|
Selenocysteine
Selenocysteine (symbol Sec or U, in older publications also as Se-Cys) is the 21st proteinogenic amino acid. Selenoproteins contain selenocysteine residues. Selenocysteine is an analogue of the more common cysteine with selenium in place of the sulfur. Selenocysteine is present in several enzymes (for example glutathione peroxidases, tetraiodothyronine 5 deiodinase, tetraiodothyronine 5′ deiodinases, thioredoxin reductases, formate dehydrogenases, glycine reductases, selenophosphate synthetase 2, methionine-''R''-sulfoxide reductase B1 (SEPX1), and some hydrogenases). It occurs in all three Domain (biology), domains of life, including important enzymes (listed above) present in humans. Selenocysteine was discovered in 1974 by biochemist Thressa Stadtman at the National Institutes of Health. Chemistry Selenocysteine is the Se-analogue of cysteine. It is rarely encountered outside of living tissue (nor is it available commercially) because of its high susceptiblility to air-oxi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Selenoprotein
In molecular biology a selenoprotein is any protein that includes a selenocysteine (Sec, U, Se-Cys) amino acid residue. Among functionally characterized selenoproteins are five glutathione peroxidases (GPX) and three thioredoxin reductases, (TrxR/TXNRD) which both contain only one Sec. Selenoprotein P is the most common selenoprotein found in the plasma. It is unusual because in humans it contains 10 Sec residues, which are split into two domains, a longer N-terminal domain that contains 1 Sec, and a shorter C-terminal domain that contains 9 Sec. The longer N-terminal domain is likely an enzymatic domain, and the shorter C-terminal domain is likely a means of safely transporting the very reactive selenium atom throughout the body. Species distribution Selenoproteins exist in all major domains of life, eukaryotes, bacteria and archaea. Among eukaryotes, selenoproteins appear to be common in animals, but rare or absent in other phyla—one has been identified in the green alga '' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Proteinogenic Amino Acid
Proteinogenic amino acids are amino acids that are incorporated biosynthetically into proteins during translation from RNA. The word "proteinogenic" means "protein creating". Throughout known life, there are 22 genetically encoded (proteinogenic) amino acids, 20 in the standard genetic code and an additional 2 ( selenocysteine and pyrrolysine) that can be incorporated by special translation mechanisms. In contrast, non-proteinogenic amino acids are amino acids that are either not incorporated into proteins (like GABA, L-DOPA, or triiodothyronine), misincorporated in place of a genetically encoded amino acid, or not produced directly and in isolation by standard cellular machinery (like hydroxyproline). The latter often results from post-translational modification of proteins. Some non-proteinogenic amino acids are incorporated into nonribosomal peptides which are synthesized by non-ribosomal peptide synthetases. Both eukaryotes and prokaryotes can incorporate selenocysteine ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Selenium
Selenium is a chemical element; it has symbol (chemistry), symbol Se and atomic number 34. It has various physical appearances, including a brick-red powder, a vitreous black solid, and a grey metallic-looking form. It seldom occurs in this elemental state or as pure ore compounds in Earth's crust. Selenium ( ) was discovered in 1817 by , who noted the similarity of the new element to the previously discovered tellurium (named for the Earth). Selenium is found in :Sulfide minerals, metal sulfide ores, where it substitutes for sulfur. Commercially, selenium is produced as a byproduct in the refining of these ores. Minerals that are pure selenide or selenate compounds are rare. The chief commercial uses for selenium today are glassmaking and pigments. Selenium is a semiconductor and is used in photocells. Applications in electronics, once important, have been mostly replaced with silicon semiconductor devices. Selenium is still used in a few types of Direct current, DC power surge ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
SEPX1
Methionine-R-sulfoxide reductase B1 is an enzyme that in humans is encoded by the ''SEPX1'' gene In biology, the word gene has two meanings. The Mendelian gene is a basic unit of heredity. The molecular gene is a sequence of nucleotides in DNA that is transcribed to produce a functional RNA. There are two types of molecular genes: protei .... This gene encodes a selenoprotein, which contains a selenocysteine (Sec) residue at its active site. The selenocysteine is encoded by the UGA codon that normally signals translation termination. The 3' UTR of selenoprotein genes have a common stem-loop structure, the sec insertion sequence (SECIS), that is necessary for the recognition of UGA as a Sec codon rather than as a stop signal. This protein belongs to the methionine sulfoxide reductase B (MsrB) family, and it is expressed in a variety of adult and fetal tissues. See also * MSRA (gene) * MSRB2 * Methionine oxidation References Further reading * * * * * * * * Selenoprot ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
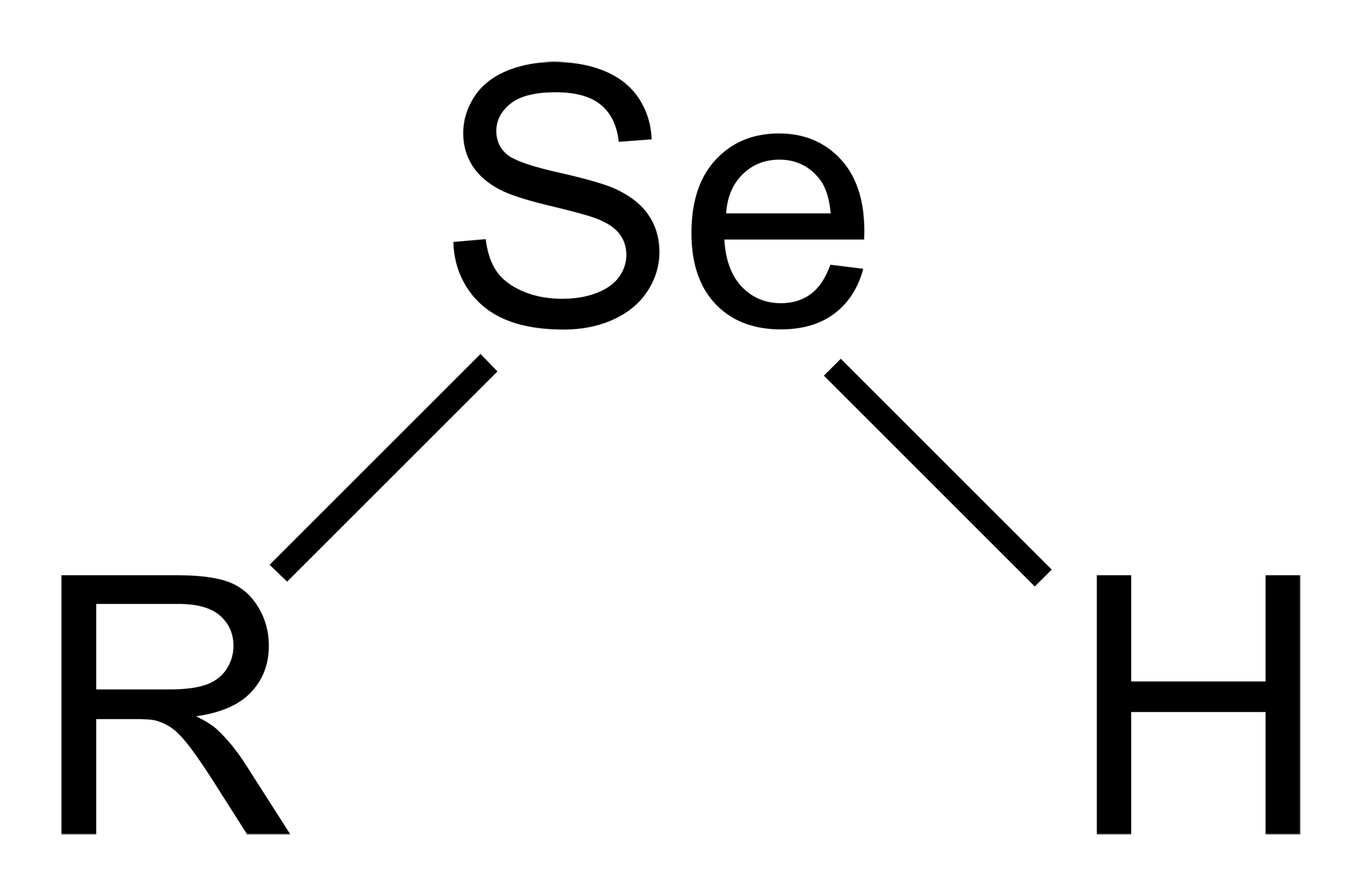
Selenol
Selenols are organic compounds that contain the functional group with the connectivity . Selenols are sometimes also called selenomercaptans and selenothiols. Selenols are one of the principal classes of organoselenium compounds. A well-known selenol is the amino acid selenocysteine. Structure and properties Selenols are structurally similar to thiols, but the bond is about 8% longer at 196 pm. The angle approaches 90°. The bonding involves almost pure p-orbitals on Se, hence the near 90 angles. The bond energy is weaker than the bond, consequently selenols are easily oxidized and serve as H-atom donors. The Se-H bond is weaker than the bond as reflected in their respective bond dissociation energy (BDE). For , the BDE is 326 kJ/mol, while for , the BDE is 368 kJ/mol. Selenols are about 1000 times stronger acids than thiols: the p''K''a of is 5.2 vs 8.3 for . Deprotonation affords the selenolate anion, , most examples of which are highly nucleophilic and rapidly o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thioredoxin Reductase
Thioredoxin reductases (TR, TrxR) () are enzymes that reduce thioredoxin (Trx). Two classes of thioredoxin reductase have been identified: one class in bacteria and some eukaryotes and one in animals. Bacterial TrxR also catalyzes the reduction of glutaredoxin like proteins known as NrdH. Both classes are flavoproteins which function as homodimers. Each monomer contains a flavin adenine dinucleotide, FAD prosthetic group, a NADPH binding domain, and an active site containing a redox-active disulfide bond. Cellular role Thioredoxin reductases are enzymes that catalyze the reduction of thioredoxin and hence they are a central component in the thioredoxin system. Together with thioredoxin (Trx) and NADPH this system's most general description is as a system for reducing disulfide bonds in cells. Electrons are taken from NADPH via TrxR and are transferred to the active site of Trx, which goes on to reduce protein disulfides or other substrates. The Trx system exists in all living ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
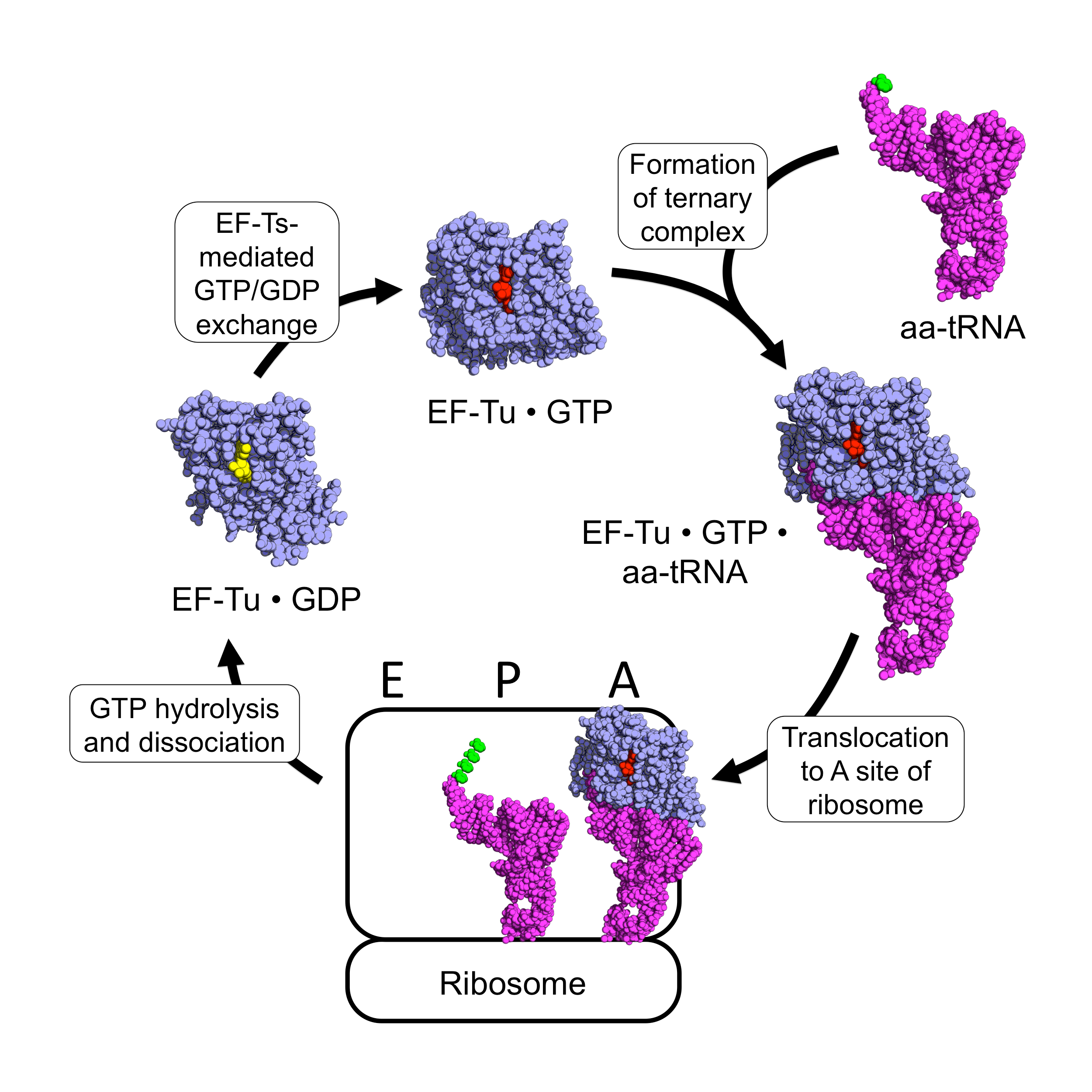
EF-Tu
EF-Tu (elongation factor thermo unstable) is a prokaryotic elongation factor responsible for catalyzing the binding of an aminoacyl-tRNA (aa-tRNA) to the ribosome. It is a G-protein, and facilitates the selection and binding of an aa-tRNA to the A-site of the ribosome. As a reflection of its crucial role in translation, EF-Tu is one of the most abundant and highly conserved proteins in prokaryotes. It is found in eukaryotic mitochondria as TUFM. As a family of elongation factors, EF-Tu also includes its eukaryotic and archaeal homolog, the alpha subunit of eEF-1 (EF-1A). Background Elongation factors are part of the mechanism that synthesizes new proteins through translation in the ribosome. Transfer RNAs (tRNAs) carry the individual amino acids that become integrated into a protein sequence, and have an anticodon for the specific amino acid that they are charged with. Messenger RNA (mRNA) carries the genetic information that encodes the primary structure of a protein, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cysteine
Cysteine (; symbol Cys or C) is a semiessential proteinogenic amino acid with the chemical formula, formula . The thiol side chain in cysteine enables the formation of Disulfide, disulfide bonds, and often participates in enzymatic reactions as a nucleophile. Cysteine is chiral, but both D and L-cysteine are found in nature. LCysteine is a protein monomer in all biota, and D-cysteine acts as a signaling molecule in mammalian nervous systems. Cysteine is named after its discovery in urine, which comes from the urinary bladder or cyst, from Ancient Greek, Greek κύστις ''kýstis'', "bladder". The thiol is susceptible to oxidation to give the disulfide bond, disulfide derivative cystine, which serves an important structural role in many proteins. In this case, the symbol Cyx is sometimes used. The deprotonated form can generally be described by the symbol Cym as well. When used as a food additive, cysteine has the E number E920. Cysteine is Genetic code, encoded by the codo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thressa Stadtman
Thressa Campbell Stadtman (February 12, 1920 – December 11, 2016) was an American biochemist, notable for her discovery of selenocysteine, and her research on selenoproteins and bioenergetics. In addition she made significant advances in amino acid metabolism, enzymes dependent on vitamin B12, and the biochemistry of microbes. Life In 1920, she was born in Sterling, New York. In 1940, she graduated from Cornell University, with a B.S. in Microbiology, and in 1942, with a M.S. in Microbiology and Nutrition. In 1949, she graduated from University of California, University of California, Berkeley, Berkeley, with a Ph.D. in Microbial Biochemistry. Her thesis was titled "Studies on Methane Fermentations", and subsequently worked as a postdoc for Christian B. Anfinsen at Harvard Medical School. She was married to Earl Reece Stadtman whom she met when they were both graduate students at the University of California, Berkeley. They were both hired by what was then the National Heart, L ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Selenocystine
Selenocystine is the amino acid with the formula . It is the oxidized derivative of the canonical amino acid selenocysteine (). The compound can also be prepared synthetically from serine. Because selenocysteine is not easily isolated or handled, it is often generated by reduction of selenocystine in situ. The selenium–selenium bond length is 2.321 Å, which is 14% longer than the disulfide bond in cystine Cystine is the oxidized derivative of the amino acid cysteine and has the formula (SCH2CH(NH2)CO2H)2. It is a white solid that is poorly soluble in water. As a residue in proteins, cystine serves two functions: a site of redox reactions and a mec ... at 2.040 Å. References {{Amino acids Alpha-Amino acids Proteinogenic amino acids Organoselenium compounds ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogenase
A hydrogenase is an enzyme that Catalysis, catalyses the reversible Redox, oxidation of molecular hydrogen (H2), as shown below: Hydrogen oxidation () is coupled to the reduction of electron acceptors such as oxygen, nitrate, Ferric, ferric ion, sulfate, carbon dioxide (), and fumarate. On the other hand, proton reduction () is coupled to the oxidation of electron donors such as ferredoxin (FNR), and serves to dispose excess electrons in cells (essential in pyruvate fermentation). Both low-molecular weight compounds and proteins such as FNRs, cytochrome ''c''3, and cytochrome ''c''6 can act as physiological electron donors or acceptors for hydrogenases. Structural classification It has been estimated that 99% of all organisms utilize hydrogen, H2. Most of these species are microbes and their ability to use H2 as a metabolite arises from the expression of metalloenzymes known as hydrogenases. Hydrogenases are sub-classified into three different types based on the active site ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |