|
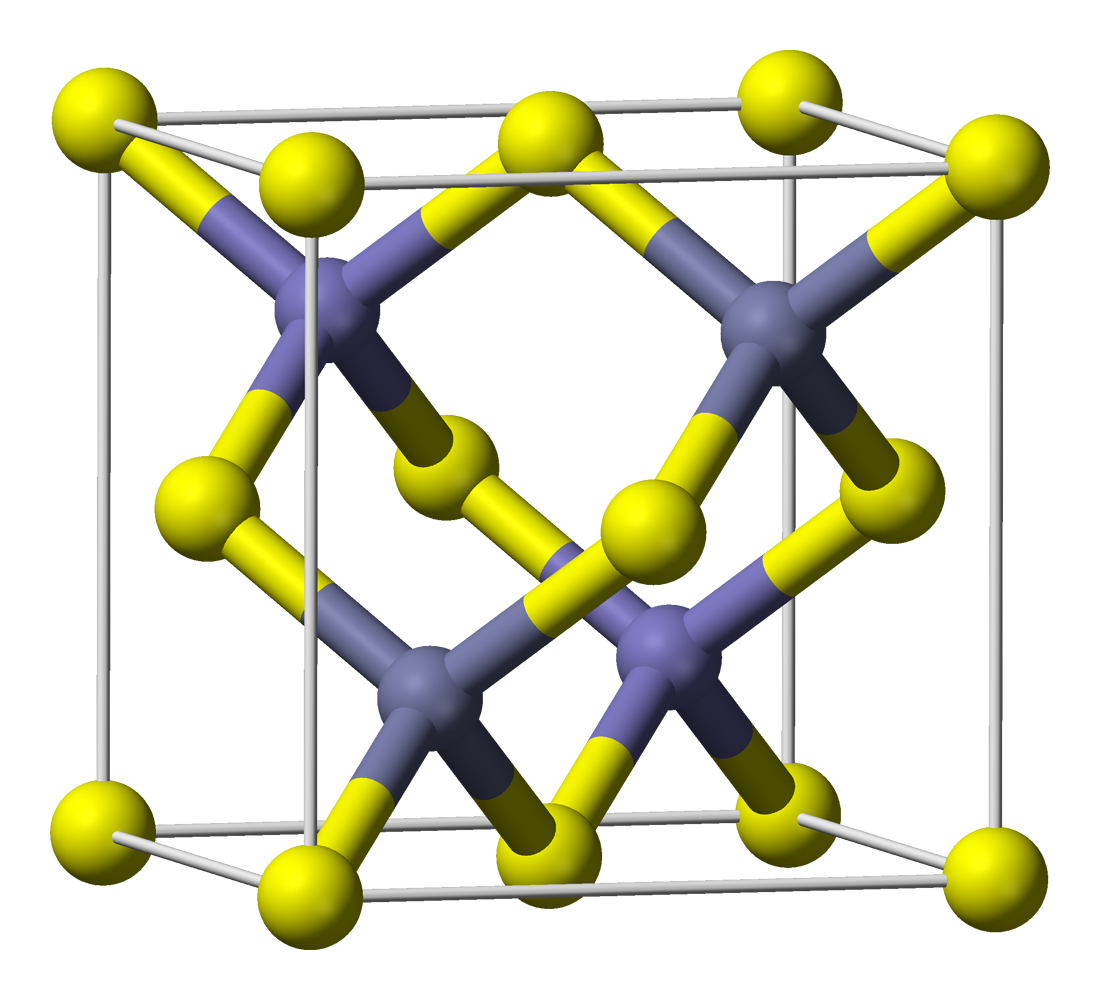
Terbium Phosphide
Terbium phosphide is an inorganic compound of terbium and phosphorus with the chemical formula TbP. Synthesis TbP can be obtained by the reaction of terbium and red phosphorus at 800–1000 °C: ::4 Tb + P4 → 4 TbP The compound can also be obtained by the reaction of sodium phosphide and anhydrous terbium chloride at 700~800 °C. Physical properties TbP undergoes a phase transition at 40 GPa from a NaCl-structure to a CsCl-structure. The compound can be sintered with zinc sulfide to make a green phosphor layer. TbP forms crystals of a cubic system, space group ''Fm''3''m''. Uses The compound is a semiconductor used in high power, high frequency applications and in laser diodes and other photo diodes. References {{Phosphides Phosphides Terbium compounds Semiconductors Rock salt crystal structure ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Terbium Nitride
Terbium nitride is a binary inorganic compound of terbium and nitrogen with the chemical formula . Physical properties Terbium nitride crystalyzes with cubic crystal system of the space group of ''F3m3''. Uses Terbium nitride is used for high-end electronics, ceramics, luminescent materials, special metallurgy, petrochemical, artificial crystal, magnetic materials. References Nitrides Terbium compounds Nitrogen compounds {{inorganic-compound-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phase Transition
In physics, chemistry, and other related fields like biology, a phase transition (or phase change) is the physical process of transition between one state of a medium and another. Commonly the term is used to refer to changes among the basic State of matter, states of matter: solid, liquid, and gas, and in rare cases, plasma (physics), plasma. A phase of a thermodynamic system and the states of matter have uniform physical property, physical properties. During a phase transition of a given medium, certain properties of the medium change as a result of the change of external conditions, such as temperature or pressure. This can be a discontinuous change; for example, a liquid may become gas upon heating to its boiling point, resulting in an abrupt change in volume. The identification of the external conditions at which a transformation occurs defines the phase transition point. Types of phase transition States of matter Phase transitions commonly refer to when a substance tran ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Terbium Compounds
Terbium compounds are Chemical compound, compounds formed by the lanthanide metal terbium (Tb). Terbium generally exhibits the +3 oxidation state in these compounds, such as in terbium(III) chloride, TbCl3, terbium(III) nitrate, Tb(NO3)3 and Tb(CH3COO)3. Compounds with terbium in the +4 oxidation state are also known, such as terbium(IV) oxide, TbO2 and BaTbF6. Terbium can also form compounds in the 0, +1 and +2 oxidation states. The trivalent terbium ion (Tb3+) is generally colorless in aqueous solution, and when it is irradiated by certain wavelengths of ultraviolet light (such as 254 nm or 365 nm) in solution or crystal form, it will emit green fluorescence. This property has given rise to applications in fields such as optics. The tetravalent terbium ion (Tb4+) is non-luminescent and its coexistence with Tb3+ will reduce the green emission of Tb3+. Properties of terbium compounds Chalcogenides Oxides Terbium has a variety of oxides. The most easily obtai ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphides
In chemistry, a phosphide is a compound containing the ion or its equivalent. Many different phosphides are known, with widely differing structures. Most commonly encountered on the binary phosphides, i.e. those materials consisting only of phosphorus and a less electronegative element. Numerous are polyphosphides, which are solids consisting of anionic chains or clusters of phosphorus. Phosphides are known with the majority of less electronegative elements with the exception of Hg, Pb, Sb, Bi, Te, and Po.Von Schnering, H.G. and Hönle , W. (1994) "Phosphides - Solid-state Chemistry" in ''Encyclopedia of Inorganic Chemistry''. R. Bruce King (ed.). John Wiley & Sons Finally, some phosphides are molecular. Binary phosphides Binary phosphides include phosphorus and one other element. An example of a group 1 phosphide is sodium phosphide (). Other notable examples include aluminium phosphide () and calcium phosphide (), which are used as pesticides, exploiting their tend ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Photo Diode
A photodiode is a semiconductor diode sensitive to photon radiation, such as visible light, infrared or ultraviolet radiation, X-rays and gamma rays. It produces an electrical current when it absorbs photons. This can be used for detection and measurement applications, or for the generation of electrical power in solar cells. Photodiodes are used in a wide range of applications throughout the electromagnetic spectrum from visible light photocells to gamma ray spectrometers. Principle of operation A photodiode is a PIN diode, PIN structure or p–n junction. When a photon of sufficient energy strikes the diode, it creates an electron–electron hole, hole pair. This mechanism is also known as the inner photoelectric effect. If the absorption occurs in the junction's depletion region, or one diffusion length away from it, these carriers are swept from the junction by the built-in electric field of the depletion region. Thus holes move toward the anode, and electrons toward the cath ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Laser Diode
file:Laser diode chip.jpg, The laser diode chip removed and placed on the eye of a needle for scale A laser diode (LD, also injection laser diode or ILD or semiconductor laser or diode laser) is a semiconductor device similar to a light-emitting diode in which a diode pumped directly with electrical current can create active laser medium, lasing conditions at the diode's p–n junction, junction. Driven by voltage, the doped p–n-transition allows for carrier generation and recombination, recombination of an electron with a electron hole, hole. Due to the drop of the electron from a higher energy level to a lower one, radiation is generated in the form of an emitted photon. This is spontaneous emission. Stimulated emission can be produced when the process is continued and further generates light with the same phase, coherence, and wavelength. The choice of the semiconductor material determines the wavelength of the emitted beam, which in today's laser diodes range from the inf ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Semiconductor
A semiconductor is a material with electrical conductivity between that of a conductor and an insulator. Its conductivity can be modified by adding impurities (" doping") to its crystal structure. When two regions with different doping levels are present in the same crystal, they form a semiconductor junction. The behavior of charge carriers, which include electrons, ions, and electron holes, at these junctions is the basis of diodes, transistors, and most modern electronics. Some examples of semiconductors are silicon, germanium, gallium arsenide, and elements near the so-called " metalloid staircase" on the periodic table. After silicon, gallium arsenide is the second-most common semiconductor and is used in laser diodes, solar cells, microwave-frequency integrated circuits, and others. Silicon is a critical element for fabricating most electronic circuits. Semiconductor devices can display a range of different useful properties, such as passing current more easil ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Space Group
In mathematics, physics and chemistry, a space group is the symmetry group of a repeating pattern in space, usually in three dimensions. The elements of a space group (its symmetry operations) are the rigid transformations of the pattern that leave it unchanged. In three dimensions, space groups are classified into 219 distinct types, or 230 types if chiral copies are considered distinct. Space groups are discrete cocompact groups of isometries of an oriented Euclidean space in any number of dimensions. In dimensions other than 3, they are sometimes called Bieberbach groups. In crystallography, space groups are also called the crystallographic or Fedorov groups, and represent a description of the symmetry of the crystal. A definitive source regarding 3-dimensional space groups is the ''International Tables for Crystallography'' . History Space groups in 2 dimensions are the 17 wallpaper groups which have been known for several centuries, though the proof that the list ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cubic System
In crystallography, the cubic (or isometric) crystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals. There are three main varieties of these crystals: *Primitive cubic (abbreviated ''cP'' and alternatively called simple cubic) *Body-centered cubic (abbreviated ''cI'' or bcc) *Face-centered cubic (abbreviated ''cF'' or fcc) Note: the term fcc is often used in synonym for the ''cubic close-packed'' or ccp structure occurring in metals. However, fcc stands for a face-centered cubic Bravais lattice, which is not necessarily close-packed when a motif is set onto the lattice points. E.g. the diamond and the zincblende lattices are fcc but not close-packed. Each is subdivided into other variants listed below. Although the ''unit cells'' in these crystals are conventionally taken to be cubes, the primitive unit cells often are not. Bravais lattices The three Bravais latices in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zinc Sulfide
Zinc sulfide (or zinc sulphide) is an inorganic compound with the chemical formula of ZnS. This is the main form of zinc found in nature, where it mainly occurs as the mineral sphalerite. Although this mineral is usually black because of various impurities, the pure material is white, and it is widely used as a pigment. In its dense synthetic form, zinc sulfide can be transparency and translucency, transparent, and it is used as a window for visible light, visible optics and infrared optics. Structure ZnS exists in two main crystalline forms. This dualism is an example of polymorphism (materials science), polymorphism. In each form, the coordination geometry at Zn and S is tetrahedral. The more stable cubic form is known also as zinc blende or sphalerite. The hexagonal form is known as the mineral wurtzite, although it also can be produced synthetically.. The transition from the sphalerite form to the wurtzite form occurs at around 1020 °C. Applications Luminescent mate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Physics Letters A
''Physics Letters'' was a scientific journal published from 1962 to 1966, when it split in two series now published by Elsevier Elsevier ( ) is a Dutch academic publishing company specializing in scientific, technical, and medical content. Its products include journals such as ''The Lancet'', ''Cell (journal), Cell'', the ScienceDirect collection of electronic journals, ...: *''Physics Letters A'': condensed matter physics, theoretical physics, nonlinear science, statistical physics, mathematical and computational physics, general and cross-disciplinary physics (including foundations), atomic, molecular and cluster physics, plasma and fluid physics, optical physics, biological physics and nanoscience. *''Physics Letters B'': nuclear physics, theoretical nuclear physics, experimental high-energy physics, theoretical high-energy physics, and astrophysics. ''Physics Letters B'' is part of the SCOAP3 initiative. References See also * List of periodicals published by Elsevier ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |