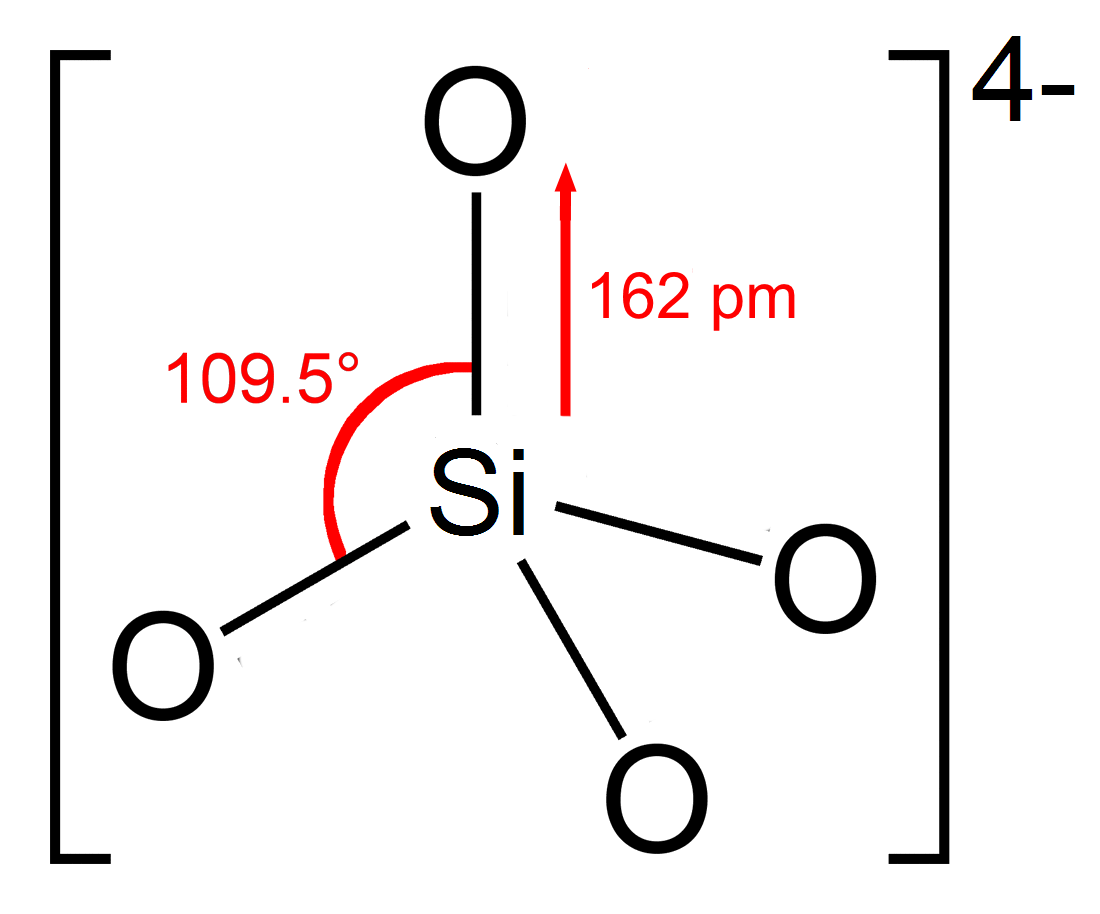
Silicates
In chemistry, a silicate is any member of a family of polyatomic anions consisting of silicon and oxygen, usually with the general formula , where . The family includes orthosilicate (), metasilicate (), and pyrosilicate (, ). The name is also used for any salt of such anions, such as sodium metasilicate; or any ester containing the corresponding chemical group, such as tetramethyl orthosilicate. The name "silicate" is sometimes extended to any anions containing silicon, even if they do not fit the general formula or contain other atoms besides oxygen; such as hexafluorosilicate .Most commonly, silicates are encountered as silicate minerals. For diverse manufacturing, technological, and artistic needs, silicates are versatile materials, both natural (such as granite, gravel, and garnet) and artificial (such as Portland cement, ceramics, glass, and waterglass). Structural principles In all silicates, silicon atom occupies the center of an idealized tetrahedron whose corner ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iron(III) Compounds
Iron () is a chemical element with symbol Fe (from la, ferrum) and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, right in front of oxygen (32.1% and 30.1%, respectively), forming much of Earth's outer and inner core. It is the fourth most common element in the Earth's crust. In its metallic state, iron is rare in the Earth's crust, limited mainly to deposition by meteorites. Iron ores, by contrast, are among the most abundant in the Earth's crust, although extracting usable metal from them requires kilns or furnaces capable of reaching or higher, about higher than that required to smelt copper. Humans started to master that process in Eurasia during the 2nd millennium BCE and the use of iron tools and weapons began to displace copper alloys, in some regions, only around 1200 BCE. That event is considered the transition from the Bronze Age to the Iron Age. In t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iron Compounds
Iron shows the characteristic chemical properties of the transition metals, namely the ability to form variable oxidation states differing by steps of one and a very large coordination and organometallic chemistry: indeed, it was the discovery of an iron compound, ferrocene, that revolutionalized the latter field in the 1950s.Greenwood and Earnshaw, p. 905 Iron is sometimes considered as a prototype for the entire block of transition metals, due to its abundance and the immense role it has played in the technological progress of humanity. Its 26 electrons are arranged in the configuration rd64s2, of which the 3d and 4s electrons are relatively close in energy, and thus it can lose a variable number of electrons and there is no clear point where further ionization becomes unprofitable. Iron forms compounds mainly in the oxidation states +2 (iron(II), "ferrous") and +3 (iron(III), "ferric"). Iron also occurs in higher oxidation states, e.g. the purple potassium ferrate (K2FeO4), ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrates
In chemistry, a hydrate is a substance that contains water Water (chemical formula ) is an inorganic, transparent, tasteless, odorless, and nearly colorless chemical substance, which is the main constituent of Earth's hydrosphere and the fluids of all known living organisms (in which it acts as a ... or its constituent elements. The chemical state of the water varies widely between different classes of hydrates, some of which were so labeled before their chemical structure was understood. Chemical nature Inorganic chemistry Hydrates are inorganic salts "containing water molecules combined in a definite ratio as an integral part of the crystal" that are either bound to a metal center or that have crystallized with the metal complex. Such hydrates are also said to contain ''water of crystallization'' or ''water of hydration''. If the water is heavy water in which the constituent hydrogen is the isotope deuterium, then the term ''deuterate'' may be used in place of ''hy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Concrete
Concrete is a composite material composed of fine and coarse aggregate bonded together with a fluid cement (cement paste) that hardens (cures) over time. Concrete is the second-most-used substance in the world after water, and is the most widely used building material. Its usage worldwide, ton for ton, is twice that of steel, wood, plastics, and aluminum combined. Globally, the ready-mix concrete industry, the largest segment of the concrete market, is projected to exceed $600 billion in revenue by 2025. This widespread use results in a number of environmental impacts. Most notably, the production process for cement produces large volumes of greenhouse gas emissions, leading to net 8% of global emissions. Other environmental concerns include widespread illegal sand mining, impacts on the surrounding environment such as increased surface runoff or urban heat island effect, and potential public health implications from toxic ingredients. Significant research and development is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cement
A cement is a binder, a chemical substance used for construction that sets, hardens, and adheres to other materials to bind them together. Cement is seldom used on its own, but rather to bind sand and gravel ( aggregate) together. Cement mixed with fine aggregate produces mortar for masonry, or with sand and gravel, produces concrete. Concrete is the most widely used material in existence and is behind only water as the planet's most-consumed resource. Cements used in construction are usually inorganic, often lime or calcium silicate based, which can be characterized as hydraulic or the less common non-hydraulic, depending on the ability of the cement to set in the presence of water (see hydraulic and non-hydraulic lime plaster). Hydraulic cements (e.g., Portland cement) set and become adhesive through a chemical reaction between the dry ingredients and water. The chemical reaction results in mineral hydrates that are not very water-soluble and so are quite durable in wa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aluminium Compounds
Aluminium (or aluminum) combines characteristics of pre- and post-transition metals. Since it has few available electrons for metallic bonding, like its heavier group 13 congeners, it has the characteristic physical properties of a post-transition metal, with longer-than-expected interatomic distances.Greenwood and Earnshaw, pp. 222–4 Furthermore, as Al3+ is a small and highly charged cation, it is strongly polarizing and aluminium compounds tend towards covalency;Greenwood and Earnshaw, pp. 224–7 this behaviour is similar to that of beryllium (Be2+), an example of a diagonal relationship.Greenwood and Earnshaw, pp. 112–3 However, unlike all other post-transition metals, the underlying core under aluminium's valence shell is that of the preceding noble gas, whereas for gallium and indium it is that of the preceding noble gas plus a filled d-subshell, and for thallium and nihonium it is that of the preceding noble gas plus filled d- and f-subshells. Hence, aluminium does not suf ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ettringite
Ettringite is a hydrous calcium aluminium sulfate mineral with formula: . It is a colorless to yellow mineral crystallizing in the trigonal system. The prismatic crystals are typically colorless, turning white on partial dehydration. It is part of the ettringite-group which includes other sulfates such as thaumasite and bentorite.Ettringite-group Mindat.org Discovery and occurrence  Ettringite was first described in 1874 by , for an occurrence near the Ettringer ,
Ettringite was first described in 1874 by , for an occurrence near the Ettringer , [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mineral Hydration
In chemistry, mineral hydration is an inorganic chemical reaction which adds water to the crystal structure of a mineral, usually creating a new mineral, usually called a ''hydrate''. In geological terms, the process of mineral hydration is known as ''retrograde alteration'' and is a process occurring in retrograde metamorphism. It commonly accompanies metasomatism and is often a feature of wall rock alteration around ore bodies. Hydration of minerals occurs generally in concert with hydrothermal circulation which may be driven by tectonic or igneous activity. Processes There are two main ways in which minerals hydrate. One is conversion of an oxide to a double hydroxide, as with the hydration of calcium oxide—CaO—to calcium hydroxide—Ca(OH)2, the other is with the incorporation of water molecules directly into the crystalline structure of a new mineral. The later process is exhibited in the hydration of feldspars to clay minerals, garnet to chlorite, or kyanite to m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Friedel's Salt
Friedel's salt is an anion exchanger mineral belonging to the family of the layered double hydroxides (LDHs). It has affinity for anions as chloride and iodide and is capable of retaining them to a certain extent in its crystallographical structure. Composition Friedel's salt is a layered double hydroxide (LDH) of general formula: : or more explicitly for a positively-charged LDH mineral: : or by directly incorporating water molecules into the Ca,Al hydroxide layer: : where chloride and hydroxide anions occupy the interlayer to compensate the excess of positive charges. In the cement chemist notation (CCN), considering that : and doubling all the stoichiometry, it could also be written in CCN as follows: : A simplified chemical composition with only Cl– in the interlayer, and without OH–, as: : can be also written in cement chemist notation as: : Friedel's salt is formed in cements initially rich in tri-calcium aluminate (C3A). Free-chloride ions directly ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

_chloride.jpg)


