Barton–Kellogg reaction on:
[Wikipedia]
[Google]
[Amazon]
The Barton–Kellogg reaction is a 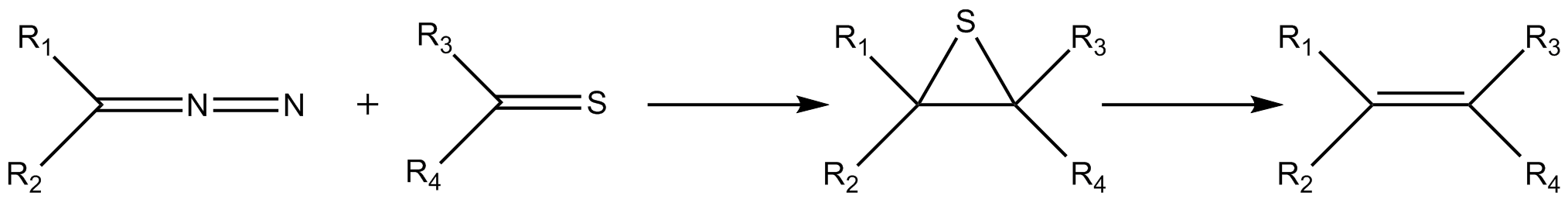 This reaction was pioneered by
This reaction was pioneered by

 The main advantage of this reaction over the
The main advantage of this reaction over the
coupling reaction A coupling reaction in organic chemistry is a general term for a variety of reactions where two fragments are joined together with the aid of a metal catalyst. In one important reaction type, a main group organometallic compound of the type R-M (R ...
between a diazo
The diazo group is an organic moiety consisting of two linked nitrogen atoms ( azo) at the terminal position. Overall charge neutral organic compounds containing the diazo group bound to a carbon atom are called diazo compounds or diazoalkanes ...
compound and a thioketone
In organic chemistry, thioketones (; also known as thiones or thiocarbonyls) are organosulfur compounds related to conventional ketones in which the oxygen has been replaced by a sulfur. Instead of a structure of , thioketones have the structure ...
, giving an alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
by way of an episulfide
In organic chemistry, episulfides are a class of organic compounds that contain a saturated, heterocyclic ring consisting of two carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetraval ...
intermediate. The Barton–Kellogg reaction is also known as Barton–Kellogg olefination and Barton olefin synthesis.
 This reaction was pioneered by
This reaction was pioneered by Hermann Staudinger
Hermann Staudinger (; 23 March 1881 – 8 September 1965) was a German organic chemist who demonstrated the existence of macromolecules, which he characterized as polymers. For this work he received the 1953 Nobel Prize in Chemistry.
He is also ...
, and also goes by the name Staudinger type diazo-thioketone coupling.
Reaction mechanism
In thereaction mechanism
In chemistry, a reaction mechanism is the step by step sequence of elementary reactions by which overall chemical change occurs.
A chemical mechanism is a theoretical conjecture that tries to describe in detail what takes place at each stage o ...
for this reaction, the diazo compound reacts as a 1,3-dipole
In organic chemistry, a 1,3-dipolar compound or 1,3-dipole is a dipolar compound with delocalized electrons and a separation of charge over three atoms. They are reactants in 1,3-dipolar cycloadditions.
The dipole has at least one resonance st ...
in a 1,3-dipolar cycloaddition with the thioketone to give a 5-membered thiadiazoline ring. This intermediate is unstable; it extrudes a molecule of nitrogen to form a thiocarbonyl ylide, which then cyclizes to form a stable episulfide
In organic chemistry, episulfides are a class of organic compounds that contain a saturated, heterocyclic ring consisting of two carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetraval ...
. Triphenylphosphine
Triphenylphosphine (IUPAC name: triphenylphosphane) is a common organophosphorus compound with the formula P(C6H5)3 and often abbreviated to P Ph3 or Ph3P. It is widely used in the synthesis of organic and organometallic compounds. PPh3 exists ...
reacts as a nucleophile
In chemistry, a nucleophile is a chemical species that forms bonds by donating an electron pair. All molecules and ions with a free pair of electrons or at least one pi bond can act as nucleophiles. Because nucleophiles donate electrons, they a ...
, opening the three-membered ring to form a sulfaphosphatane. In a manner similar to the Wittig reaction
The Wittig reaction or Wittig olefination is a chemical reaction of an aldehyde or ketone with a triphenyl phosphonium ylide called a Wittig reagent. Wittig reactions are most commonly used to convert aldehydes and ketones to alkenes. Most ...
, this structure then expels triphenylphosphine sulfide
Triphenylphosphine sulfide (IUPAC name: triphenyl-''λ''5-phosphanethione) is the organophosphorus compound with the formula , usually written (where Ph = phenyl). It is a colourless solid, which is soluble in a variety of organic solvents.
Struc ...
to produce an alkene.
Scope
The diazo compound can be obtained from aketone
In organic chemistry, a ketone is a functional group with the structure R–C(=O)–R', where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group –C(=O)– (which contains a carbon-oxygen double bon ...
by reaction with hydrazine
Hydrazine is an inorganic compound with the chemical formula . It is a simple pnictogen hydride, and is a colourless flammable liquid with an ammonia-like odour. Hydrazine is highly toxic unless handled in solution as, for example, hydrazin ...
to a hydrazone
Hydrazones are a class of organic compounds with the structure . They are related to ketones and aldehydes by the replacement of the oxygen =O with the = functional group. They are formed usually by the action of hydrazine on ketones or aldehydes. ...
followed by oxidation
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a ...
. Many reagents exist for this conversion for example silver(I) oxide
Silver oxide is the chemical compound with the formula Ag2O. It is a fine black or dark brown powder that is used to prepare other silver compounds.
Preparation
Silver oxide can be prepared by combining aqueous solutions of silver nitrate and ...
and (bis(trifluoroacetoxy)iodo)benzene
(Bis(trifluoroacetoxy)iodo)benzene, , is a hypervalent iodine compound used as a reagent in organic chemistry. It can be used to carry out the Hofmann rearrangement under acidic conditions.
Preparation
The syntheses of all aryl hypervalent iod ...
. The thioketone
In organic chemistry, thioketones (; also known as thiones or thiocarbonyls) are organosulfur compounds related to conventional ketones in which the oxygen has been replaced by a sulfur. Instead of a structure of , thioketones have the structure ...
required for this reaction can be obtained from a ketone and phosphorus pentasulfide
Phosphorus pentasulfide is the inorganic compound with the formula ( monomer) or ( dimer). This yellow solid is the one of two phosphorus sulfides of commercial value. Samples often appear greenish-gray due to impurities. It is soluble in carb ...
. Desulfurization of the episulfide can be accomplished by many phosphines and also by copper
Copper is a chemical element with the symbol Cu (from la, cuprum) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkish ...
powder.
McMurry reaction
The McMurry reaction is an organic reaction in which two ketone or aldehyde groups are coupled to form an alkene using a titanium chloride compound such as titanium(III) chloride and a reducing agent. The reaction is named after its co-discoverer, ...
is the notion that the reaction can take place with two different ketones. In this regard the diazo-thioketone coupling is a cross-coupling rather than a homocoupling.
References
# # # # # # # {{DEFAULTSORT:Barton-Kellogg Reaction Olefination reactions Carbon-carbon bond forming reactions Name reactions