|
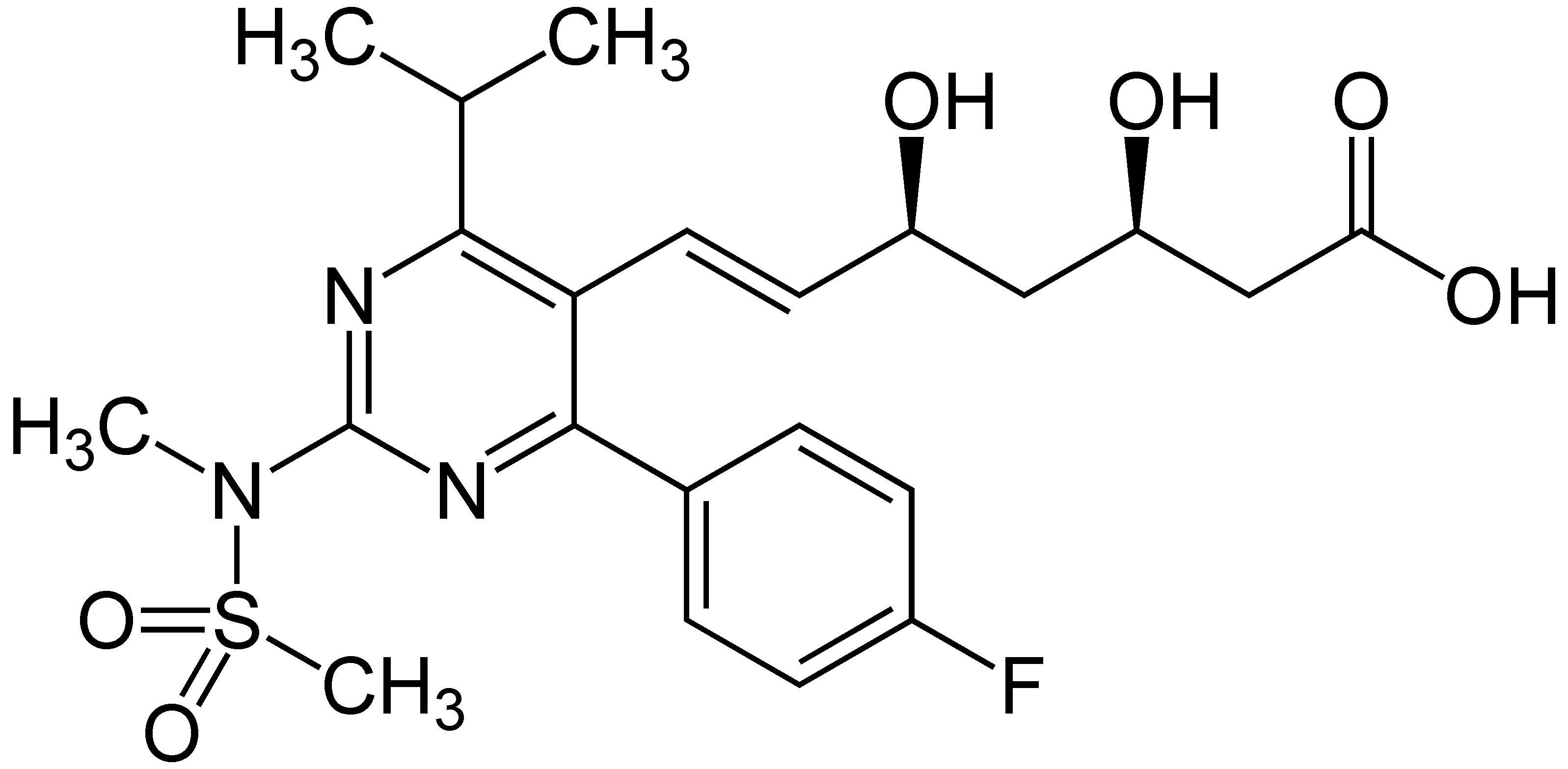
Luxturna
Voretigene neparvovec, sold under the brand name Luxturna, is a gene therapy medication for the treatment of Leber congenital amaurosis. Leber's congenital amaurosis, or biallelic RPE65-mediated inherited retinal disease, is an inherited disorder causing progressive blindness. Voretigene is the first treatment available for this condition. The gene therapy is not a cure for the condition, but substantially improves vision in those treated. It is given as a subretinal injection. Voretigene neparvovec was approved for medical use in the United States in December 2017, Australia in August 2020 and in Canada, in October 2020. It is the first ''in vivo'' gene therapy approved by the US Food and Drug Administration (FDA). Medical uses Voretigene neparvovec is indicated for the treatment of people with vision loss due to inherited retinal dystrophy caused by confirmed biallelic RPE65 mutations and who have sufficient viable retinal cells. Chemistry and production Voretigene nepar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gene Therapy
Gene therapy is a medical field which focuses on the genetic modification of cells to produce a therapeutic effect or the treatment of disease by repairing or reconstructing defective genetic material. The first attempt at modifying human DNA was performed in 1980, by Martin Cline, but the first successful nuclear gene transfer in humans, approved by the National Institutes of Health, was performed in May 1989. The first therapeutic use of gene transfer as well as the first direct insertion of human DNA into the nuclear genome was performed by French Anderson in a trial starting in September 1990. It is thought to be able to cure many genetic disorders or treat them over time. Between 1989 and December 2018, over 2,900 clinical trials were conducted, with more than half of them in phase I.Gene Therapy Cli ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gene Therapy
Gene therapy is a medical field which focuses on the genetic modification of cells to produce a therapeutic effect or the treatment of disease by repairing or reconstructing defective genetic material. The first attempt at modifying human DNA was performed in 1980, by Martin Cline, but the first successful nuclear gene transfer in humans, approved by the National Institutes of Health, was performed in May 1989. The first therapeutic use of gene transfer as well as the first direct insertion of human DNA into the nuclear genome was performed by French Anderson in a trial starting in September 1990. It is thought to be able to cure many genetic disorders or treat them over time. Between 1989 and December 2018, over 2,900 clinical trials were conducted, with more than half of them in phase I.Gene Therapy Cli ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Leber Congenital Amaurosis
Leber congenital amaurosis (LCA) is a rare inherited eye disease that appears at birth or in the first few months of life. It affects about 1 in 40,000 newborns. LCA was first described by Theodor Leber in the 19th century. It should not be confused with Leber's hereditary optic neuropathy, which is a different disease also described by Theodor Leber. One form of LCA was successfully treated with gene therapy in 2008. Signs and symptoms The term congenital refers to a condition present from birth (not acquired) and amaurosis refers to a loss of vision not associated with a lesion. However, beyond these general descriptions, the presentation of LCA can vary, because it is associated with multiple genes. LCA is typically characterized by nystagmus, sluggish or absent pupillary responses, and severe vision loss or blindness. Genetics It is usually autosomal recessive; however, importantly for family planning, it is sometimes autosomal dominant. It is a disorder thought to be ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Priority Review (FDA)
Priority review is a program of the United States Food and Drug Administration (FDA) to expedite the review process for drugs that are expected to have a particularly great impact on the treatment of a disease. The priority review voucher program is a program that grants a voucher for priority review to a drug developer as an incentive to develop treatments for disease indications with limited profitability. Priority review vouchers are currently earned by pharmaceutical companies for the development and approval of drugs treating neglected tropical diseases, rare pediatric diseases, and "medical countermeasures" for terrorism. The voucher can be used for future drugs that could have wider indications for use, but the company is required to pay a fee (approximately $2.8 million) to use the voucher. When seeking approval for a drug, manufacturers can apply to the FDA for priority review. This is granted when a drug is intended to treat a serious condition and would "provide a sig ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Priority Review
Priority review is a program of the United States Food and Drug Administration (FDA) to expedite the review process for drugs that are expected to have a particularly great impact on the treatment of a disease. The priority review voucher program is a program that grants a voucher for priority review to a drug developer as an incentive to develop treatments for disease indications with limited profitability. Priority review vouchers are currently earned by pharmaceutical companies for the development and approval of drugs treating neglected tropical diseases, rare pediatric diseases, and "medical countermeasures" for terrorism. The voucher can be used for future drugs that could have wider indications for use, but the company is required to pay a fee (approximately $2.8 million) to use the voucher. When seeking approval for a drug, manufacturers can apply to the FDA for priority review. This is granted when a drug is intended to treat a serious condition and would "provide a si ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Spark Therapeutics
Spark Therapeutics, Inc. is a developer of gene therapy treatments, which treat debilitating genetic diseases. It is a subsidiary of Hoffmann-La Roche. History The company was founded in 2013 by Katherine A. High, Jeffrey Marrazzo, and Steven Altschuler in an effort to commercially develop treatments against haemophilia that High was working on at Children's Hospital of Philadelphia. In January 2015, the company became a public company, trading under the ticker $ONCE via a $161 million initial public offering led by Chief Legal Officer Joseph La Barge. In December 2017, the U.S. Food and Drug Administration approved LUXTURNATM (voretigene neparvovec-rzyl) for the treatment of patients with viable retinal cells and confirmed biallelic RPE65 mutation-associated retinal dystrophy, a genetic blinding condition caused by mutations in the RPE65 gene. The company is currently developing several gene therapies to target a suite of diseases, including Haemophilia A and B, and several cen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
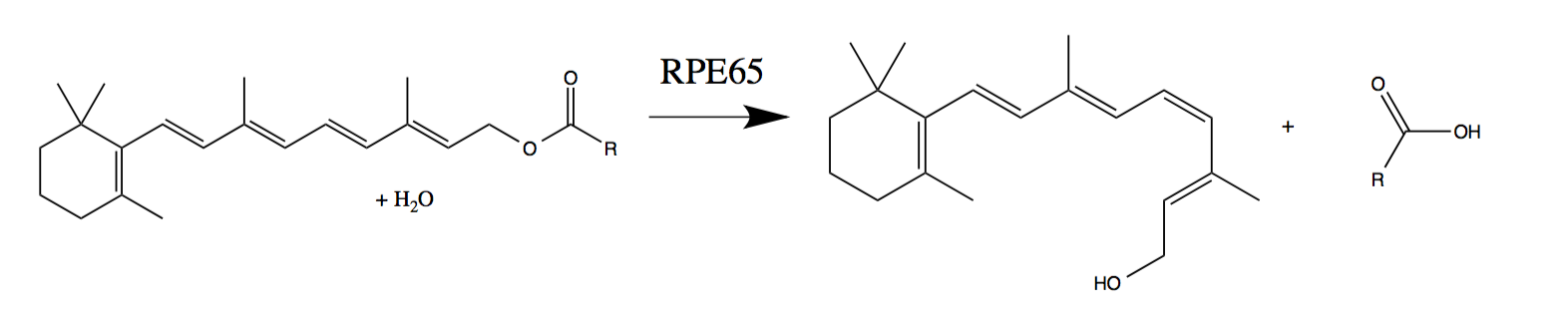
RPE65
Retinal pigment epithelium-specific 65 kDa protein, also known as retinoid isomerohydrolase, is an enzyme of the vertebrate visual cycle that is encoded in humans by the ''RPE65'' gene. RPE65 is expressed in the retinal pigment epithelium (RPE, a layer of epithelial cells that nourish the photoreceptor cells) and is responsible for the conversion of all-trans- retinyl esters to 11-cis-retinol during phototransduction. 11-cis-retinol is then used in visual pigment regeneration in photoreceptor cells. RPE65 belongs to the carotenoid oxygenase family of enzymes. Function RPE65 is a critical enzyme in the vertebrate visual cycle found in the retinal pigmented epithelium. It is also found in rods and cones. The photoisomerization of 11-cis-retinal to all-trans-retinal initiates the phototransduction pathway through which the brain detects light. All-trans-retinol is not photoactive and therefore must be reconverted to 11-cis-retinal before it can recombine with opsin to form an acti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
HEK 293 Cells
Human embryonic kidney 293 cells, also often referred to as HEK 293, HEK-293, 293 cells, or less precisely as HEK cells, are a specific immortalised cell line derived from a spontaneously miscarried or aborted fetus or human embryonic kidney cells grown in tissue culture taken from a female fetus in 1973. HEK 293 cells have been widely used in cell biology research for many years, because of their reliable growth and propensity for transfection. They are also used by the biotechnology industry to produce therapeutic proteins and viruses for gene therapy as well as safety testing for a vast array of chemicals. 293T (or HEK 293T) is a derivative human cell line that expresses a mutant version of the SV40 large T antigen. It is very commonly used in biological research for making proteins and producing recombinant retroviruses. History HEK 293 cells were generated in 1973 by transfection of cultures of normal human embryonic kidney cells with sheared adenovirus 5 DNA in Alex v ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phase III Clinical Trial
The phases of clinical research are the stages in which scientists conduct experiments with a health intervention to obtain sufficient evidence for a process considered effective as a medical treatment. For drug development, the clinical phases start with testing for safety in a few human subjects, then expand to many study participants (potentially tens of thousands) to determine if the treatment is effective. Clinical research is conducted on drug candidates, vaccine candidates, new medical devices, and new diagnostic assays. Summary Clinical trials testing potential medical products are commonly classified into four phases. The drug development process will normally proceed through all four phases over many years. If the drug successfully passes through Phases I, II, and III, it will usually be approved by the national regulatory authority for use in the general population. Phase IV trials are 'post-marketing' or 'surveillance' studies conducted to monitor safety over sever ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Biologics License Application
A biologics license application (BLA) is defined by the U.S. Food and Drug Administration (FDA) as follows: The biologics license application is a request for permission to introduce, or deliver for introduction, a biologic product into interstate commerce (21 CFR 601.2). The BLA is regulated under 21 CFR 600 – 680. A BLA is submitted by any legal person or entity who is engaged in manufacture or an applicant for a license who takes responsibility for compliance with product and establishment standards. Form 356h specifies the requirements for a BLA. This includes: * Applicant information * Product/manufacturing information * Pre-clinical studies * Clinical studies * Labeling Some biological products are regulated by the Center for Drug Evaluation and Research (CDER) while others are regulated by Center for Biologics Evaluation and Research The Center for Biologics Evaluation and Research (CBER) is one of six main centers for the Food and Drug Administration, U.S. Food and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Retinitis Pigmentosa
Retinitis pigmentosa (RP) is a genetic disorder of the eyes that causes loss of vision. Symptoms include trouble seeing at night and decreasing peripheral vision (side and upper or lower visual field). As peripheral vision worsens, people may experience "tunnel vision". Complete blindness is uncommon. Onset of symptoms is generally gradual and often begins in childhood. Retinitis pigmentosa is generally inherited from one or both parents or rarely it can be caused by a miscoding during DNA division. It is caused by genetic miscoding of proteins in one of more than 300 genes involved. The underlying mechanism involves the progressive loss of rod photoreceptor cells that line the retina of the eyeball. The rod cells secrete a neuroprotective substance (Rod-derived cone viability factor, RdCVF) that protects the cone cells from apoptosis (cell death). However, when the rod cells die, this substance is no longer provided. This is generally followed by the loss of cone photorecept ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Orphan Drug
An orphan drug is a pharmaceutical agent developed to treat medical conditions which, because they are so rare, would not be profitable to produce without government assistance. The conditions are referred to as orphan diseases. The assignment of orphan status to a disease and to drugs developed to treat it is a matter of public policy in many countries and has yielded medical breakthroughs that might not otherwise have been achieved, due to the economics of drug research and development. In the U.S. and the EU, it is easier to gain marketing approval for an orphan drug. There may be other financial incentives, such as an extended period of exclusivity, during which the producer has sole rights to market the drug. All are intended to encourage development of drugs which would otherwise lack sufficient profit motive to attract corporate research budgets and personnel. Definition According to the US Food and Drug Administration (FDA), an orphan drug is defined as one "intended for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |