|
Vulcanized Rubber
Vulcanization (British English: vulcanisation) is a range of processes for hardening rubbers. The term originally referred exclusively to the treatment of natural rubber with sulfur, which remains the most common practice. It has also grown to include the hardening of other (synthetic) rubbers via various means. Examples include silicone rubber via RTV silicone, room temperature vulcanizing and chloroprene rubber (neoprene) using metal oxides. Vulcanization can be defined as the curing (chemistry), curing of elastomers, with the terms 'vulcanization' and 'curing' sometimes used interchangeably in this context. It works by forming cross-links between sections of the polymer chain which results in increased rigidity and durability, as well as other changes in the mechanical and electrical properties of the material. Vulcanization, in common with the curing of other thermosetting polymers, is generally irreversible. The word was suggested by William Brockedon (a friend of Thomas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mesoamerican
Mesoamerica is a historical region and cultural area that begins in the southern part of North America and extends to the Pacific coast of Central America, thus comprising the lands of central and southern Mexico, all of Belize, Guatemala, El Salvador, and parts of Honduras, Nicaragua and northwestern part of Costa Rica. As a cultural area, Mesoamerica is defined by a mosaic of cultural traits developed and shared by its indigenous cultures. In the pre-Columbian era, many indigenous societies flourished in Mesoamerica for more than 3,000 years before the Spanish colonization of the Americas began on Hispaniola in 1493. In world history, Mesoamerica was the site of two historical transformations: (i) primary urban generation, and (ii) the formation of New World cultures from the mixtures of the indigenous Mesoamerican peoples with the European, African, and Asian peoples who were introduced by the Spanish colonization of the Americas. Mesoamerica is one of the six areas in the w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polysulfide
Polysulfides are a class of chemical compounds derived from anionic chains of sulfur atoms. There are two main classes of polysulfides: inorganic and organic. The inorganic polysulfides have the general formula . These anions are the conjugate bases of polysulfanes . Organic polysulfides generally have the formulae , where R is an alkyl or aryl group. Polysulfide salts and complexes The alkali metal polysulfides arise by treatment of a solution of the sulfide with elemental sulfur, e.g. sodium sulfide to sodium polysulfide: In some cases, these anions have been obtained as organic salts, which are soluble in organic solvents. The energy released in the reaction of sodium and elemental sulfur is the basis of battery technology. The sodium–sulfur battery and the lithium–sulfur battery require high temperatures to maintain liquid polysulfide and -conductive membranes that are unreactive toward sodium, sulfur, and sodium sulfide. Polysulfides are ligands in coordination ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkene
In organic chemistry, an alkene, or olefin, is a hydrocarbon containing a carbon–carbon double bond. The double bond may be internal or at the terminal position. Terminal alkenes are also known as Alpha-olefin, α-olefins. The International Union of Pure and Applied Chemistry (IUPAC) Preferred IUPAC name, recommends using the name "alkene" only for Open-chain compound, acyclic hydrocarbons with just one double bond; alkadiene, alkatriene, etc., or polyene for acyclic hydrocarbons with two or more double bonds; cycloalkene, cycloalkadiene, etc. for Cyclic compound, cyclic ones; and "olefin" for the general class – cyclic or acyclic, with one or more double bonds. Acyclic alkenes, with only one double bond and no other functional groups (also known as mono-enes) form a homologous series of hydrocarbons with the general formula with ''n'' being a >1 natural number (which is two hydrogens less than the corresponding alkane). When ''n'' is four or more, isomers are possible, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Allyl
In organic chemistry, an allyl group is a substituent with the structural formula . It consists of a methylene bridge () attached to a vinyl group (). The name is derived from the scientific name for garlic, . In 1844, Theodor Wertheim isolated an allyl derivative from garlic oil and named it "". The term allyl applies to many compounds related to , some of which are of practical or of everyday importance, for example, allyl chloride. Allylation is any chemical reaction that adds an allyl group to a Substrate (chemistry), substrate. Nomenclature A site adjacent to the unsaturated carbon atom is called the allylic position or allylic site. A group attached at this site is sometimes described as allylic. Thus, "has an allylic hydroxyl group". Allylic Carbon–hydrogen bond, C−H bonds are about 15% weaker than the C−H bonds in ordinary Orbital hybridisation, sp3 carbon centers and are thus more reactive. Benzylic and allylic are related in terms of structure, bond strength ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Styrene-butadiene
Styrene-butadiene or styrene-butadiene rubber (SBR) describe families of synthetic rubbers derived from styrene and butadiene (the version developed by Goodyear is called Neolite). These materials have good abrasion resistance and good aging stability when protected by additives. In 2012, more than 5.4 million tonnes of SBR were processed worldwide. About 50% of car tires are made from various types of SBR. The styrene/butadiene ratio influences the properties of the polymer: with high styrene content, the rubbers are harder and less rubbery. SBR is not to be confused with the thermoplastic elastomer, styrene-butadiene block copolymer, although being derived from the same monomers. Types SBR is derived from two monomers, styrene and butadiene. The mixture of these two monomers is polymerized by two processes: from solution (S-SBR) or as an emulsion (E-SBR). E-SBR is more widely used. Emulsion polymerization E-SBR produced by emulsion polymerization is initiated by f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polyisoprene
Polyisoprene is, strictly speaking, a collective name for polymers that are produced by polymerization of isoprene. In practice polyisoprene is commonly used to refer to synthetic ''cis''-1,4-polyisoprene, made by the industrial polymerisation of isoprene. Natural forms of polyisoprene are also used in substantial quantities, the most important being "natural rubber" (mostly ''cis''-1,4-polyisoprene), which is derived from the sap of trees. Both synthetic polyisoprene and natural rubber are highly elastic and consequently used to make tires and a variety of other applications. The ''trans'' isomer, which is much harder than the ''cis'' isomer, has also seen significant use in the past. It too has been synthesised and extracted from plant sap, the latter resin being known as gutta-percha. These were widely used as an electrical insulator and as components of golf balls. Annual worldwide production of synthetic polyisoprene was 13 million tons in 2007 and 16 million tons in 202 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
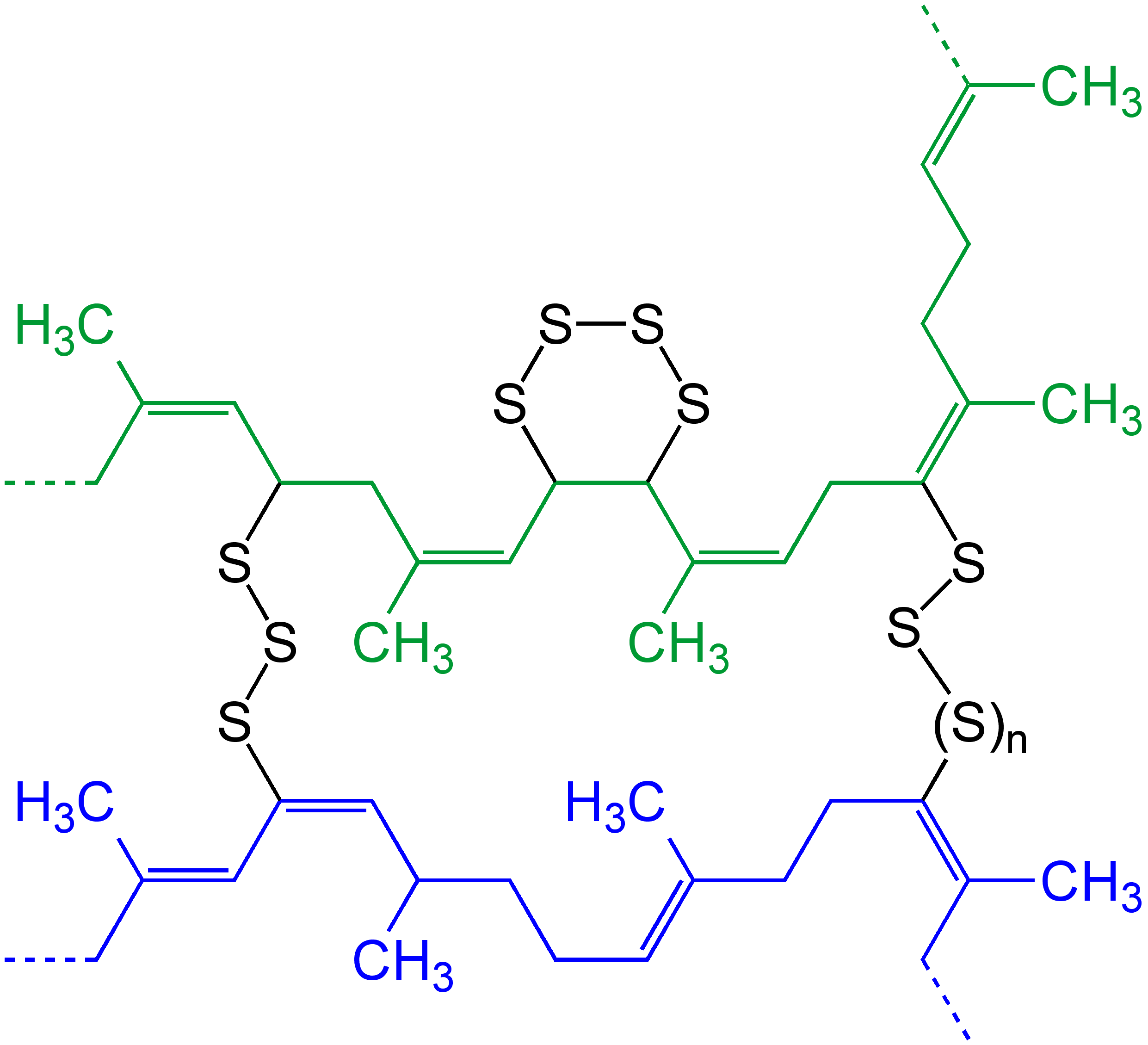
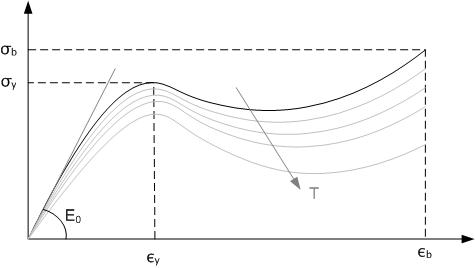
Sulfur Vulcanization
Sulfur vulcanization is a chemical process for converting natural rubber or related polymers into materials of varying hardness, elasticity, and mechanical durability by heating them with sulfur or sulfur-containing compounds. Sulfur forms cross-linking bridges between sections of polymer chains which affects the mechanical properties. Many products are made with vulcanized rubber, including tires, shoe soles, hoses, and conveyor belts. The term is derived from Vulcan (mythology), Vulcan, the Roman god of fire. The main polymers subjected to sulfur vulcanization are polyisoprene (natural rubber, NR), polybutadiene rubber (BR) and styrene-butadiene rubber (SBR), and ethylene propylene diene monomer rubber (EPDM rubber). All of these materials contain allyl, alkene groups adjacent to methylene groups. Other specialty rubbers may also be vulcanized, such as nitrile rubber (NBR) and butyl rubber (IIR). Vulcanization, in common with the curing of other thermosetting polymers, is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polyolefin
A polyolefin is a type of polymer with the general formula (CH2CHR)n where R is an alkyl group. They are usually derived from a small set of simple olefins (alkenes). Dominant in a commercial sense are polyethylene and polypropylene. More specialized polyolefins include Polyisobutene, polyisobutylene and polymethylpentene. They are all colorless or white oils or solids. Many copolymers are known, such as polybutene, which derives from a mixture of different butene isomers. The name of each polyolefin indicates the olefin from which it is prepared; for example, polyethylene is derived from ethylene, and polymethylpentene is derived from 4-methyl-1-pentene. Polyolefins are not olefins themselves because the double bond of each olefin monomer is opened in order to form the polymer. Monomers having more than one double bond such as butadiene and isoprene yield polymers that contain double bonds (polybutadiene and polyisoprene) and are usually not considered polyolefins. Polyolefins are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
The Employment Of Women In Britain, 1914-1918 Q28267
''The'' is a grammatical article in English, denoting nouns that are already or about to be mentioned, under discussion, implied or otherwise presumed familiar to listeners, readers, or speakers. It is the definite article in English. ''The'' is the most frequently used word in the English language; studies and analyses of texts have found it to account for seven percent of all printed English-language words. It is derived from gendered articles in Old English which combined in Middle English and now has a single form used with nouns of any gender. The word can be used with both singular and plural nouns, and with a noun that starts with any letter. This is different from many other languages, which have different forms of the definite article for different genders or numbers. Pronunciation In most dialects, "the" is pronounced as (with the voiced dental fricative followed by a schwa) when followed by a consonant sound, and as (homophone of the archaic pronoun ''thee' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Peroxide
In chemistry, peroxides are a group of Chemical compound, compounds with the structure , where the R's represent a radical (a portion of a complete molecule; not necessarily a free radical) and O's are single oxygen atoms. Oxygen atoms are joined to each other and to adjacent elements through Single bond, single covalent bonds, denoted by dashes or lines. The group in a peroxide is often called the peroxide group, though some nomenclature discrepancies exist. This linkage is recognized as a common polyatomic ion, and exists in many molecules. General structure The characteristic structure of any regular peroxide is the oxygen–oxygen covalent single bond, which connects the two main atoms together. In the event that the molecule has no chemical Substituent, substituents, the peroxide group will have a [−2] Formal charge, net charge. Each oxygen atom has a charge of negative one, as 5 of its Valence electron, valence electrons remain in the outermost Atomic orbital, orbital ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thermoplastic
A thermoplastic, or thermosoftening plastic, is any plastic polymer material that becomes pliable or moldable at a certain elevated temperature and solidifies upon cooling. Most thermoplastics have a high molecular weight. The polymer chains associate by intermolecular forces, which weaken rapidly with increased temperature, yielding a viscous liquid. In this state, thermoplastics may be reshaped, and are typically used to produce parts by various polymer processing techniques such as injection molding, compression molding, calendering, and extrusion. Thermoplastics differ from thermosetting polymers (or "thermosets"), which form irreversible chemical bonds during the curing process. Thermosets do not melt when heated, but typically decompose and do not reform upon cooling. Above its glass transition temperature and below its melting point, the physical properties of a thermoplastic change drastically without an associated phase change. Some thermoplastics do not fully ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |