|
Polyetheramines
Polyetheramines are a group of chemicals that are aliphatic organic species based on both ether and amine groups. They are produced by reacting either ethylene oxide or propylene oxide with polyols and then aminating them. There are a number of commercially available molecules with different CAS Registry Number, CAS numbers and molecular weights. They often come with a prefix of M, D or T for monofunctional, difunctional and trifunctional respectively. D-230 would mean difunctional with a molecular weight of 230. A key use is for curing epoxy resins. Use as epoxy resin curatives One of the primary uses of polyether amines is as an epoxy curing agent. They are commercially available as mono, di and tri-functional. Difunctional versions tend to give flexibility and thus toughness to epoxy resin systems. Studies have been done on the cured properties of epoxy systems using different functionality polyetheramines. As epoxy curatives, they may then be further formulated into CASE appli ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mannich Reaction
In organic chemistry, the Mannich reaction is a three-component organic reaction that involves the amino alkylation of an acidic proton next to a carbonyl () functional group by formaldehyde () and a primary or secondary amine () or ammonia (). The final product is a β-amino-carbonyl compound also known as a Mannich base. Reactions between aldimines and α-methylene carbonyls are also considered Mannich reactions because these imines form between amines and aldehydes. The reaction is named after Carl Mannich. The Mannich reaction starts with the nucleophilic addition of an amine to a carbonyl group followed by dehydration to the Schiff base. The Schiff base is an electrophile which reacts in a second step in an electrophilic addition with an enol formed from a carbonyl compound containing an acidic alpha-proton. The Mannich reaction is a condensation reaction. In the Mannich reaction, primary or secondary amines or ammonia react with formaldehyde to form a Schiff base. T ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethylene Oxide
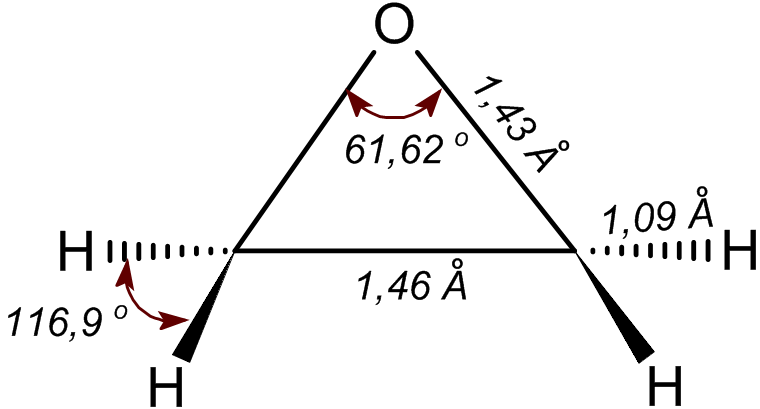
Ethylene oxide is an organic compound with the chemical formula, formula . It is a cyclic ether and the simplest epoxide: a three-membered ring (chemistry), ring consisting of one oxygen atom and two carbon atoms. Ethylene oxide is a colorless and flammable gas with a faintly sweet odor. Because it is a strained ring, ethylene oxide easily participates in a number of addition reactions that result in ring-opening. Ethylene oxide is isomeric with acetaldehyde and with vinyl alcohol. Ethylene oxide is industrially produced by oxidation of ethylene in the presence of a silver catalyst. The reactivity that is responsible for many of ethylene oxide's hazards also makes it useful. Although too dangerous for direct household use and generally unfamiliar to consumers, ethylene oxide is used for making many consumer products as well as non-consumer chemicals and intermediates. These products include detergents, thickeners, solvents, plastics, and various organic chemicals such as ethylen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Propylene Oxide
Propylene oxide is an epoxide with the molecular formula C3H6O. This colourless volatile liquid with an odour similar to ether, is produced on a large scale industrially. Its major application is its use for the production of polyether polyols for use in making polyurethane plastics. It is a chiral epoxide, although it is commonly used as a racemic mixture. This compound is sometimes called 1,2-propylene oxide to distinguish it from its isomer 1,3-propylene oxide, better known as oxetane. Production Industrial production of propylene oxide starts from propylene. Two general approaches are employed, one involving chlorohydrin formation and the other involving oxidation. In 2005, about half of the world production was through chlorohydrin technology and one half via oxidation routes. The latter approach is growing in importance. Chlorohydrin route The traditional route proceeds via the conversion of propylene to propylene chlorohydrin according to the following simplified sch ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polyol
In organic chemistry, a polyol is an organic compound containing multiple hydroxyl groups (). The term "polyol" can have slightly different meanings depending on whether it is used in food science or polymer chemistry. Polyols containing two, three and four hydroxyl groups are diols, triols, and tetrols, respectively. Classification Polyols may be classified according to their chemistry. Some of these chemistries are polyether, polyester, polycarbonate and also acrylic polyols. Polyether polyols may be further subdivided and classified as polyethylene oxide or polyethylene glycol (PEG), polypropylene glycol (PPG) and Polytetrahydrofuran or PTMEG. These have 2, 3 and 4 carbons respectively per oxygen atom in the repeat unit. Polycaprolactone polyols are also commercially available. There is also an increasing trend to use biobased (and hence renewable) polyols. Uses Polyether polyols have numerous uses. As an example, polyurethane foam is a big user of polyether polyols. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Epoxy
Epoxy is the family of basic components or Curing (chemistry), cured end products of epoxy Resin, resins. Epoxy resins, also known as polyepoxides, are a class of reactive prepolymers and polymers which contain epoxide groups. The epoxide functional group is also collectively called ''epoxy''. The IUPAC name for an epoxide group is an oxirane. Epoxy resins may be reacted (cross-linked) either with themselves through catalytic homopolymerisation, or with a wide range of co-reactants including polyfunctional amines, acids (and acid anhydrides), phenols, alcohols and thiols (sometimes called mercaptans). These co-reactants are often referred to as hardeners or curatives, and the cross-linking reaction is commonly referred to as Curing (chemistry), curing. Reaction of polyepoxides with themselves or with polyfunctional hardeners forms a thermosetting polymer, often with favorable mechanical properties and high thermal and chemical resistance. Epoxy has a wide range of application ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coating
A coating is a covering that is applied to the surface of an object, or substrate. The purpose of applying the coating may be decorative, functional, or both. Coatings may be applied as liquids, gases or solids e.g. powder coatings. Paints and lacquers are coatings that mostly have dual uses, which are protecting the substrate and being decorative, although some artists paints are only for decoration, and the paint on large industrial pipes is for identification (e.g. blue for process water, red for fire-fighting control) in addition to preventing corrosion. Along with corrosion resistance, functional coatings may also be applied to change the surface properties of the substrate, such as adhesion, wettability, or wear resistance.Howarth G.A "Synthesis of a legislation compliant corrosion protection coating system based on urethane, oxazolidine and waterborne epoxy technology" Master of Science Thesis April 1997 Imperial College London In other cases the coating adds a co ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Adhesive
Adhesive, also known as glue, cement, mucilage, or paste, is any non-metallic substance applied to one or both surfaces of two separate items that binds them together and resists their separation. The use of adhesives offers certain advantages over other binding techniques such as sewing, mechanical fastenings, and welding. These include the ability to bind different materials together, the more efficient distribution of stress across a joint, the cost-effectiveness of an easily mechanized process, and greater flexibility in design. Disadvantages of adhesive use include decreased stability at high temperatures, relative weakness in bonding large objects with a small bonding surface area, and greater difficulty in separating objects during testing. Adhesives are typically organized by the method of adhesion followed by ''reactive'' or ''non-reactive'', a term which refers to whether the adhesive chemically reacts in order to harden. Alternatively, they can be organized either ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sealant
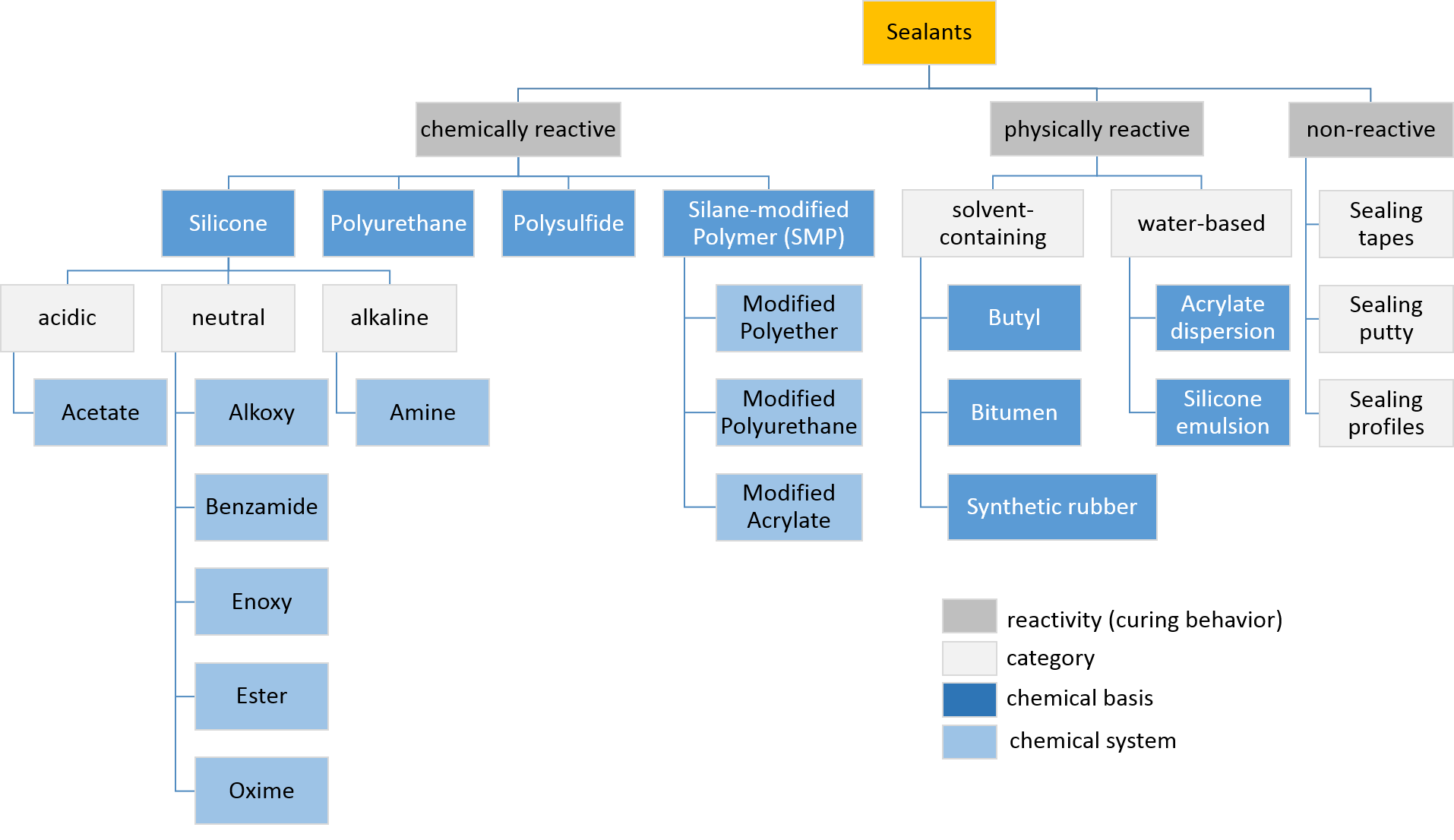
Sealant is a substance used to block the passage of fluids through openings in materials, a type of Seal (mechanical), mechanical seal. In building construction ''sealant'' is sometimes synonymous with ''caulk'' (especially if acrylic latex or polyurethane based) and also serve the purposes of blocking dust, sound and heat transmission. Sealants may be weak or strong, flexible or rigid, permanent or temporary. Sealants are not adhesives but some have adhesive qualities and are called ''adhesive-sealants'' or ''structural sealants''. History Sealants were first used in prehistory in the broadest sense as mud, grass and reeds to seal dwellings from the weather such as the daub in wattle and daub and thatching. Natural sealants and adhesive-sealants included plant resins such as pine pitch and birch pitch, bitumen, wax, tar, natural gum, clay (mud) mortar, lime mortar, lead, blood and egg. In the 17th century glazing putty was first used to seal window glass made with linseed oil a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Elastomer
An elastomer is a polymer with viscoelasticity (i.e. both viscosity and elasticity) and with weak intermolecular forces, generally low Young's modulus (E) and high failure strain compared with other materials. The term, a portmanteau of ''elastic polymer'', is often used interchangeably with ''rubber'', although the latter is preferred when referring to vulcanisates. Each of the monomers which link to form the polymer is usually a compound of several elements among carbon, hydrogen, oxygen and silicon. Elastomers are amorphous polymers maintained above their glass transition temperature, so that considerable molecular reconformation is feasible without breaking of covalent bonds. Rubber-like solids with elastic properties are called elastomers. Polymer chains are held together in these materials by relatively weak intermolecular bonds, which permit the polymers to stretch in response to macroscopic stresses. Elastomers are usually thermosets (requiring vulcanization ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mannich Base
A Mannich base is a beta- amino-ketone, which is formed in the reaction of an amine, formaldehyde (or an aldehyde) and a carbon acid. The Mannich base is an endproduct in the Mannich reaction, which is nucleophilic addition reaction of a non-enolizable aldehyde and any primary or secondary amine to produce resonance stabilized imine (iminium ion or imine salt). The addition of a carbanion from a CH acidic compound (any enolizable carbonyl compound, amide, carbamate, hydantoin or urea) to the imine gives the Mannich base.Mannich base applied in this example: Reactivity With primary or secondary amines, Mannich bases react with additional aldehyde and carbon acid to larger adducts HN(CH2CH2COR)2 and N(CH2CH2COR)3. With multiple acidic hydrogen atoms on the carbon acid higher adducts are also possible. Ammonia can be split off in an elimination reaction An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |