|
Peng-Robinson Equation
Cubic equations of state are a specific class of thermodynamic models for modeling the pressure of a gas as a function of temperature and density and which can be rewritten as a cubic function of the molar volume. Equations of state are generally applied in the fields of physical chemistry and chemical engineering, particularly in the modeling of vapor–liquid equilibrium and chemical engineering process design. Van der Waals equation of state The van der Waals equation of state may be written as : \left(p + \frac\right)\left(V_\text - b\right) = RT where T is the absolute temperature, p is the pressure, V_\text is the molar volume and R is the universal gas constant. Note that V_\text = V / n, where V is the volume, and n=N/N_\text, where n is the number of moles, N is the number of particles, and N_\text is the Avogadro constant. These definitions apply to all equations of state below as well. Proposed in 1873, the van der Waals equation of state was one of the first to pe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Equation Of State
In physics and chemistry, an equation of state is a thermodynamic equation relating state variables, which describe the state of matter under a given set of physical conditions, such as pressure, volume, temperature, or internal energy. Most modern equations of state are formulated in the Helmholtz free energy. Equations of state are useful in describing the properties of pure substances and mixtures in liquids, gases, and solid states as well as the state of matter in the interior of stars. Though there are many equations of state, none accurately predicts properties of substances under all conditions. The quest for a universal equation of state has spanned three centuries. Overview At present, there is no single equation of state that accurately predicts the properties of all substances under all conditions. An example of an equation of state correlates densities of gases and liquids to temperatures and pressures, known as the ideal gas law, which is roughly accurate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Compressibility Factor
In thermodynamics, the compressibility factor (Z), also known as the compression factor or the gas deviation factor, describes the deviation of a real gas from ideal gas behaviour. It is simply defined as the ratio of the molar volume of a gas to the molar volume of an ideal gas at the same temperature and pressure. It is a useful thermodynamic property for modifying the ideal gas law to account for the real gas behaviour.Properties of Natural Gases . Includes a chart of compressibility factors versus reduced pressure and reduced temperature (on last page of the PDF document) In general, deviation from ideal behaviour becomes more significant the closer a gas is to a phase change, the lower the temperat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vapor–liquid Equilibrium
In thermodynamics and chemical engineering, the vapor–liquid equilibrium (VLE) describes the distribution of a chemical species between the vapor phase and a liquid phase. The Vapor quality, concentration of a vapor in contact with its liquid, especially at Thermodynamic equilibrium, equilibrium, is often expressed in terms of vapor pressure, which will be a partial pressure (a part of the total gas pressure) if any other gas(es) are present with the vapor. The equilibrium vapor pressure of a liquid is in general strongly dependent on temperature. At vapor–liquid equilibrium, a liquid with individual components in certain concentrations will have an equilibrium vapor in which the concentrations or partial pressures of the vapor components have certain values depending on all of the liquid component concentrations and the temperature. The converse is also true: if a vapor with components at certain concentrations or partial pressures is in vapor–liquid equilibrium with its ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Departure Function
In thermodynamics, a departure function is defined for any thermodynamic property as the difference between the property as computed for an ideal gas and the property of the species as it exists in the real world, for a specified temperature ''T'' and pressure ''P''. Common departure functions include those for enthalpy, entropy, and internal energy. Departure functions are used to calculate real fluid extensive properties (i.e. properties which are computed as a difference between two states). A departure function gives the difference between the real state, at a finite volume or non-zero pressure and temperature, and the ideal state, usually at zero pressure or infinite volume and temperature. For example, to evaluate enthalpy change between two points ''h''(''v''1,''T''1) and ''h''(''v''2,''T''2) we first compute the enthalpy departure function between volume ''v''1 and infinite volume at ''T'' = ''T''1, then add to that the ideal gas enthalpy change due to the tempe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ding Yu Peng
Ding-Yu Peng is a professor of Chemical Engineering at the University of Saskatchewan. Under the direction of Donald B. Robinson, Peng introduced a two-parameter cubic equation of state now known as the Peng–Robinson equation of state during the 1970s while a research engineer at the University of Alberta. Early life Ding-Yu Peng was born in China sometime in 1943. Education Peng completed a degree in chemical engineering at the National Taiwan University in 1966. He studied for one year at Syracuse University during 1968-1969. Subsequently, he followed Leonard I. Stiel to the University of Missouri and obtained his PhD. in chemical engineering in 1973. Work under Robinson In late 1974, while working as a post-doctoral fellow under Robinson, the Natural Gas Processors Association requested a better gas model than was available at that time. Work on the Peng–Robinson equation of state was completed in 1975, and the results were published the following year. Teachi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
University Of Alberta
The University of Alberta (also known as U of A or UAlberta, ) is a public research university located in Edmonton, Alberta, Canada. It was founded in 1908 by Alexander Cameron Rutherford, the first premier of Alberta, and Henry Marshall Tory, the university's first president. It was enabled through the ''Post-secondary Learning Act.'' The university is considered a "comprehensive academic and research university" (CARU), which means that it offers a range of academic and professional programs that generally lead to undergraduate and graduate level credentials. The university comprises four campuses in Edmonton, an Augustana Campus in Camrose, Alberta, Camrose, and a staff centre in downtown Calgary. The original north campus consists of 150 buildings covering 50 city blocks on the south rim of the North Saskatchewan River valley parks system, North Saskatchewan River valley, across and west from downtown Edmonton. About 37,000 students from Canada and 150 other countries partici ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vapor–liquid Equilibrium
In thermodynamics and chemical engineering, the vapor–liquid equilibrium (VLE) describes the distribution of a chemical species between the vapor phase and a liquid phase. The Vapor quality, concentration of a vapor in contact with its liquid, especially at Thermodynamic equilibrium, equilibrium, is often expressed in terms of vapor pressure, which will be a partial pressure (a part of the total gas pressure) if any other gas(es) are present with the vapor. The equilibrium vapor pressure of a liquid is in general strongly dependent on temperature. At vapor–liquid equilibrium, a liquid with individual components in certain concentrations will have an equilibrium vapor in which the concentrations or partial pressures of the vapor components have certain values depending on all of the liquid component concentrations and the temperature. The converse is also true: if a vapor with components at certain concentrations or partial pressures is in vapor–liquid equilibrium with its ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
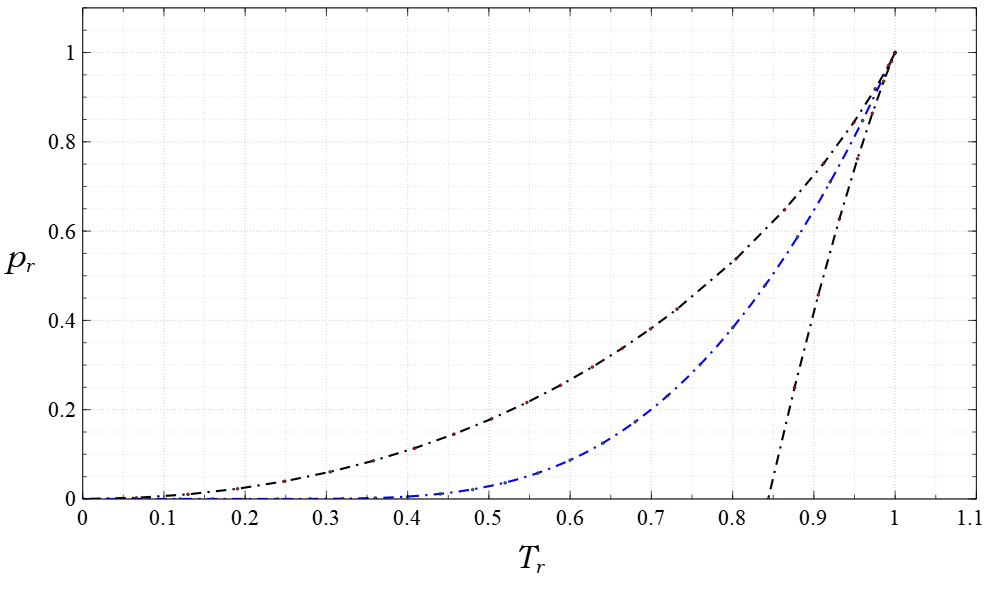
Acentric Factor
The acentric factor is a conceptual number introduced by Kenneth Pitzer in 1955, proven to be useful in the description of fluids. It has become a standard for the phase characterization of single and pure components, along with other state description parameters such as molecular weight, critical temperature, critical pressure, and critical volume (or critical compressibility). The acentric factor is also said to be a measure of the non-sphericity (centricity) of molecules. Pitzer defined from the relationship : \omega = -\log_(p^\text_\text) - 1 \text T_\text = 0.7, where p^\text_\text = p^\text / p_c is the reduced saturation vapor pressure, and T_\text = T / T_c is the reduced temperature. Pitzer developed this factor by studying the vapor-pressure curves of various pure substances. Thermodynamically, the vapor-pressure curve for pure components can be mathematically described using the Clausius–Clapeyron equation. The integrated form of equation is mainly used fo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrogen
Nitrogen is a chemical element; it has Symbol (chemistry), symbol N and atomic number 7. Nitrogen is a Nonmetal (chemistry), nonmetal and the lightest member of pnictogen, group 15 of the periodic table, often called the Pnictogen, pnictogens. It is a common element in the universe, estimated at Abundance of the chemical elements, seventh in total abundance in the Milky Way and the Solar System. At standard temperature and pressure, two atoms of the element chemical bond, bond to form N2, a colourless and odourless diatomic molecule, diatomic gas. N2 forms about 78% of Atmosphere of Earth, Earth's atmosphere, making it the most abundant chemical species in air. Because of the volatility of nitrogen compounds, nitrogen is relatively rare in the solid parts of the Earth. It was first discovered and isolated by Scottish physician Daniel Rutherford in 1772 and independently by Carl Wilhelm Scheele and Henry Cavendish at about the same time. The name was suggested by French chemist ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Water
Water is an inorganic compound with the chemical formula . It is a transparent, tasteless, odorless, and Color of water, nearly colorless chemical substance. It is the main constituent of Earth's hydrosphere and the fluids of all known living organisms (in which it acts as a solvent). It is vital for all known forms of life, despite not providing food energy or organic micronutrients. Its chemical formula, , indicates that each of its molecules contains one oxygen and two hydrogen atoms, connected by covalent bonds. The hydrogen atoms are attached to the oxygen atom at an angle of 104.45°. In liquid form, is also called "water" at standard temperature and pressure. Because Earth's environment is relatively close to water's triple point, water exists on Earth as a solid, a liquid, and a gas. It forms precipitation in the form of rain and aerosols in the form of fog. Clouds consist of suspended droplets of water and ice, its solid state. When finely divided, crystalline ice ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon Dioxide
Carbon dioxide is a chemical compound with the chemical formula . It is made up of molecules that each have one carbon atom covalent bond, covalently double bonded to two oxygen atoms. It is found in a gas state at room temperature and at normally-encountered concentrations it is odorless. As the source of carbon in the carbon cycle, atmospheric is the primary carbon source for life on Earth. In the air, carbon dioxide is transparent to visible light but absorbs infrared, infrared radiation, acting as a greenhouse gas. Carbon dioxide is soluble in water and is found in groundwater, lakes, ice caps, and seawater. It is a trace gas Carbon dioxide in Earth's atmosphere, in Earth's atmosphere at 421 parts per million (ppm), or about 0.042% (as of May 2022) having risen from pre-industrial levels of 280 ppm or about 0.028%. Burning fossil fuels is the main cause of these increased concentrations, which are the primary cause of climate change.IPCC (2022Summary for pol ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Van Der Waals Equation
The van der Waals equation is a mathematical formula that describes the behavior of real gases. It is an equation of state that relates the pressure, volume, Avogadro's law, number of molecules, and temperature in a fluid. The equation modifies the ideal gas law in two ways: first, it considers particles to have a finite diameter (whereas an ideal gas consists of point particles); second, its particles interact with each other (unlike an ideal gas, whose particles move as though alone in the volume). The equation is named after Dutch physicist Johannes Diderik van der Waals, who first derived it in 1873 as part of his doctoral thesis. Van der Waals based the equation on the idea that fluids are composed of discrete particles, which few scientists believed existed. However, the equation accurately predicted the behavior of a fluid around its Critical point (thermodynamics), critical point, which had been discovered a few years earlier. Its qualitative and quantitative agreement w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |