|
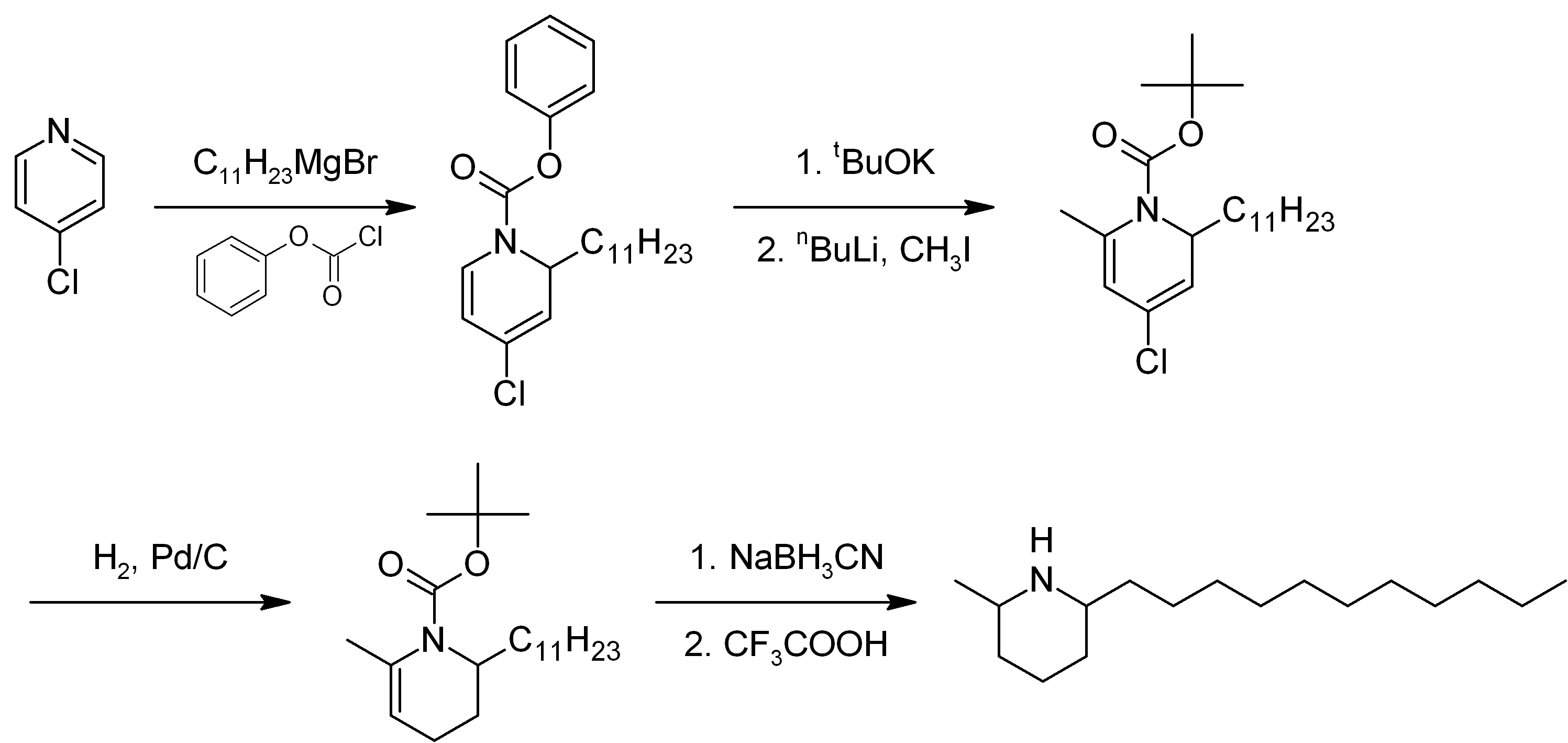
Solenopsin
Solenopsin is a lipophilic alkaloid with the molecular formula C17H35N found in the venom of fire ants (''Solenopsis''). It is considered the primary toxin in the venom and may be the component responsible for the cardiorespiratory failure in people who experience excessive fire ant stings. Structurally solenopsins are a piperidine ring with a methyl group substitution at position 2 and a long hydrophobic chain at position 6. They are typically oily at room temperature, water-insoluble, and present an absorbance peak at 232 nanometers. Fire ant venom contains other chemically related piperidines which make purification of solenopsin from ants difficult. Therefore, solenopsin and related compounds have been the target of organic synthesis from which pure compounds can be produced for individual study. Originally synthesized in 1993, several groups have designed novel and creative methods of synthesizing enantiopure solenopsin and other alkaloidal components of ant venom. To ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkaloid
Alkaloids are a broad class of natural product, naturally occurring organic compounds that contain at least one nitrogen atom. Some synthetic compounds of similar structure may also be termed alkaloids. Alkaloids are produced by a large variety of organisms including bacteria, fungus, fungi, Medicinal plant, plants, and animals. They can be purified from crude extracts of these organisms by acid-base extraction, or solvent extractions followed by silica-gel column chromatography. Alkaloids have a wide range of pharmacology, pharmacological activities including antimalarial medication, antimalarial (e.g. quinine), asthma, antiasthma (e.g. ephedrine), chemotherapy, anticancer (e.g. omacetaxine mepesuccinate, homoharringtonine), cholinomimetic (e.g. galantamine), vasodilation, vasodilatory (e.g. vincamine), Antiarrhythmic agent, antiarrhythmic (e.g. quinidine), analgesic (e.g. morphine), antibacterial (e.g. chelerythrine), and anti-diabetic, antihyperglycemic activities (e.g. berb ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fire Ant
Fire ants are several species of ants in the genus ''Solenopsis'', which includes over 200 species. ''Solenopsis'' are stinging ants, and most of their common names reflect this, for example, ginger ants and tropical fire ants. Many of the names shared by this genus are often used interchangeably to refer to other species of ant, such as the term red ant, mostly because of their similar coloration despite not being in the genus ''Solenopsis''. Both '' Myrmica rubra'' and '' Pogonomyrmex barbatus'' are common examples of non-Solenopsis ants being termed red ants. None of these common names apply to all species of ''Solenopsis'' nor exclusively to species of ''Solenopsis''; for example, several species of weaver ants of the genus '' Oecophylla'' in Southeast Asia are colloquially called "fire ants" because of their similar coloration and painful bites, but the two genera are not closely related. '' Wasmannia auropunctata'' is another unrelated ant more commonly called the "littl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Piperidine
Piperidine is an organic compound with the molecular formula (CH2)5NH. This heterocyclic amine consists of a six-membered ring containing five methylene bridges (–CH2–) and one amine bridge (–NH–). It is a colorless liquid with an odor described as objectionable, typical of amines. The name comes from the genus name '' Piper'', which is the Latin word for pepper. Although piperidine is a common organic compound, it is best known as a representative structure element within many pharmaceuticals and alkaloids, such as natural-occurring solenopsins. Production Piperidine was first reported in 1850 by the Scottish chemist Thomas Anderson and again, independently, in 1852 by the French chemist Auguste Cahours, who named it. Both of them obtained piperidine by reacting piperine with nitric acid. Industrially, piperidine is produced by the hydrogenation of pyridine, usually over a molybdenum disulfide catalyst: : C5H5N + 3 H2 → C5H10NH Pyridine can also be reduce ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitric Oxide Synthase
Nitric oxide synthases (NOSs) are a family of enzymes catalyzing the production of nitric oxide (NO) from L-arginine. NO is an important cellular signaling molecule. It helps modulate vascular tone, insulin secretion, airway tone, and peristalsis, and is involved in angiogenesis and neural development. It may function as a retrograde neurotransmitter. Nitric oxide is mediated in mammals by the calcium-calmodulin controlled isoenzymes eNOS (endothelial NOS) and nNOS (neuronal NOS). The inducible isoform, iNOS, involved in immune response, binds calmodulin at physiologically relevant concentrations, and produces NO as an immune defense mechanism, as NO is a free radical with an unpaired electron. It is the proximate cause of septic shock and may function in autoimmune disease. In the context of eukaryote biology, ''nitric oxide synthase'' refers to nitric-oxide synthase (NADPH) (), which catalyzes the reaction: * 2 L-arginine + 3 NADPH + 3 H+ + 4 O2 \rightleftharpoons 2 citrulli ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Insecticide
Insecticides are pesticides used to kill insects. They include ovicides and larvicides used against insect eggs and larvae, respectively. The major use of insecticides is in agriculture, but they are also used in home and garden settings, industrial buildings, for vector control, and control of insect parasites of animals and humans. Acaricides, which kill mites and ticks, are not strictly insecticides, but are usually classified together with insecticides. Some insecticides (including common bug sprays) are effective against other non-insect arthropods as well, such as scorpions, spiders, etc. Insecticides are distinct from insect repellents, which repel but do not kill. Sales In 2016 insecticides were estimated to account for 18% of worldwide pesticide sales. Worldwide sales of insecticides in 2018 were estimated as $ 18.4 billion, of which 25% were neonicotinoids, 17% were pyrethroids, 13% were diamides, and the rest were many other classes which sold for less th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Angiogenesis
Angiogenesis is the physiological process through which new blood vessels form from pre-existing vessels, formed in the earlier stage of vasculogenesis. Angiogenesis continues the growth of the vasculature mainly by processes of sprouting and splitting, but processes such as coalescent angiogenesis, vessel elongation and vessel cooption also play a role. Vasculogenesis is the embryonic formation of endothelial cells from mesoderm cell precursors, and from neovascularization, although discussions are not always precise (especially in older texts). The first vessels in the developing embryo form through vasculogenesis, after which angiogenesis is responsible for most, if not all, blood vessel growth during development and in disease. Angiogenesis is a normal and vital process in growth and development, as well as in wound healing and in the formation of granulation tissue. However, it is also a fundamental step in the transition of tumors from a benign state to a malign ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
In Vitro
''In vitro'' (meaning ''in glass'', or ''in the glass'') Research, studies are performed with Cell (biology), cells or biological molecules outside their normal biological context. Colloquially called "test-tube experiments", these studies in biology and its subdisciplines are traditionally done in labware such as test tubes, flasks, Petri dishes, and microtiter plates. Studies conducted using components of an organism that have been isolated from their usual biological surroundings permit a more detailed or more convenient analysis than can be done with whole organisms; however, results obtained from ''in vitro'' experiments may not fully or accurately predict the effects on a whole organism. In contrast to ''in vitro'' experiments, ''in vivo'' studies are those conducted in living organisms, including humans, known as clinical trials, and whole plants. Definition ''In vitro'' (Latin language, Latin for "in glass"; often not italicized in English usage) studies are conducted ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphoinositide 3-kinase
Phosphoinositide 3-kinases (PI3Ks), also called phosphatidylinositol 3-kinases, are a family of enzymes involved in cellular functions such as cell growth, proliferation, differentiation, motility, survival and intracellular trafficking, which in turn are involved in cancer. PI3Ks are a family of related intracellular signal transducer enzymes capable of phosphorylating the 3 position hydroxyl group of the inositol ring of phosphatidylinositol (PtdIns). The pathway, with oncogene PIK3CA and tumor suppressor gene PTEN, is implicated in the sensitivity of cancer tumors to insulin and IGF1, and in calorie restriction. Discovery The discovery of PI3Ks by Lewis Cantley and colleagues began with their identification of a previously unknown phosphoinositide kinase associated with the polyoma middle T protein. They observed unique substrate specificity and chromatographic properties of the products of the lipid kinase, leading to the discovery that this phosphoinositide kinas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lipophilicity
Lipophilicity (from Greek λίπος "fat" and φίλος "friendly") is the ability of a chemical compound to dissolve in fats, oils, lipids, and non-polar solvents such as hexane or toluene. Such compounds are called lipophilic (translated as "fat-loving" or "fat-liking"). Such non-polar solvents are themselves lipophilic, and the adage "like dissolves like" generally holds true. Thus lipophilic substances tend to dissolve in other lipophilic substances, whereas hydrophilic ("water-loving") substances tend to dissolve in water and other hydrophilic substances. Lipophilicity, hydrophobicity, and non-polarity may describe the same tendency towards participation in the London dispersion force, as the terms are often used interchangeably. However, the terms "lipophilic" and "hydrophobic" are not synonymous, as can be seen with silicones and fluorocarbons, which are hydrophobic but not lipophilic. __TOC__ Surfactants Hydrocarbon-based surfactants are compounds that are amph ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Arginine
Arginine is the amino acid with the formula (H2N)(HN)CN(H)(CH2)3CH(NH2)CO2H. The molecule features a guanidinium, guanidino group appended to a standard amino acid framework. At physiological pH, the carboxylic acid is deprotonated (−CO2−) and both the amino and guanidino groups are protonated, resulting in a cation. Only the -arginine (symbol Arg or R) enantiomer is found naturally. Arg residues are common components of proteins. It is Genetic code, encoded by the DNA codon table, codons CGU, CGC, CGA, CGG, AGA, and AGG. The guanidine group in arginine is the Precursor (chemistry), precursor for the biosynthesis of nitric oxide. Like all amino acids, it is a white, water-soluble solid. The one-letter symbol R was assigned to arginine for its phonetic similarity. History Arginine was first isolated in 1886 from Lupinus luteus, yellow lupin seedlings by the German chemist Ernst Schulze (chemist), Ernst Schulze and his assistant Ernst Steiger. He named it from the Greek ''árg ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
2,6-Lutidine
2,6-Lutidine is a natural heterocyclic aromatic organic compound with the formula (CH3)2C5H3N. It is one of several dimethyl-substituted derivative of pyridine, all of which are referred to as Lutidine, lutidines. It is a colorless liquid with mildly basic properties and a pungent, noxious odor. Occurrence and production It was first isolated from the base (chemistry), basic fraction of coal tar and from Dippel's oil, bone oil. A laboratory route involves condensation of ethyl acetoacetate, formaldehyde, and an ammonia source to give a bis(carboxy ester) of a 2,6-dimethyl-1,4-dihydropyridine, which, after hydrolysis, undergoes decarboxylation. It is produced industrially by the reaction of formaldehyde, acetone, and ammonia. Uses 2,6-Lutidine has been evaluated for use as a food additive owing to its nutty aroma when present in solution at very low concentrations. Due to the steric effects of the two methyl groups, 2,6-lutidine is less nucleophilic than pyridine. Protonatio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |