separase on:
[Wikipedia]
[Google]
[Amazon]
Separase, also known as separin, is a cysteine protease responsible for triggering anaphase by hydrolysing cohesin, which is the
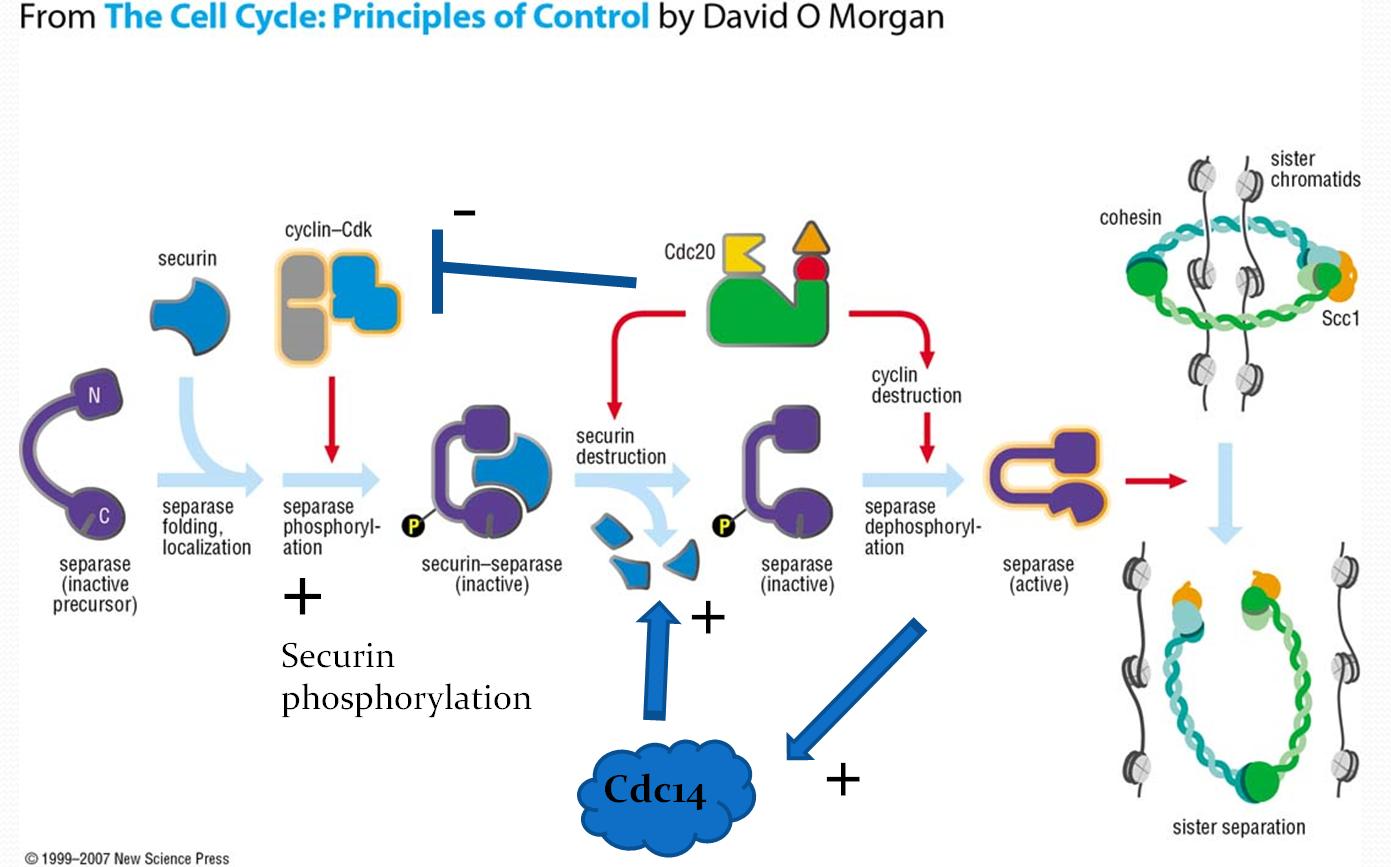
 When the cell is not dividing, separase is prevented from cleaving cohesin through its association with either securin or upon phosphorylation of a specific serine residue in separase by the cyclin-CDK complex. Separase phosphorylation leads to a stable association with CDK1-cyclin B1. Securin or CDK1-cyclin B binding is mutually exclusive. In both complexes, separase is inhibited by pseudosubstrate motifs that block substrate binding at the catalytic site and at nearby docking sites. However, while securin contains its own pseudosubstrate motifs to occlude substrate binding, the CDK1–cyclin B complex inhibits separase by rigidifying pseudosubstrate motifs from flexible loops in separase itself, leading to an auto-inhibition of the proteolytic activity of separase. Regulation through these distinct binding partners provides two layers of negative regulation to prevent inappropriate cohesin cleavage. Note that separase cannot function without initially forming the securin-separase complex in most organisms. This is because securin helps properly fold separase into the functional conformation. However, yeast does not appear to require securin to form functional separase because anaphase occurs in yeast even with a securin deletion.
On the signal for anaphase, securin is ubiquitinated and hydrolysed, releasing separase for dephosphorylation by the APC-Cdc20 complex. Active separase can then cleave Scc1 for release of the sister chromatids.
Separase initiates the activation of Cdc14 in early anaphase and Cdc14 has been found to dephosphorylate securin, thereby increasing its efficiency as a substrate for degradation. The presence of this positive feedback loop offers a potential mechanism for giving anaphase a more switch-like behavior.
When the cell is not dividing, separase is prevented from cleaving cohesin through its association with either securin or upon phosphorylation of a specific serine residue in separase by the cyclin-CDK complex. Separase phosphorylation leads to a stable association with CDK1-cyclin B1. Securin or CDK1-cyclin B binding is mutually exclusive. In both complexes, separase is inhibited by pseudosubstrate motifs that block substrate binding at the catalytic site and at nearby docking sites. However, while securin contains its own pseudosubstrate motifs to occlude substrate binding, the CDK1–cyclin B complex inhibits separase by rigidifying pseudosubstrate motifs from flexible loops in separase itself, leading to an auto-inhibition of the proteolytic activity of separase. Regulation through these distinct binding partners provides two layers of negative regulation to prevent inappropriate cohesin cleavage. Note that separase cannot function without initially forming the securin-separase complex in most organisms. This is because securin helps properly fold separase into the functional conformation. However, yeast does not appear to require securin to form functional separase because anaphase occurs in yeast even with a securin deletion.
On the signal for anaphase, securin is ubiquitinated and hydrolysed, releasing separase for dephosphorylation by the APC-Cdc20 complex. Active separase can then cleave Scc1 for release of the sister chromatids.
Separase initiates the activation of Cdc14 in early anaphase and Cdc14 has been found to dephosphorylate securin, thereby increasing its efficiency as a substrate for degradation. The presence of this positive feedback loop offers a potential mechanism for giving anaphase a more switch-like behavior.
{{NLM content Mitosis Cell biology EC 3.4.22
protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residue (biochemistry), residues. Proteins perform a vast array of functions within organisms, including Enzyme catalysis, catalysing metab ...
responsible for binding sister chromatids during the early stage of anaphase. In humans, separin is encoded by the ''ESPL1'' gene
In biology, the word gene has two meanings. The Mendelian gene is a basic unit of heredity. The molecular gene is a sequence of nucleotides in DNA that is transcribed to produce a functional RNA. There are two types of molecular genes: protei ...
.
History
In '' S. cerevisiae'', separase is encoded by the ''esp1'' gene. Esp1 was discovered byKim Nasmyth
Kim Ashley Nasmyth (born 18 October 1952) is an English geneticist, the Whitley Professor of Biochemistry at the University of Oxford, a Fellow of Trinity College, Oxford, former scientific director of the Research Institute of Molecular Pathol ...
and coworkers in 1998. In 2021, structures of human separase were determined in complex with either securin or CDK1-cyclin B1-CKS1 using cryo-EM by scientists of the University of Geneva.
Function
Stable cohesion between sister chromatids before anaphase and their timely separation during anaphase are critical for cell division and chromosome inheritance. In vertebrates, sister chromatid cohesion is released in 2 steps via distinct mechanisms. The first step involves phosphorylation of STAG1 or STAG2 in the cohesin complex. The second step involves cleavage of the cohesin subunit SCC1 (RAD21
Double-strand-break repair protein rad21 homolog is a protein that in humans is encoded by the ''RAD21'' gene. ''RAD21'' (also known as ''Mcd1'', ''Scc1'', ''KIAA0078'', ''NXP1'', ''HR21''), an essential gene, encodes a DNA repair#Double-strand b ...
) by separase, which initiates the final separation of sister chromatids.
In ''S. cerevisiae'', Esp1 is coded by ESP1 and is regulated by the securin Pds1. The two sister chromatids are initially bound together by the cohesin complex until the beginning of anaphase, at which point the mitotic spindle pulls the two sister chromatids apart, leaving each of the two daughter cells with an equivalent number of sister chromatids. The proteins that bind the two sister chromatids, disallowing any premature sister chromatid separation, are a part of the cohesin protein family. One of these cohesin proteins crucial for sister chromatid cohesion is Scc1. Esp1 is a separase protein that cleaves the cohesin subunit Scc1 (RAD21), allowing sister chromatids to separate at the onset of anaphase during mitosis.
Regulation
 When the cell is not dividing, separase is prevented from cleaving cohesin through its association with either securin or upon phosphorylation of a specific serine residue in separase by the cyclin-CDK complex. Separase phosphorylation leads to a stable association with CDK1-cyclin B1. Securin or CDK1-cyclin B binding is mutually exclusive. In both complexes, separase is inhibited by pseudosubstrate motifs that block substrate binding at the catalytic site and at nearby docking sites. However, while securin contains its own pseudosubstrate motifs to occlude substrate binding, the CDK1–cyclin B complex inhibits separase by rigidifying pseudosubstrate motifs from flexible loops in separase itself, leading to an auto-inhibition of the proteolytic activity of separase. Regulation through these distinct binding partners provides two layers of negative regulation to prevent inappropriate cohesin cleavage. Note that separase cannot function without initially forming the securin-separase complex in most organisms. This is because securin helps properly fold separase into the functional conformation. However, yeast does not appear to require securin to form functional separase because anaphase occurs in yeast even with a securin deletion.
On the signal for anaphase, securin is ubiquitinated and hydrolysed, releasing separase for dephosphorylation by the APC-Cdc20 complex. Active separase can then cleave Scc1 for release of the sister chromatids.
Separase initiates the activation of Cdc14 in early anaphase and Cdc14 has been found to dephosphorylate securin, thereby increasing its efficiency as a substrate for degradation. The presence of this positive feedback loop offers a potential mechanism for giving anaphase a more switch-like behavior.
When the cell is not dividing, separase is prevented from cleaving cohesin through its association with either securin or upon phosphorylation of a specific serine residue in separase by the cyclin-CDK complex. Separase phosphorylation leads to a stable association with CDK1-cyclin B1. Securin or CDK1-cyclin B binding is mutually exclusive. In both complexes, separase is inhibited by pseudosubstrate motifs that block substrate binding at the catalytic site and at nearby docking sites. However, while securin contains its own pseudosubstrate motifs to occlude substrate binding, the CDK1–cyclin B complex inhibits separase by rigidifying pseudosubstrate motifs from flexible loops in separase itself, leading to an auto-inhibition of the proteolytic activity of separase. Regulation through these distinct binding partners provides two layers of negative regulation to prevent inappropriate cohesin cleavage. Note that separase cannot function without initially forming the securin-separase complex in most organisms. This is because securin helps properly fold separase into the functional conformation. However, yeast does not appear to require securin to form functional separase because anaphase occurs in yeast even with a securin deletion.
On the signal for anaphase, securin is ubiquitinated and hydrolysed, releasing separase for dephosphorylation by the APC-Cdc20 complex. Active separase can then cleave Scc1 for release of the sister chromatids.
Separase initiates the activation of Cdc14 in early anaphase and Cdc14 has been found to dephosphorylate securin, thereby increasing its efficiency as a substrate for degradation. The presence of this positive feedback loop offers a potential mechanism for giving anaphase a more switch-like behavior.

References
Further reading
* * * *External links
* * https://web.archive.org/web/20041117073907/http://ncbi.nih.gov/entrez/query.fcgi?db=Nucleotide * Video by David Morgan explaining action of securin and separin (in MP4 format): http://media.hhmi.org/ibio/morgan/morgan_3.mp4 * and in other formats{{NLM content Mitosis Cell biology EC 3.4.22