|
Sieverts' Law
Sieverts's law, in physical metallurgy and in chemistry, is a rule to predict the solubility of gases in metals. It is named after German chemist Adolf Sieverts (1874–1947). The law states that the solubility of a diatomic gas in metal is proportional to the square root of the partial pressure of the gas in thermodynamic equilibrium.C. K. Gupta, "Chemical metallurgy: principles and practice", Wiley-VCH, 2003, p.273. Hydrogen, oxygen and nitrogen are examples of dissolved diatomic gases of frequent interest in metallurgy. Justification Sieverts's law can be readily rationalized by considering the reaction of dissolution of the gas in the metal, which involves dissociation of the molecule of the gas. For example, for nitrogen: : N2 (molecular gas) 2 N (dissolved atoms) For the above reaction, the equilibrium constant is :K = \frac, where: : ''c''at is the concentration of the dissolved atoms into the metal (in the case above, atomic nitrogen N), : ''p''mol is the partia ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
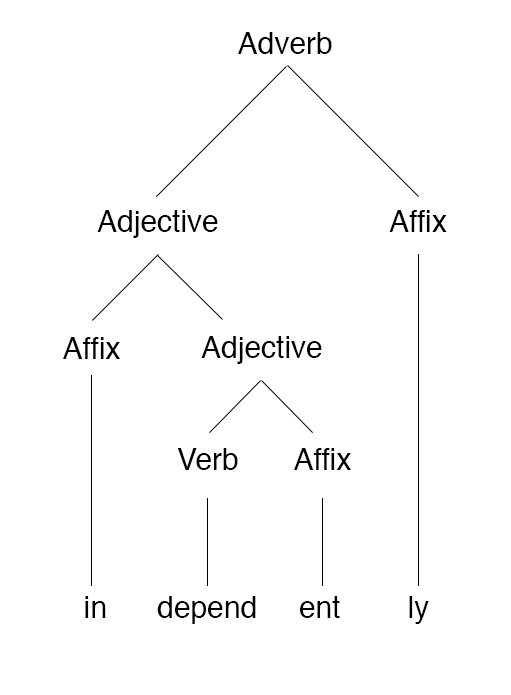
Morphology (linguistics)
In linguistics, morphology is the study of words, including the principles by which they are formed, and how they relate to one another within a language. Most approaches to morphology investigate the structure of words in terms of morphemes, which are the smallest units in a language with some independent meaning. Morphemes include roots that can exist as words by themselves, but also categories such as affixes that can only appear as part of a larger word. For example, in English the root ''catch'' and the suffix ''-ing'' are both morphemes; ''catch'' may appear as its own word, or it may be combined with ''-ing'' to form the new word ''catching''. Morphology also analyzes how words behave as parts of speech, and how they may be inflected to express grammatical categories including number, tense, and aspect. Concepts such as productivity are concerned with how speakers create words in specific contexts, which evolves over the history of a language. The basic fields of ling ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen
Hydrogen is a chemical element; it has chemical symbol, symbol H and atomic number 1. It is the lightest and abundance of the chemical elements, most abundant chemical element in the universe, constituting about 75% of all baryon, normal matter. Under standard conditions, hydrogen is a gas of diatomic molecules with the chemical formula, formula , called dihydrogen, or sometimes hydrogen gas, molecular hydrogen, or simply hydrogen. Dihydrogen is colorless, odorless, non-toxic, and highly combustible. Stars, including the Sun, mainly consist of hydrogen in a plasma state, while on Earth, hydrogen is found as the gas (dihydrogen) and in molecular forms, such as in water and organic compounds. The most common isotope of hydrogen (H) consists of one proton, one electron, and no neutrons. Hydrogen gas was first produced artificially in the 17th century by the reaction of acids with metals. Henry Cavendish, in 1766–1781, identified hydrogen gas as a distinct substance and discovere ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Graham's Law
Graham's law of effusion (also called Graham's law of diffusion) was formulated by Scottish physical chemist Thomas Graham in 1848. Keith J. Laidler and John M. Meiser, ''Physical Chemistry'' (Benjamin/Cummings 1982), pp. 18–19 Graham found experimentally that the rate of effusion of a gas is inversely proportional to the square root of the molar mass of its particles. This formula is stated as: :=\sqrt, where: :Rate1 is the rate of effusion for the first gas. (volume or number of moles per unit time). :Rate2 is the rate of effusion for the second gas. :''M1'' is the molar mass of gas 1 :''M2'' is the molar mass of gas 2. Graham's law states that the rate of diffusion or of effusion of a gas is inversely proportional to the square root of its molecular weight. Thus, if the molecular weight of one gas is four times that of another, it would diffuse through a porous plug or escape through a small pinhole in a vessel at half the rate of the other (heavier gases diffuse mor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Henry's Law
In physical chemistry, Henry's law is a gas law that states that the amount of dissolved gas in a liquid is directly proportional at equilibrium to its partial pressure above the liquid. The proportionality factor is called Henry's law constant. It was formulated by the English chemist William Henry, who studied the topic in the early 19th century. In simple words, it states that the partial pressure of a gas in the vapour phase is directly proportional to the mole fraction of a gas in solution. An example where Henry's law is at play is the depth-dependent dissolution of oxygen and nitrogen in the blood of underwater divers that changes during decompression, going to decompression sickness. An everyday example is carbonated soft drinks, which contain dissolved carbon dioxide. Before opening, the gas above the drink in its container is almost pure carbon dioxide, at a pressure higher than atmospheric pressure. After the bottle is opened, this gas escapes, moving the partial ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Equilibrium Constant
The equilibrium constant of a chemical reaction is the value of its reaction quotient at chemical equilibrium, a state approached by a dynamic chemical system after sufficient time has elapsed at which its composition has no measurable tendency towards further change. For a given set of reaction conditions, the equilibrium constant is independent of the initial analytical concentrations of the reactant and product species in the mixture. Thus, given the initial composition of a system, known equilibrium constant values can be used to determine the Chemical equilibrium#Composition of a mixture, composition of the system at equilibrium. However, reaction parameters like temperature, solvent, and ionic strength may all influence the value of the equilibrium constant. A knowledge of equilibrium constants is essential for the understanding of many chemical systems, as well as the biochemical processes such as oxygen transport by hemoglobin in blood and acid–base homeostasis in the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dissociation (chemistry)
Dissociation in chemistry is a general process in which molecules (or ionic compounds such as salt (chemistry), salts, or coordination complex, complexes) separate or split into other things such as atoms, ions, or radical (chemistry), radicals, usually in a reversible manner. For instance, when an acid dissolves in water, a covalent bond between an electronegativity, electronegative atom and a hydrogen atom is broken by heterolytic fission, which gives a proton (H+) and a negative ion. Dissociation is the opposite of association or recombination. Dissociation constant For reversible dissociations in a chemical equilibrium :AB A + B the dissociation constant ''K''d is the ratio of dissociated to undissociated compound :K_d = \mathrm where the brackets denote the equilibrium concentrations of the species. Dissociation degree The dissociation degree \alpha is the fraction of original solute molecules that have dissociated. It is usually indicated by the Greek symbol α. More acc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrogen
Nitrogen is a chemical element; it has Symbol (chemistry), symbol N and atomic number 7. Nitrogen is a Nonmetal (chemistry), nonmetal and the lightest member of pnictogen, group 15 of the periodic table, often called the Pnictogen, pnictogens. It is a common element in the universe, estimated at Abundance of the chemical elements, seventh in total abundance in the Milky Way and the Solar System. At standard temperature and pressure, two atoms of the element chemical bond, bond to form N2, a colourless and odourless diatomic molecule, diatomic gas. N2 forms about 78% of Atmosphere of Earth, Earth's atmosphere, making it the most abundant chemical species in air. Because of the volatility of nitrogen compounds, nitrogen is relatively rare in the solid parts of the Earth. It was first discovered and isolated by Scottish physician Daniel Rutherford in 1772 and independently by Carl Wilhelm Scheele and Henry Cavendish at about the same time. The name was suggested by French chemist ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxygen
Oxygen is a chemical element; it has chemical symbol, symbol O and atomic number 8. It is a member of the chalcogen group (periodic table), group in the periodic table, a highly reactivity (chemistry), reactive nonmetal (chemistry), nonmetal, and a potent oxidizing agent that readily forms oxides with most elements as well as with other chemical compound, compounds. Oxygen is abundance of elements in Earth's crust, the most abundant element in Earth's crust, making up almost half of the Earth's crust in the form of various oxides such as water, carbon dioxide, iron oxides and silicates.Atkins, P.; Jones, L.; Laverman, L. (2016).''Chemical Principles'', 7th edition. Freeman. It is abundance of chemical elements, the third-most abundant element in the universe after hydrogen and helium. At standard temperature and pressure, two oxygen atoms will chemical bond, bind covalent bond, covalently to form dioxygen, a colorless and odorless diatomic gas with the chemical formula ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thermodynamic Equilibrium
Thermodynamic equilibrium is a notion of thermodynamics with axiomatic status referring to an internal state of a single thermodynamic system, or a relation between several thermodynamic systems connected by more or less permeable or impermeable walls. In thermodynamic equilibrium, there are no net macroscopic flows of mass nor of energy within a system or between systems. In a system that is in its own state of internal thermodynamic equilibrium, not only is there an absence of macroscopic change, but there is an “absence of any ''tendency'' toward change on a macroscopic scale.” Systems in mutual thermodynamic equilibrium are simultaneously in mutual thermal, mechanical, chemical, and radiative equilibria. Systems can be in one kind of mutual equilibrium, while not in others. In thermodynamic equilibrium, all kinds of equilibrium hold at once and indefinitely, unless disturbed by a thermodynamic operation. In a macroscopic equilibrium, perfectly or almost perfectly ba ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Linguistics
Linguistics is the scientific study of language. The areas of linguistic analysis are syntax (rules governing the structure of sentences), semantics (meaning), Morphology (linguistics), morphology (structure of words), phonetics (speech sounds and equivalent gestures in sign languages), phonology (the abstract sound system of a particular language, and analogous systems of sign languages), and pragmatics (how the context of use contributes to meaning). Subdisciplines such as biolinguistics (the study of the biological variables and evolution of language) and psycholinguistics (the study of psychological factors in human language) bridge many of these divisions. Linguistics encompasses Outline of linguistics, many branches and subfields that span both theoretical and practical applications. Theoretical linguistics is concerned with understanding the universal grammar, universal and Philosophy of language#Nature of language, fundamental nature of language and developing a general ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Partial Pressure
In a mixture of gases, each constituent gas has a partial pressure which is the notional pressure of that constituent gas as if it alone occupied the entire volume of the original mixture at the same temperature. The total pressure of an ideal gas mixture is the sum of the partial pressures of the gases in the mixture (Dalton's Law). In respiratory physiology, the partial pressure of a dissolved gas in liquid (such as oxygen in arterial blood) is also defined as the partial pressure of that gas as it would be undissolved in gas phase yet in equilibrium with the liquid. This concept is also known as blood gas tension. In this sense, the diffusion of a gas liquid is said to be driven by differences in partial pressure (not concentration). In chemistry and thermodynamics, this concept is generalized to non-ideal gases and instead called fugacity. The partial pressure of a gas is a measure of its Thermodynamic activity, thermodynamic activity. Gases dissolve, diffuse, and react accor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diatomic Molecule
Diatomic molecules () are molecules composed of only two atoms, of the same or different chemical elements. If a diatomic molecule consists of two atoms of the same element, such as hydrogen () or oxygen (), then it is said to be homonuclear molecule, homonuclear. Otherwise, if a diatomic molecule consists of two different atoms, such as carbon monoxide () or nitric oxide (), the molecule is said to be heteronuclear molecule, heteronuclear. The bond in a homonuclear diatomic molecule is non-polar. The only chemical elements that form stable homonuclear diatomic molecules at standard temperature and pressure (STP) (or at typical laboratory conditions of 1 bar (pressure), bar and 25 °C) are the gases hydrogen (), nitrogen (), oxygen (), fluorine (), and chlorine (), and the liquid bromine (). The noble gases (helium, neon, argon, krypton, xenon, and radon) are also gases at STP, but they are monatomic. The homonuclear diatomic gases and noble gases together are called "ele ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |