|
Rhodococcus Gannanensis
''Rhodococcus'' is a genus of aerobic, nonsporulating, nonmotile Gram-positive bacteria closely related to ''Mycobacterium'' and ''Corynebacterium''. While a few species are pathogenic, most are benign, and have been found to thrive in a broad range of environments, including soil, water, and eukaryotic cells. Some species have large genomes, including the 9.7 megabasepair genome (67% G/C) of ''Rhodococcus'' sp. RHA1. Strains of ''Rhodococcus'' are important owing to their ability to catabolize a wide range of compounds and produce bioactive steroids, acrylamide, and acrylic acid, and their involvement in fossil fuel biodesulfurization. This genetic and catabolic diversity is not only due to the large bacterial chromosome, but also to the presence of three large linear plasmids. ''Rhodococcus'' is also an experimentally advantageous system owing to a relatively fast growth rate and simple developmental cycle, but is not well characterized. Another important application of ''Rh ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Friedrich Wilhelm Zopf
Friedrich (or Friederich) Wilhelm Zopf (12 December 1846 – 24 June 1909) was a well-known German botanist and mycologist. He dedicated to his whole life with fungal biology, particularly in classification of fungi and dye production in fungi and lichens. Besides, his textbook on fungi called “Die pilze in morphologischer, physiologischer, biologischer und systematischer beziehung (Translation: The mushrooms in morphological, physiological, biological and systematic relationship)” in 1890 was also an outstanding work on the subject for many decades. The unicellular achlorophic microalgae ''Prototheca zopfii'' is named after him because of his profound suggestions and contributions to Krüger's pioneering work in ''Prototheca''. Thus, his numerous contributions gave him a special status in mycological history. Early life Wilhelm Zopf was born in Roßleben in Thuringia in 1846. Before going into biological science area, he has been an elementary school teacher in Thondorf at ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cellular Respiration
Cellular respiration is the process of oxidizing biological fuels using an inorganic electron acceptor, such as oxygen, to drive production of adenosine triphosphate (ATP), which stores chemical energy in a biologically accessible form. Cellular respiration may be described as a set of metabolic reactions and processes that take place in the cells of organisms to transfer chemical energy from nutrients to ATP, with the flow of electrons to an electron acceptor, and then release waste products. If the electron acceptor is oxygen, the process is more specifically known as aerobic cellular respiration. If the electron acceptor is a molecule other than oxygen, this is anaerobic cellular respiration. Fermentation, which is also an anaerobic process, is not respiration, as no external electron acceptor is involved. The reactions involved in respiration are catabolic reactions, which break large molecules into smaller ones, producing large amounts of energy (ATP). Respiration ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
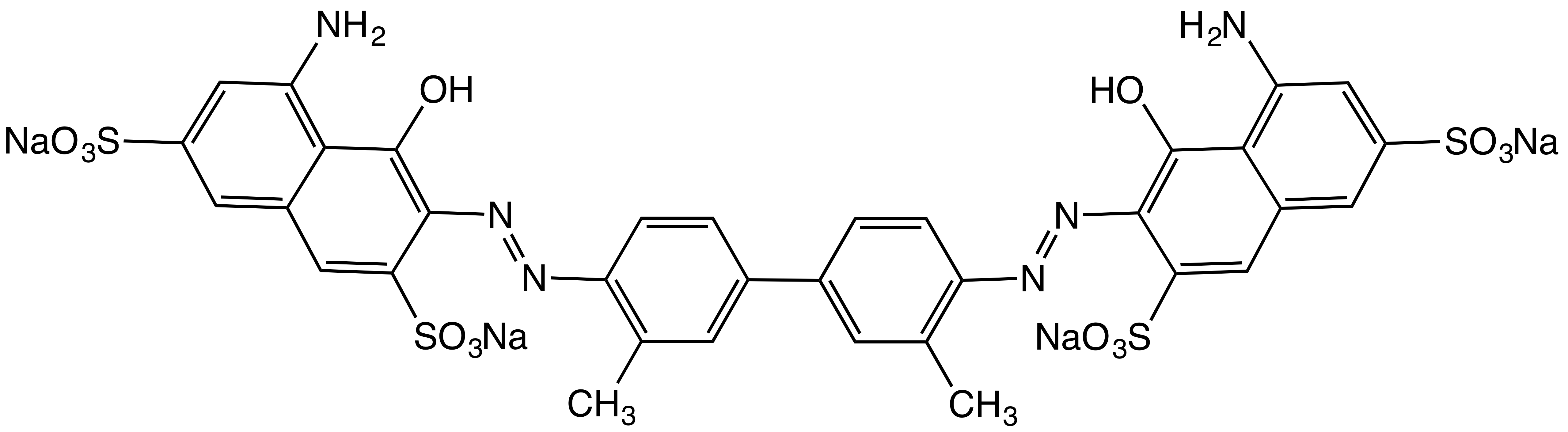
Azo Dye
Azo dyes are organic compounds bearing the functional group R−N=N−R′, in which R and R′ are usually aryl and substituted aryl groups. They are a commercially important family of azo compounds, i.e. compounds containing the C−N=N−C linkage. Azo dyes are synthetic dyes and do not occur naturally. Most azo dyes contain only one azo group but there are some that contain two or three azo groups, called "diazo dyes" and "triazo dyes" respectively. Azo dyes comprise 60–70% of all dyes used in food and textile industries. Azo dyes are widely used to treat textiles, leather articles, and some foods. Chemically related derivatives of azo dyes include azo pigments, which are insoluble in water and other solvents. Classes Many kinds of azo dyes are known, and several classification systems exist. Some classes include disperse dyes, metal-complex dyes, reactive dyes, and substantive dyes. Also called direct dyes, substantive dyes are employed for cellulose-based textil ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Caesium
Caesium (IUPAC spelling; also spelled cesium in American English) is a chemical element; it has Symbol (chemistry), symbol Cs and atomic number 55. It is a soft, silvery-golden alkali metal with a melting point of , which makes it one of only five elemental metals that are liquid at or near room temperature. Caesium has physical and chemical properties similar to those of rubidium and potassium. It is pyrophoricity, pyrophoric and reacts with water even at . It is the least electronegativity, electronegative stable element, with a value of 0.79 on the Pauling scale. It has only one stable isotope, caesium-133. Caesium is mined mostly from pollucite. Caesium-137, a fission product, is extracted from waste produced by nuclear reactor technology, nuclear reactors. It has the largest atomic radius of all elements whose radii have been measured or calculated, at about 260 picometres. The German chemist Robert Bunsen and physicist Gustav Kirchhoff discovered caesium in 1860 by the new ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heavy Metals
upright=1.2, Crystals of lead.html" ;"title="osmium, a heavy metal nearly twice as dense as lead">osmium, a heavy metal nearly twice as dense as lead Heavy metals is a controversial and ambiguous term for metallic elements with relatively high density, densities, atomic weights, or atomic numbers. The criteria used, and whether metalloids are included, vary depending on the author and context and it has been argued that the term "heavy metal" should be avoided. A heavy metal may be defined on the basis of density, atomic number or chemical behaviour. More specific definitions have been published, none of which have been widely accepted. The definitions surveyed in this article encompass up to 96 out of the 118 known chemical elements; only mercury, lead and bismuth meet all of them. Despite this lack of agreement, the term (plural or singular) is widely used in science. A density of more than 5 g/cm3 is sometimes quoted as a commonly used criterion and is used in the b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protocatechuic Acid
Protocatechuic acid (PCA) is a dihydroxybenzoic acid, a type of phenolic acid. It is a major metabolite of antioxidant polyphenols found in green tea. It has mixed effects on normal and cancer cells in ''in vitro'' and ''in vivo'' studies. It is produced commercially from vanillin. Biological effects Protocatechuic acid (PCA) is antioxidant and anti-inflammatory. PCA extracted from ''Hibiscus sabdariffa'' protected against chemically induced liver toxicity ''in vivo''. ''In vitro'' testing documented antioxidant and anti-inflammatory activity of PCA, while liver protection ''in vivo'' was measured by chemical markers and histological assessment. PCA has been reported to induce apoptosis of human leukemia cells, as well as malignant HSG1 cells taken from human oral cavities, but PCA was found to have mixed effects on TPA-induced mouse skin tumours. Depending on the amount of PCA and the time before application, PCA could reduce or enhance tumour growth. Similarly, PCA was repo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzoate
Benzoic acid () is a white (or colorless) solid organic compound with the formula , whose structure consists of a benzene ring () with a carboxyl () substituent. The benzoyl group is often abbreviated "Bz" (not to be confused with "Bn," which is used for benzyl), thus benzoic acid is also denoted as BzOH, since the benzoyl group has the formula –. It is the simplest aromatic carboxylic acid. The name is derived from gum benzoin, which was for a long time its only source. Benzoic acid occurs naturally in many plants and serves as an intermediate in the biosynthesis of many secondary metabolites. Salts of benzoic acid are used as food preservatives. Benzoic acid is an important precursor for the industrial synthesis of many other organic substances. The salts and esters of benzoic acid are known as benzoates (). History Benzoic acid was discovered in the sixteenth century. The dry distillation of gum benzoin was first described by Nostradamus (1556), and then by Ale ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Catechol
Catechol ( or ), also known as pyrocatechol or 1,2-dihydroxybenzene, is an organic compound with the molecular formula . It is the ''ortho'' isomer of the three isomeric benzenediols. This colorless compound occurs naturally in trace amounts. It was first discovered by destructive distillation of the plant extract catechin. About 20,000 tonnes of catechol are now synthetically produced annually as a commodity organic chemical, mainly as a precursor to pesticides, flavors, and fragrances. Small amounts of catechol occur in fruits and vegetables. Isolation and synthesis Catechol was first isolated in 1839 by Edgar Hugo Emil Reinsch (1809–1884) by distilling it from the solid tannic preparation catechin, which is the residuum of catechu, the boiled or concentrated juice of ''Mimosa catechu'' ('' Acacia catechu''). Upon heating catechin above its decomposition point, a substance that Reinsch first named ''Brenz-Katechusäure'' (burned catechu acid) sublimated as a white efflo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyridine
Pyridine is a basic (chemistry), basic heterocyclic compound, heterocyclic organic compound with the chemical formula . It is structurally related to benzene, with one methine group replaced by a nitrogen atom . It is a highly flammable, weakly alkaline, water-miscible liquid with a distinctive, unpleasant fish-like smell. Pyridine is colorless, but older or impure samples can appear yellow, due to the formation of extended, unsaturated Polymer, polymeric chains, which show significant electrical conductivity. The pyridine ring occurs in many important compounds, including agrochemicals, pharmaceuticals, and vitamins. Historically, pyridine was produced from coal tar. As of 2016, it is synthesized on the scale of about 20,000 tons per year worldwide. Properties Physical properties Pyridine is diamagnetism, diamagnetic. Its critical point (thermodynamics), critical parameters are: pressure 5.63 MPa, temperature 619 K and volume 248 cm3/mol. In the temperatur ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Quinoline
Quinoline is a heterocyclic aromatic organic compound with the chemical formula C9H7N. It is a colorless hygroscopic liquid with a strong odor. Aged samples, especially if exposed to light, become yellow and later brown. Quinoline is only slightly soluble in cold water but dissolves readily in hot water and most organic solvents. Quinoline itself has few applications, but many of its derivatives are useful in diverse applications. A prominent example is quinine, an alkaloid found in plants. Over 200 biologically active quinoline and quinazoline alkaloids are identified. 4-Hydroxy-2-alkylquinolines (HAQs) are involved in antibiotic resistance. Occurrence and isolation Quinoline was first extracted from coal tar in 1834 by German chemist Friedlieb Ferdinand Runge; he called quinoline ''leukol'' ("white oil" in Greek). Coal tar remains the principal source of commercial quinoline. In 1842, French chemist Charles Gerhardt obtained a compound by dry distilling quinine, st ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzotrifluoride
Trifluorotoluene is an organic compound with the formula of C6H5CF3. This colorless fluorocarbon is used as a specialty solvent in organic synthesis and an intermediate in the production of pesticides and pharmaceuticals.Banks, R.E. Organofluorine Chemicals and their Industrial Applications, Ellis Horwood LTD, Chichester, 1979. Synthesis For small-scale laboratory preparations, trifluorotoluene is synthesized by coupling an aromatic halide and trifluoromethyl iodide in the presence of a copper catalyst: :PhX + CF3I → PhCF3 (where X = I, Br) Industrial production is done by reacting benzotrichloride with hydrogen fluoride in a pressurized reactor.Siegemund, Günter "Aromatic Compounds with Fluorinated Side-Chains" in Ullmann’s Encyclopedia of Industrial Chemistry 2005, Wiley-VCH. . :PhCCl3 + 3 HF → PhCF3 + 3 HCl Uses Trifluorotoluene has a variety of niche uses. Low toxicity alternative to dichloromethane According to Ogawa and Curran, trifluorotoluene is similar to di ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dioxygenases
Dioxygenases are oxidoreductase enzymes. Aerobic life, from simple single-celled bacteria species to complex eukaryotic organisms, has evolved to depend on the oxidizing power of dioxygen in various metabolic pathways. From energetic adenosine triphosphate (ATP) generation to xenobiotic degradation, the use of dioxygen as a biological oxidant is widespread and varied in the exact mechanism of its use. Enzymes employ many different schemes to use dioxygen, and this largely depends on the substrate and reaction at hand. Comparison with monooxygenases In the monooxygenases, only a single atom of dioxygen is incorporated into a substrate with the other being reduced to a water molecule. The dioxygenases () catalyze the oxidation of a substrate without the reduction of one oxygen atom from dioxygen into a water molecule. However, this definition is ambiguous because it does not take into account how many substrates are involved in the reaction. The majority of dioxygenases fully in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |