|
3-methoxytyramine
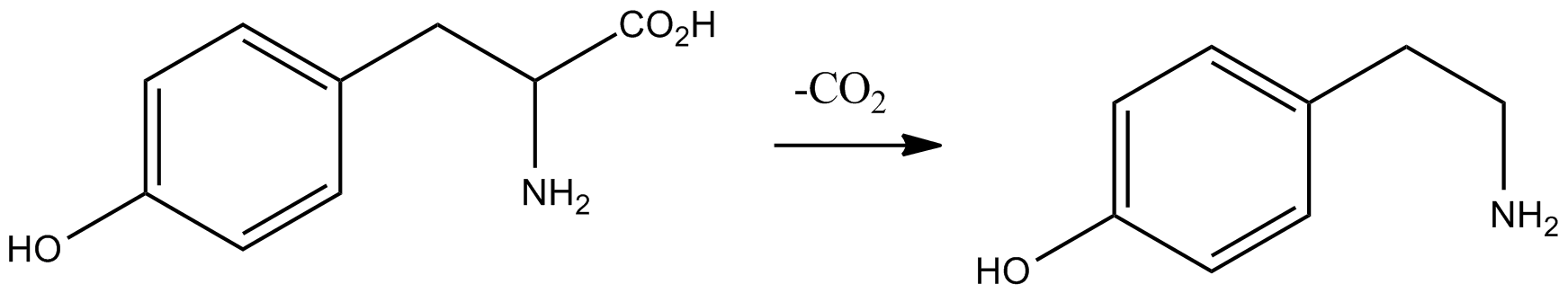
3-Methoxytyramine (3-MT), also known as 3-methoxy-4-hydroxyphenethylamine, is a human trace amine that occurs as a metabolite of the neurotransmitter dopamine. It is formed by the introduction of a methyl group to dopamine by the enzyme catechol-O-methyl transferase (COMT). 3-MT can be further metabolized by the enzyme monoamine oxidase (MAO) to form homovanillic acid (HVA), which is then typically excreted in the urine. Originally thought to be physiologically inactive, 3-MT has recently been shown to act as an agonist of human TAAR1. Occurrence 3-Methoxytyramine occurs naturally in the prickly pear cactus (genus ''Opuntia''), and is in general widespread throughout the Cactaceae. It has also been found in crown gall tumors on ''Nicotiana'' sp. In humans, 3-methoxytyramine is a trace amine that occurs as a metabolite of dopamine. See also * Tyramine Tyramine ( ) (also spelled tyramin), also known under several other names, is a naturally occurring trace amine d ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trace Amine
Trace amines are an endogenous group of trace amine-associated receptor 1 (TAAR1) agonists – and hence, monoaminergic neuromodulators – that are structurally and metabolically related to classical monoamine neurotransmitters. Compared to the classical monoamines, they are present in trace concentrations. They are distributed heterogeneously throughout the mammalian brain and peripheral nervous tissues and exhibit high rates of metabolism. Although they can be synthesized within parent monoamine neurotransmitter systems, there is evidence that suggests that some of them may comprise their own independent neurotransmitter systems. Trace amines play significant roles in regulating the quantity of monoamine neurotransmitters in the synaptic cleft of monoamine neurons with . They have well-characterized presynaptic ''amphetamine-like'' effects on these monoamine neurons via TAAR1 activation; specifically, by activating TAAR1 in neurons they promote the release and prevent reuptak ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Catechol-O-methyl Transferase
Catechol-''O''-methyltransferase (COMT; ) is one of several enzymes that degrade catecholamines (neurotransmitters such as dopamine, epinephrine, and norepinephrine), catecholestrogens, and various drugs and substances having a catechol structure. In humans, catechol-''O''-methyltransferase protein is encoded by the COMT gene. Two isoforms of COMT are produced: the soluble short form (S-COMT) and the membrane bound long form (MB-COMT). As the regulation of catecholamines is impaired in a number of medical conditions, several pharmaceutical drugs target COMT to alter its activity and therefore the availability of catecholamines. COMT was first discovered by the biochemist Julius Axelrod in 1957. Function Catechol-''O''-methyltransferase is involved in the inactivation of the catecholamine neurotransmitters (dopamine, epinephrine, and norepinephrine). The enzyme introduces a methyl group to the catecholamine, which is donated by S-adenosyl methionine (SAM). Any compound having a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trace Amines
Trace amines are an endogenous group of TAAR1 agonist, trace amine-associated receptor 1 (TAAR1) agonists – and hence, monoaminergic neuromodulators – that are structurally and metabolically related to classical monoamine neurotransmitters. Compared to the classical monoamines, they are present in trace concentrations. They are distributed heterogeneously throughout the mammalian brain and peripheral nervous tissues and exhibit high rates of metabolism. Although they can be synthesized within parent monoamine neurotransmitter systems, there is evidence that suggests that some of them may comprise their own independent neurotransmitter systems. Trace amines play significant roles in regulating the quantity of monoamine neurotransmitters in the synaptic cleft of monoamine neurons with . They have well-characterized presynaptic ''amphetamine-like'' effects on these monoamine neurons via TAAR1 activation; specifically, by activating TAAR1 in neurons they promote the release and p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dopamine
Dopamine (DA, a contraction of 3,4-dihydroxyphenethylamine) is a neuromodulatory molecule that plays several important roles in cells. It is an organic compound, organic chemical of the catecholamine and phenethylamine families. Dopamine constitutes about 80% of the catecholamine content in the brain. It is an amine synthesized by removing a carboxyl group from a molecule of its precursor (chemistry), precursor chemical, L-DOPA, which is biosynthesis, synthesized in the brain and kidneys. Dopamine is also synthesized in plants and most animals. In the brain, dopamine functions as a neurotransmitter—a chemical released by neurons (nerve cells) to send signals to other nerve cells. Neurotransmitters are synthesized in specific regions of the brain, but affect many regions systemically. The brain includes several distinct dopaminergic pathway, dopamine pathways, one of which plays a major role in the motivational component of reward system, reward-motivated behavior. The anticipa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cactaceae
A cactus (, or less commonly, cactus) is a member of the plant family Cactaceae, a family comprising about 127 genera with some 1750 known species of the order Caryophyllales. The word ''cactus'' derives, through Latin, from the Ancient Greek word (''káktos''), a name originally used by Theophrastus for a spiny plant whose identity is now not certain. Cacti occur in a wide range of shapes and sizes. Although some species live in quite humid environments, most cacti live in habitats subject to at least some drought. Many live in extremely dry environments, even being found in the Atacama Desert, one of the driest places on Earth. Because of this, cacti show many adaptations to conserve water. For example, almost all cacti are succulents, meaning they have thickened, fleshy parts adapted to store water. Unlike many other succulents, the stem is the only part of most cacti where this vital process takes place. Most species of cacti have lost true leaves, retaining only Thorns, s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
TAAR1 Agonists
Trace amine-associated receptor 1 (TAAR1) is a trace amine-associated receptor (TAAR) protein that in humans is encoded by the ''TAAR1'' gene. TAAR1 is an intracellular amine-activated and G protein-coupled receptor (GPCR) that is primarily expressed in several peripheral organs and cells (e.g., the stomach, small intestine, duodenum, and white blood cells), astrocytes, and in the intracellular milieu within the presynaptic plasma membrane (i.e., axon terminal) of monoamine neurons in the central nervous system (CNS). TAAR1 was discovered in 2001 by two independent groups of investigators, Borowski ''et al.'' and Bunzow ''et al.'' TAAR1 is one of six functional human trace amine-associated receptors, which are so named for their ability to bind endogenous amines that occur in tissues at trace concentrations. TAAR1 plays a significant role in regulating neurotransmission in dopamine, norepinephrine, and serotonin neurons in the CNS; it also affects immune system and neuroimmun ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phenols
In organic chemistry, phenols, sometimes called phenolics, are a class of chemical compounds consisting of one or more hydroxyl groups (— O H) bonded directly to an aromatic hydrocarbon group. The simplest is phenol, . Phenolic compounds are classified as simple phenols or polyphenols based on the number of phenol units in the molecule. Phenols are both synthesized industrially and produced by plants and microorganisms. Properties Acidity Phenols are more acidic than typical alcohols. The acidity of the hydroxyl group in phenols is commonly intermediate between that of aliphatic alcohols and carboxylic acids (their pKa is usually between 10 and 12). Deprotonation of a phenol forms a corresponding negative phenolate ion or phenoxide ion, and the corresponding salts are called phenolates or phenoxides (aryloxides according to the IUPAC Gold Book). Condensation with aldehydes and ketones Phenols are susceptible to Electrophilic aromatic substitutions. Condensation with formald ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phenethylamine Alkaloids
Phenethylamine (PEA) is an organic compound, natural product, natural monoamine alkaloid, and trace amine, which acts as a central nervous system stimulant in humans. In the brain, phenethylamine regulates monoamine neurotransmitter, monoamine neurotransmission by binding to trace amine-associated receptor 1 (TAAR1) and inhibiting vesicular monoamine transporter 2 (VMAT2) in monoamine neurons. To a lesser extent, it also acts as a neurotransmitter in the human central nervous system. In mammals, phenethylamine biosynthesis, is produced from the amino acid L-phenylalanine by the enzyme aromatic L-amino acid decarboxylase via enzymatic decarboxylation. In addition to its presence in mammals, phenethylamine is found in many other organisms and foods, such as chocolate, especially after microorganism, microbial fermentation (food), fermentation. Phenethylamine is sold as a dietary supplement for purported Mood (psychology), mood and weight loss-related therapeutic benefits; howeve ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phenethylamines
Substituted phenethylamines (or simply phenethylamines) are a chemical class of organic compounds that are based upon the phenethylamine structure; the class is composed of all the derivative compounds of phenethylamine which can be formed by replacing, or substituting, one or more hydrogen atoms in the phenethylamine core structure with substituents. The structural formula of any substituted phenethylamine contains a phenyl ring that is joined to an amino (NH) group via a two-carbon sidechain. Hence, any substituted phenethylamine can be classified according to the substitution of hydrogen (H) atoms on phenethylamine's phenyl ring, sidechain, or amino group with a specific group of atoms. Many substituted phenethylamines are psychoactive drugs which belong to a variety of different drug classes, including central nervous system stimulants (e.g., amphetamine), hallucinogens (e.g., dl- 2,5-dimethoxy-4-methylamphetamine DOM), entactogens (e.g., 3,4-methylenedioxyamphe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
3,4-Dimethoxyphenethylamine
3,4- Dimethoxy phenethylamine (DMPEA) is a chemical compound of the phenethylamine class. It is an analogue of the major human neurotransmitter dopamine where the 3- and 4-position hydroxy groups have been replaced with methoxy groups. It is also closely related to mescaline which is 3,4,5-trimethoxyphenethylamine. Chemistry One of the earliest syntheses of DMPEA (then referred to as "homoveratrylamine") was that of Pictet and Finkelstein, who made it in a multi-step sequence starting from vanillin. A similar sequence was subsequently reported by Buck and Perkin, as follows: : 3,4-Dimethoxybenzaldehyde (veratraldehyde) → 3,4-Dimethoxycinnamic acid → 3,4-Dimethoxyphenylpropionic acid → 3,4-Dimethoxyphenylpropionamide → 3,4-Dimethoxyphenethylamine A much shorter synthesis is given by Shulgin and Shulgin:A. Shulgin and A. Shulgin (1991). "PiHKAL A Chemical Love Story", pp. 614-616, Transform Press, Berkeley. Derivatives A known use was in the synthesis of Bevanto ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tyramine
Tyramine ( ) (also spelled tyramin), also known under several other names, is a naturally occurring trace amine derived from the amino acid tyrosine. Tyramine acts as a catecholamine releasing agent. Notably, it is unable to cross the blood-brain barrier, resulting in only non-psychoactive peripheral sympathomimetic effects following ingestion. A hypertensive crisis can result, however, from ingestion of tyramine-rich foods in conjunction with the use of monoamine oxidase inhibitors (MAOIs). Occurrence Tyramine occurs widely in plants and animals, and is metabolized by various enzymes, including monoamine oxidases. In foods, it often is produced by the decarboxylation of tyrosine during fermentation or decay. Foods that are fermented, cured, pickled, aged, or spoiled have high amounts of tyramine. Tyramine levels go up when foods are at room temperature or go past their freshness date. Specific foods containing considerable amounts of tyramine include: * strong or ag ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Crown Gall
''Agrobacterium'' is a genus of Gram-negative bacteria established by H. J. Conn that uses horizontal gene transfer to cause tumors in plants. ''Agrobacterium tumefaciens'' is the most commonly studied species in this genus. ''Agrobacterium'' is well known for its ability to transfer DNA between itself and plants, and for this reason it has become an important tool for genetic engineering. Nomenclatural History Leading up to the 1990s, the genus ''Agrobacterium'' was used as a wastebasket taxon. With the advent of 16S sequencing, many ''Agrobacterium'' species (especially the marine species) were reassigned to genera such as ''Ahrensia'', ''Pseudorhodobacter'', ''Ruegeria'', and ''Stappia''. The remaining ''Agrobacterium'' species were assigned to three biovars: biovar 1 (''Agrobacterium tumefaciens''), biovar 2 (''Agrobacterium rhizogenes''), and biovar 3 (''Agrobacterium vitis''). In the early 2000s, ''Agrobacterium'' was synonymized with the genus ''Rhizobium''. This move pr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |