|
Silver Sulfide
Silver sulfide is an inorganic compound with the formula . A dense black solid, it is the only sulfide of silver. It is useful as a photosensitizer in photography. It constitutes the tarnish that forms over time on silverware and other silver objects. Silver sulfide is insoluble in most solvents, but is degraded by strong acids. Silver sulfide is a network solid made up of silver (electronegativity of 1.98) and sulfur (electronegativity of 2.58) where the bonds have low ionic character (approximately 10%). Formation Silver sulfide naturally occurs as the tarnish on silverware. When combined with silver, hydrogen sulfide gas creates a layer of black silver sulfide patina on the silver, protecting the inner silver from further conversion to silver sulfide. Silver whiskers can form when silver sulfide forms on the surface of silver electrical contacts operating in an atmosphere rich in hydrogen sulfide and high humidity. Such atmospheres can exist in sewage treatment and paper m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Cyanide
Hydrogen cyanide (formerly known as prussic acid) is a chemical compound with the chemical formula, formula HCN and structural formula . It is a highly toxic and flammable liquid that boiling, boils slightly above room temperature, at . HCN is produced on an industrial scale and is a highly valued Precursor (chemistry), precursor to many chemical compounds ranging from polymers to pharmaceuticals. Large-scale applications are for the production of potassium cyanide and adiponitrile, used in mining and plastics, respectively. It is more toxic than solid cyanide compounds due to its Volatility (chemistry), volatile nature. A solution of hydrogen cyanide in water (molecule), water, represented as HCN(aqueous, aq), is called ''hydrocyanic acid''. The Salt (chemistry), salts of the cyanide anion are known as cyanides. Whether hydrogen cyanide is an organic compound or not is a topic of debate among chemists, and opinions vary from author to author. Traditionally, it is considered ino ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solubility
In chemistry, solubility is the ability of a chemical substance, substance, the solute, to form a solution (chemistry), solution with another substance, the solvent. Insolubility is the opposite property, the inability of the solute to form such a solution. The extent of the solubility of a substance in a specific solvent is generally measured as the concentration of the solute in a wikt:saturated#Chemistry, saturated solution, one in which no more solute can be dissolved. At this point, the two substances are said to be at the solubility equilibrium. For some solutes and solvents, there may be no such limit, in which case the two substances are said to be "miscibility, miscible in all proportions" (or just "miscible"). The solute can be a solid, a liquid, or a gas, while the solvent is usually solid or liquid. Both may be pure substances, or may themselves be solutions. Gases are always miscible in all proportions, except in very extreme situations,J. de Swaan Arons and G. A. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfides
Sulfide (also sulphide in British English) is an inorganic anion of sulfur with the chemical formula S2− or a compound containing one or more S2− ions. Solutions of sulfide salts are corrosive. ''Sulfide'' also refers to large families of inorganic and organic compounds, e.g. lead sulfide and dimethyl sulfide. Hydrogen sulfide (H2S) and bisulfide (HS−) are the conjugate acids of sulfide. Chemical properties The sulfide ion does not exist in aqueous alkaline solutions of Na2S. Instead sulfide converts to hydrosulfide: :S2− + H2O → SH− + OH− Upon treatment with an acid, sulfide salts convert to hydrogen sulfide: :S2− + H+ → SH− :SH− + H+ → H2S Oxidation of sulfide is a complicated process. Depending on the conditions, the oxidation can produce elemental sulfur, polysulfides, polythionates, sulfite, or sulfate. Metal sulfides react with halogens, forming sulfur and metal salts. :8 MgS + 8 I2 → S8 + 8 MgI2 Metal derivati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Qualitative Inorganic Analysis
Classical qualitative inorganic analysis is a method of analytical chemistry which seeks to find the elemental composition of inorganic compounds. It is mainly focused on detecting ions in an aqueous solution, therefore materials in other forms may need to be brought to this state before using standard methods. The solution is then treated with various reagents to test for reactions characteristic of certain ions, which may cause color change, precipitation and other visible changes. Qualitative inorganic analysis is that branch or method of analytical chemistry which seeks to establish the elemental composition of inorganic compounds through various reagents. Physical appearance of some inorganic compounds Detecting cations According to their properties, cations are usually classified into six groups. Each group has a common reagent which can be used to separate them from the solution. To obtain meaningful results, the separation must be done in the sequence specified bel ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Michael Faraday
Michael Faraday (; 22 September 1791 – 25 August 1867) was an English chemist and physicist who contributed to the study of electrochemistry and electromagnetism. His main discoveries include the principles underlying electromagnetic induction, diamagnetism, and electrolysis. Although Faraday received little formal education, as a self-made man, he was one of the most influential scientists in history. It was by his research on the magnetic field around a Electrical conductor, conductor carrying a direct current that Faraday established the concept of the electromagnetic field in physics. Faraday also established that magnetism could Faraday effect, affect rays of light and that there was an underlying relationship between the two phenomena. the 1911 ''Encyclopædia Britannica''. He similarly discovered the principles of electromagnetic induction, diamagnetism, and the Faraday's laws of electrolysis, laws of electrolysis. His inventions of electric motor, electromagnetic rotar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pseudomorph
In mineralogy, a pseudomorph is a mineral or mineral compound that appears in an atypical form (crystal system), resulting from a substitution process in which the appearance and dimensions remain constant, but the original mineral is replaced by another due to alteration, or chemical substitution, dissolution and refilling, structural changes or incrustation. The name literally means "false form". Terminology for pseudomorphs is "''replacer'' after ''original''", as in ''brookite'' after ''rutile''. The term ''pseudomorphoses'' was initially used by Haüy. Substitution pseudomorph A substitution pseudomorph is a pseudomorph in which one mineral or other material is replaced by another. The original shape of the mineral remains unchanged, but chemical composition, color, hardness, and other properties change to those of the replacing mineral. This happens typically when a mineral of one composition changes by chemical reaction to another of similar composition, retaining t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Argentite
In mineralogy, argentite () is cubic silver sulfide (Ag2S), which can only exist at temperatures above , , or . When it cools to ordinary temperatures it turns into its monoclinic polymorph, acanthite. The International Mineralogical Association has decided to reject argentite as a proper mineral. The name "argentite" sometimes also refers to pseudomorphs of argentite: specimens of acanthite which still display some of the outward signs of the cubic crystal form, even though their actual crystal structure is monoclinic due to the lower temperature. This form of acanthite is occasionally found as uneven cubes and octahedra, but more often as dendritic or earthy masses, with a blackish lead-grey color and metallic luster. Argentite belongs to the galena group. Cleavage, which is so prominent a feature in galena, here presents only in traces. The mineral is perfectly sectile and has a shining streak; hardness 2.5, specific gravity is 7.2–7.4. It occurs in mineral veins, and w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acanthite
Acanthite is a form of silver sulfide with the chemical formula Ag2S. It crystallizes in the monoclinic system and is the stable form of silver sulfide below . Argentite is the stable form above that temperature. As argentite cools below that temperature its cubic form is distorted to the monoclinic form of acanthite. Below 173 °C acanthite forms directly. Acanthite is the only stable form in normal air temperature. Occurrence Acanthite is a common silver mineral in moderately low-temperature hydrothermal veins and in zones of supergene enrichment. It occurs in association with native silver, pyrargyrite, proustite, polybasite, stephanite, aguilarite, galena, chalcopyrite, sphalerite, calcite and quartz. Acanthite was first described in 1855 for an occurrence in the Jáchymov (Joachimsthal) district, Ore Mountains, Bohemia (today Karlovy Vary Region, Czech Republic). The name is from the Greek "akantha" meaning thorn or arrow, in reference to its crystal shape. Gall ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
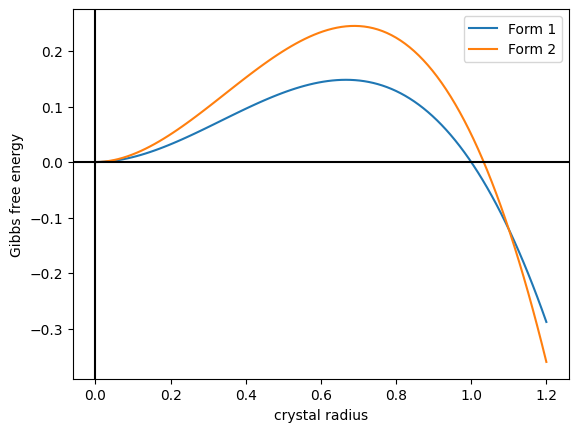
Polymorphism (materials Science)
In crystallography, polymorphism is the phenomenon where a compound or element can crystallize into more than one crystal structure. The preceding definition has evolved over many years and is still under discussion today. Discussion of the defining characteristics of polymorphism involves distinguishing among types of transitions and structural changes occurring in polymorphism versus those in other phenomena. Overview Phase transitions (phase changes) that help describe polymorphism include polymorphic transitions as well as melting and vaporization transitions. According to IUPAC, a polymorphic transition is "A reversible transition of a solid crystalline phase at a certain temperature and pressure (the inversion point) to another phase of the same chemical composition with a different crystal structure." Additionally, Walter McCrone described the phases in polymorphic matter as "different in crystal structure but identical in the liquid or vapor states." McCrone also def ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Paper Mill
A paper mill is a factory devoted to making paper from vegetable fibres such as wood pulp, old rags, and other ingredients. Prior to the invention and adoption of the Fourdrinier machine and other types of paper machine that use an endless belt, all paper in a paper mill was made by hand, one sheet at a time, by specialized laborers. History Historical investigations into the origin of the paper mill are complicated by differing definitions and loose terminology from modern authors: Many modern scholars use the term to refer indiscriminately to all kinds of mills, whether powered by humans, by animals or by water. Their propensity to refer to any ancient paper manufacturing center as a "mill", without further specifying its exact power source, has increased the difficulty of identifying the particularly efficient and historically important water-powered type. Human and animal-powered mills The use of human and animal powered mills was known to Muslim and Chinese paper ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sewage Treatment
Sewage treatment is a type of wastewater treatment which aims to remove contaminants from sewage to produce an effluent that is suitable to discharge to the surrounding environment or an intended reuse application, thereby preventing water pollution from raw sewage discharges. Sewage contains wastewater from households and businesses and possibly pre-treated Industrial wastewater treatment, industrial wastewater. There are a large number of sewage treatment processes to choose from. These can range from Decentralized wastewater system, decentralized systems (including on-site treatment systems) to large centralized systems involving a network of pipes and pump stations (called sewerage) which convey the sewage to a treatment plant. For cities that have a combined sewer, the sewers will also carry urban runoff (stormwater) to the sewage treatment plant. Sewage treatment often involves two main stages, called primary and secondary treatment, while advanced treatment also incorpo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Whisker (metallurgy)
Metal whiskering is a phenomenon that occurs in electrical devices when metals form long whisker-like projections over time. Tin whiskers were noticed and documented in the vacuum tube era of electronics early in the 20th century in equipment that used pure, or almost pure, tin solder in their production. It was noticed that small metal hairs or tendrils grew between metal solder pads, causing short circuits. Metal whiskers form in the presence of compressive stress. Germanium, zinc, cadmium, and even lead whiskers have been documented. Many techniques are used to mitigate the problem, including changes to the annealing (metallurgy), annealing process (heating and cooling), the addition of elements like copper and nickel, and the inclusion of conformal coatings. Traditionally, lead has been added to slow down whisker growth in tin-based solders. Following the RoHS, Restriction of Hazardous Substances Directive (RoHS), the European Union banned the use of lead in most consumer elec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |