|
Palmitoyl-CoA
Palmitoyl-CoA is an acyl-CoA thioester. It is an "activated" form of palmitic acid and can be transported into the mitochondrial matrix by the carnitine shuttle system (which transports fatty acyl-CoA molecules into the mitochondria), and once inside can participate in beta-oxidation. Alternatively, palmitoyl-CoA is used as a substrate in the biosynthesis of sphingosine (this biosynthetic pathway does not require transfer into the mitochondria). Biosynthesis Palmitoyl CoA formed from palmitic acid, in the reaction below. Palmitate + CoA-SH + ATP -> Palmitoyl-CoA + AMP + Pyrophosphate This reaction is often referred to as the "activation" of a fatty acid. The activation is catalyzed by palmitoyl-coenzyme A synthetase and the reaction proceeds through a two step mechanism, in which palmitoyl-AMP is an intermediate. The reaction is driven to completion by the exergonic hydrolysis of pyrophosphate. The activation of fatty acids occurs in the cytosol and beta-oxidation occurs i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acyl-CoA
Acyl-CoA is a group of coenzymes that metabolize fatty acids. Acyl-CoA's are susceptible to beta oxidation, forming, ultimately, acetyl-CoA. The acetyl-CoA enters the citric acid cycle, eventually forming several equivalents of ATP. In this way, fats are converted to ATP, the universal biochemical energy carrier. Functions Fatty acid activation Fats are broken down by conversion to acyl-CoA. This conversion is one response to high energy demands such as exercise. The oxidative degradation of fatty acids is a two-step process, catalyzed by acyl-CoA synthetase. Fatty acids are converted to their acyl phosphate, the precursor to acyl-CoA. The latter conversion is mediated by acyl-CoA synthase" :acyl-P + HS-CoA → acyl-S-CoA + Pi + H+ Three types of acyl-CoA synthases are employed, depending on the chain length of the fatty acid. For example, the substrates for medium chain acyl-CoA synthase are 4-11 carbon fatty acids. The enzyme acyl-CoA thioesterase takes of the acyl-CoA t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
NADH
Nicotinamide adenine dinucleotide (NAD) is a coenzyme central to metabolism. Found in all living cells, NAD is called a dinucleotide because it consists of two nucleotides joined through their phosphate groups. One nucleotide contains an adenine nucleobase and the other nicotinamide. NAD exists in two forms: an oxidized and reduced form, abbreviated as NAD and NADH (H for hydrogen), respectively. In metabolism, nicotinamide adenine dinucleotide is involved in redox reactions, carrying electrons from one reaction to another. The cofactor is, therefore, found in two forms in cells: NAD is an oxidizing agent – it accepts electrons from other molecules and becomes reduced. This reaction, also with H+, forms NADH, which can then be used as a reducing agent to donate electrons. These electron transfer reactions are the main function of NAD. However, it is also used in other cellular processes, most notably as a substrate of enzymes in adding or removing chemical groups t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thioesters Of Coenzyme A
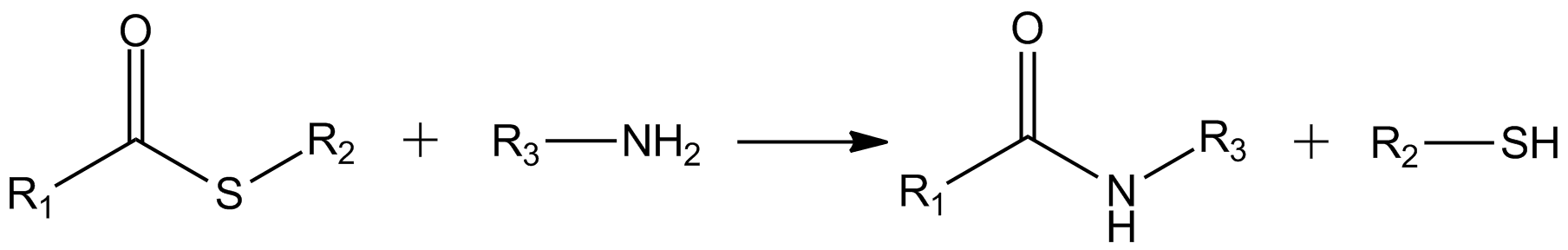
In organic chemistry, thioesters are organosulfur compounds with the functional group . They are analogous to carboxylate esters () with the sulfur in the thioester playing the role of the linking oxygen in the carboxylate ester, as implied by the ''thio-'' prefix. They are the product of esterification between a carboxylic acid () and a thiol (). In biochemistry, the best-known thioesters are derivatives of coenzyme A, e.g., acetyl-CoA.Matthys J. Janssen "Carboxylic Acids and Esters" in PATAI's Chemistry of Functional Groups: Carboxylic Acids and Esters, Saul Patai, Ed. John Wiley, 1969, New York: pp. 705–764. Synthesis The most typical route to thioester involves the reaction of an acid chloride with an alkali metal salt of a thiol: :RSNa + R'COCl -> R'COSR + NaCl Another common route entails the displacement of halides by the alkali metal salt of a thiocarboxylic acid. For example, thioacetate esters are commonly prepared by alkylation of potassium thioacetate: :CH3C ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coenzyme A
Coenzyme A (CoA, SHCoA, CoASH) is a coenzyme, notable for its role in the synthesis and oxidation of fatty acids, and the oxidation of pyruvate in the citric acid cycle. All genomes sequenced to date encode enzymes that use coenzyme A as a substrate, and around 4% of cellular enzymes use it (or a thioester) as a substrate. In humans, CoA biosynthesis requires cysteine, pantothenic acid, pantothenate (vitamin B5), and adenosine triphosphate (ATP). In acetyl-CoA, its acetyl form, coenzyme A is a highly versatile molecule, serving metabolic functions in both the Anabolism, anabolic and Catabolism, catabolic pathways. Acetyl-CoA is utilised in the post-translational regulation and allosteric regulation of pyruvate dehydrogenase and carboxylase to maintain and support the partition of Pyruvic acid, pyruvate synthesis and degradation. Discovery of structure Coenzyme A was identified by Fritz Lipmann in 1946, who also later gave it its name. Its structure was determined during the e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Palmitic Acid
Palmitic acid (hexadecanoic acid in IUPAC nomenclature) is a fatty acid with a 16-carbon chain. It is the most common saturated fatty acid found in animals, plants and microorganisms.Gunstone, F. D., John L. Harwood, and Albert J. Dijkstra. The Lipid Handbook, 3rd ed. Boca Raton: CRC Press, 2007. , Its chemical formula is CH3(CH2)14COOH, and its C:D (the total number of carbon atoms to the number of carbon-carbon double-bonds) is 16:0. It is a major component of the oil from the fruit of oil palms ( palm oil), making up to 44% of total fats. Meats, cheeses, butter, and other dairy products also contain palmitic acid, amounting to 50–60% of total fats. Palmitates are the salts and esters of palmitic acid. The palmitate anion is the observed form of palmitic acid at physiologic pH (7.4). Occurrence and production Palmitic acid was discovered by Edmond Frémy in 1840, in saponified palm oil. This remains the primary industrial route for its production, with the triglycerides (fa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sphingosine Synthesis Corrected
Sphingosine (2-amino-4-trans-octadecene-1,3-diol) is an 18-carbon amino alcohol with an unsaturated hydrocarbon chain, which forms a primary part of sphingolipids, a class of cell membrane lipids that include sphingomyelin, an important phospholipid. Functions Sphingosine can be phosphorylated in vivo via two kinases, sphingosine kinase type 1 and sphingosine kinase type 2. This leads to the formation of sphingosine-1-phosphate, a potent signaling lipid. Sphingolipid metabolites, such as ceramides, sphingosine and sphingosine-1-phosphate, are lipid signaling molecules involved in diverse cellular processes. Biosynthesis Sphingosine is synthesized from palmitoyl CoA and serine in a condensation required to yield dihydrosphingosine. Dehydrosphingosine is then reduced by NADPH to dihydrosphingosine (sphinganine), acylated to dihydroceramide finally oxidized by FAD to ceramide. Sphingosine is then solely formed via degradation of sphingolipid in the lysosome. See also ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Serine C-palmitoyltransferase
In enzymology, a serine C-palmitoyltransferase () is an enzyme that catalyzes the chemical reaction: :palmitoyl-CoA + L-serine \rightleftharpoons CoA + 3-dehydro-D-sphinganine + CO2 Thus, the two substrates of this enzyme are palmitoyl-CoA and L-serine, whereas its 3 products are CoA, 3-dehydro-D-sphinganine, and CO2. This reaction is a key step in the biosynthesis of sphingosine which is a precursor of many other sphingolipids. This enzyme participates in sphingolipid metabolism. It employs one cofactor, pyridoxal phosphate. Nomenclature This enzyme belongs to the family of transferases, specifically those acyltransferases transferring groups other than aminoacyl groups. The systematic name of this enzyme class is palmitoyl-CoA:L-serine C-palmitoyltransferase (decarboxylating). Other names in common use include: * serine palmitoyltransferase, * SPT, 3-oxosphinganine synthetase, and * acyl-CoA:serine C-2 acyltransferase decarboxylating. Structure Serine C-pa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Beta Oxidation
In biochemistry and metabolism, beta-oxidation is the catabolic process by which fatty acid molecules are broken down in the cytosol in prokaryotes and in the mitochondria in eukaryotes to generate acetyl-CoA, which enters the citric acid cycle, and NADH and FADH2, which are co-enzymes used in the electron transport chain. It is named as such because the beta carbon of the fatty acid undergoes oxidation to a carbonyl group. Beta-oxidation is primarily facilitated by the mitochondrial trifunctional protein, an enzyme complex associated with the inner mitochondrial membrane, although very long chain fatty acids are oxidized in peroxisomes. The overall reaction for one cycle of beta oxidation is: :C''n''-acyl-CoA + FAD + + + CoA → C''n''-2-acyl-CoA + + NADH + + acetyl-CoA Activation and membrane transport Free fatty acids cannot penetrate any biological membrane due to their negative charge. Free fatty acids must cross the cell membrane through specific transport pro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Palmitic Acid
Palmitic acid (hexadecanoic acid in IUPAC nomenclature) is a fatty acid with a 16-carbon chain. It is the most common saturated fatty acid found in animals, plants and microorganisms.Gunstone, F. D., John L. Harwood, and Albert J. Dijkstra. The Lipid Handbook, 3rd ed. Boca Raton: CRC Press, 2007. , Its chemical formula is CH3(CH2)14COOH, and its C:D (the total number of carbon atoms to the number of carbon-carbon double-bonds) is 16:0. It is a major component of the oil from the fruit of oil palms ( palm oil), making up to 44% of total fats. Meats, cheeses, butter, and other dairy products also contain palmitic acid, amounting to 50–60% of total fats. Palmitates are the salts and esters of palmitic acid. The palmitate anion is the observed form of palmitic acid at physiologic pH (7.4). Occurrence and production Palmitic acid was discovered by Edmond Frémy in 1840, in saponified palm oil. This remains the primary industrial route for its production, with the triglycerides (fa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carnitine O-palmitoyltransferase
Carnitine O-palmitoyltransferase (also called carnitine palmitoyltransferase) is a mitochondrial transferase enzyme () involved in the metabolism of palmitoylcarnitine into palmitoyl-CoA. A related transferase is carnitine acyltransferase. Molecules Image:Palmitoylcarnitine.PNG , Palmitoylcarnitine Image:Palmitoyl coenzyme A.svg, Palmitoyl CoA Pathway Human forms There are four different forms of CPT in humans: * CPT1A – associated with Carnitine palmitoyltransferase I deficiency * CPT1B * CPT1C * CPT2 – associated with carnitine palmitoyltransferase II deficiency See also * References External links * – Acyltransferases ChoActase / COT / CPT family in PROSITE Choline/Carnitine o-acyltransferase familyin Pfam Pfam is a database of protein families that includes their annotations and multiple sequence alignments generated using hidden Markov models. The most recent version, Pfam 35.0, was released in November 2021 and contains 19,632 families. Uses ... ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |