|
Octene
Octene is an alkene with the formula . Several isomers of octene are known, depending on the position and the geometry of the double bond in the carbon chain. The simplest isomer is 1-octene, an alpha-olefin used primarily as a co-monomer in production of polyethylene via the solution polymerization process. Several useful structural isomers of the octenes are obtained by dimerization of isobutene and 1-butene. These branched alkenes are used to alkylate phenols to give precursors to detergents.Helmut Fiege, Heinz-Werner Voges, Toshikazu Hamamoto, Sumio Umemura, Tadao Iwata, Hisaya Miki, Yasuhiro Fujita, Hans-Josef Buysch, Dorothea Garbe, Wilfried Paulus "Phenol Derivatives" in Ullmann's Encyclopedia of Industrial Chemistry 2002, Wiley-VCH, Weinheim.. The dimerisation of isobutane generates diisobutene Diisobutene (also known as Diisobutylene and Isooctene) refers to a pair of organic compounds with the overall formula C8H16. The isomers have the same carbon skeleton but diffe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkene
In organic chemistry, an alkene, or olefin, is a hydrocarbon containing a carbon–carbon double bond. The double bond may be internal or at the terminal position. Terminal alkenes are also known as Alpha-olefin, α-olefins. The International Union of Pure and Applied Chemistry (IUPAC) Preferred IUPAC name, recommends using the name "alkene" only for Open-chain compound, acyclic hydrocarbons with just one double bond; alkadiene, alkatriene, etc., or polyene for acyclic hydrocarbons with two or more double bonds; cycloalkene, cycloalkadiene, etc. for Cyclic compound, cyclic ones; and "olefin" for the general class – cyclic or acyclic, with one or more double bonds. Acyclic alkenes, with only one double bond and no other functional groups (also known as mono-enes) form a homologous series of hydrocarbons with the general formula with ''n'' being a >1 natural number (which is two hydrogens less than the corresponding alkane). When ''n'' is four or more, isomers are possible, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isomers
In chemistry, isomers are molecules or polyatomic ions with identical molecular formula – that is, the same number of atoms of each element – but distinct arrangements of atoms in space. ''Isomerism'' refers to the existence or possibility of isomers. Isomers do not necessarily share similar chemical or physical properties. Two main forms of isomerism are structural (or constitutional) isomerism, in which '' bonds'' between the atoms differ; and stereoisomerism (or spatial isomerism), in which the bonds are the same but the ''relative positions'' of the atoms differ. Isomeric relationships form a hierarchy. Two chemicals might be the same constitutional isomer, but upon deeper analysis be stereoisomers of each other. Two molecules that are the same stereoisomer as each other might be in different conformational forms or be different isotopologues. The depth of analysis depends on the field of study or the chemical and physical properties of interest. The English word ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
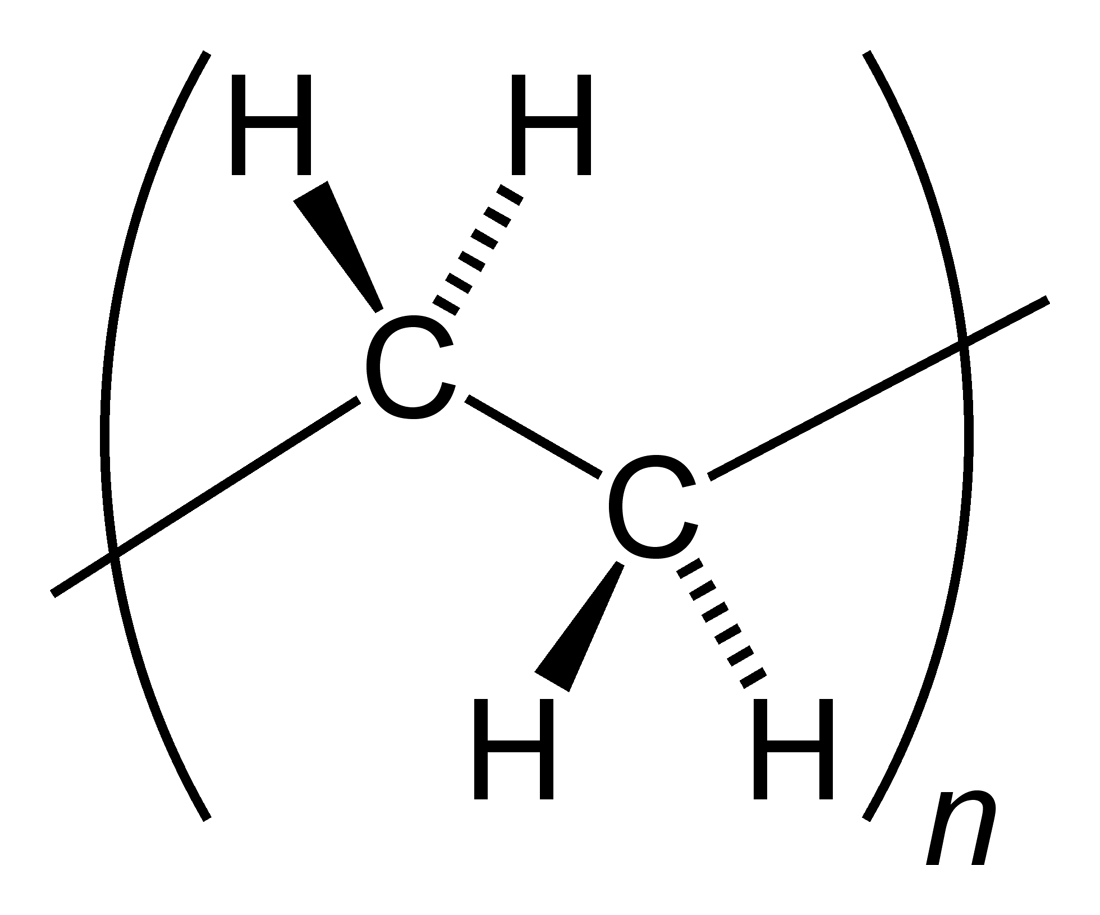
Polyethylene
Polyethylene or polythene (abbreviated PE; IUPAC name polyethene or poly(methylene)) is the most commonly produced plastic. It is a polymer, primarily used for packaging (plastic bags, plastic films, geomembranes and containers including bottles, cups, jars, etc.). , over 100 million tonnes of polyethylene resins are being produced annually, accounting for 34% of the total plastics market. Many kinds of polyethylene are known, with most having the chemical formula (C2H4)''n''. PE is usually a mixture of similar polymers of ethylene, with various values of ''n''. It can be ''low-density'' or ''high-density'' and many variations thereof. Its properties can be modified further by crosslinking or copolymerization. All forms are nontoxic as well as chemically resilient, contributing to polyethylene's popularity as a multi-use plastic. However, polyethylene's chemical resilience also makes it a long-lived and decomposition-resistant pollutant when disposed of improperly. Being a h ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Monomer
A monomer ( ; ''mono-'', "one" + '' -mer'', "part") is a molecule that can react together with other monomer molecules to form a larger polymer chain or two- or three-dimensional network in a process called polymerization. Classification Chemistry classifies monomers by type, and two broad classes based on the type of polymer they form. By type: * natural vs synthetic, e.g. glycine vs caprolactam, respectively * polar vs nonpolar, e.g. vinyl acetate vs ethylene, respectively * cyclic vs linear, e.g. ethylene oxide vs ethylene glycol, respectively By type of polymer they form: * those that participate in condensation polymerization * those that participate in addition polymerization Differing stoichiometry causes each class to create its respective form of polymer. : The polymerization of one kind of monomer gives a homopolymer. Many polymers are copolymers, meaning that they are derived from two different monomers. In the case of condensation polymerizations, t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diisobutene
Diisobutene (also known as Diisobutylene and Isooctene) refers to a pair of organic compounds with the overall formula C8H16. The isomers have the same carbon skeleton but differ in the location of the C=C bond. Both are colorless liquids with very similar physical properties. These compounds arise via the acid catalysis, acid catalyzed dimerization of isobutene, a reaction that proceeds via the carbocation . The process also leads to some triisobutenes and tetraisobutenes. Applications Hydrogenation is performed at a significant scale to give isooctane, which is an important fuel additive. Diisobutene is used as precursors to isononylol and octylphenols by hydroformylation/hydrogenation and phenol alkylation, respectively. Both are precursors to plasticizers. The isononylol (3,5,5-trimethyl-hexan-1-ol) is a precursor to 3,5,5-trimethylhexyl acetate, a commercial fragrance. Diisobutenes were once of interest as components for automotive fuels. See also * 1-Octene - the corres ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
C8H16
The molecular formula C8H16 (molar mass: 112.21 g/mol, exact mass: 112.1252 u) may refer to: * Cyclooctane * Methylcycloheptane * Dimethylcyclohexanes * * Octene Octene is an alkene with the formula . Several isomers of octene are known, depending on the position and the geometry of the double bond in the carbon chain. The simplest isomer is 1-octene, an alpha-olefin used primarily as a co-monomer in p ...s ** 1-Octene {{MolFormDisambig Molecular formulas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Double Bond
In chemistry, a double bond is a covalent bond between two atoms involving four bonding electrons as opposed to two in a single bond. Double bonds occur most commonly between two carbon atoms, for example in alkenes. Many double bonds exist between two different elements: for example, in a carbonyl group between a carbon atom and an oxygen atom. Other common double bonds are found in azo compounds (N=N), imines (C=N), and sulfoxides (S=O). In a skeletal formula, a double bond is drawn as two parallel lines (=) between the two connected atoms; typographically, the equals sign is used for this. Double bonds were introduced in chemical notation by Russian chemist Alexander Butlerov. Double bonds involving carbon are stronger and shorter than single bonds. The bond order is two. Double bonds are also electron-rich, which makes them potentially more reactive in the presence of a strong electron acceptor (as in addition reactions of the halogens). File:Ethene structural.svg, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alpha-olefin
In organic chemistry, terminal alkenes (alpha-olefins, α-olefins, or 1-alkenes) are a family of organic compounds which are alkenes (also known as olefins) with a chemical formula , distinguished by having a double bond at the primary, Alpha and beta carbon, alpha (α), or Locant#Numeric locants, 1- position.''Petrochemicals in Nontechnical Language'', 3rd Edition, Donald L. Burdick and William L. Leffler, This location of a double bond enhances the reactivity of the compound and makes it useful for a number of applications. Classification There are two types of alpha-olefins, branched alpha olefin, branched and linear alpha olefin, linear (or normal). The chemical properties of branched alpha-olefins with a branch at either the second (vinylidene) or the third carbon number are significantly different from the properties of linear alpha-olefins and those with branches on the fourth carbon number and further from the start of the chain. Examples of linear alpha-olefins are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polymerization
In polymer chemistry, polymerization (American English), or polymerisation (British English), is a process of reacting monomer molecules together in a chemical reaction to form polymer chains or three-dimensional networks. There are many forms of polymerization and different systems exist to categorize them. In chemical compounds, polymerization can occur via a variety of reaction mechanisms that vary in complexity due to the functional groups present in the reactants and their inherent steric effects. In more straightforward polymerizations, alkenes form polymers through relatively simple radical reactions; in contrast, reactions involving substitution at a carbonyl group require more complex synthesis due to the way in which reactants polymerize. As alkenes can polymerize in somewhat straightforward radical reactions, they form useful compounds such as polyethylene and polyvinyl chloride (PVC), which are produced in high tonnages each year due to their usefulnes ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Structural Isomers
In chemistry, a structural isomer (or constitutional isomer in the IUPAC nomenclature) of a chemical compound, compound is a compound that contains the same number and type of atoms, but with a different connectivity (i.e. arrangement of chemical bond, bonds) between them. The term metamer was formerly used for the same concept. For example, butanol , methyl propyl ether , and diethyl ether have the same molecular formula but are three distinct structural isomers. The concept applies also to polyatomic ions with the same total charge. A classical example is the cyanate ion and the fulminate ion . It is also extended to ionic compounds, so that (for example) ammonium cyanate and urea are considered structural isomers,William F. Bynum, E. Janet Browne, Roy Porter (2014)''Dictionary of the History of Science'' page 218. and so are methylammonium formate and ammonium acetate . Structural isomerism is the most radical type of isomerism. It is opposed to stereoisomerism, in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |