|
Lucas Cell
A Lucas cell is a type of scintillation counter. It is used to acquire a gas sample, filter out the radioactive particulates through a special filter and then count the radioactive decay. The inside of the gas chamber is coated with ZnS( Ag) - a chemical that emits light when struck by alpha particles. A photomultiplier tube at the top of the chamber counts the photons and sends the count to a data logger. Radon measurement A Lucas cell can be used to measure radon gas concentrations. Radon itself is an inert gas. Its danger lies in the fact that it undergoes radioactive decay. The radon decay products may lodge in the lungs and bombard them with alpha and beta particles, thus increasing the risk of lung cancer. See also * Geiger counter A Geiger counter (, ; also known as a Geiger–Müller counter or G-M counter) is an electronic instrument for detecting and measuring ionizing radiation with the use of a Geiger–Müller tube. It is widely used in applications such as ra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lucas Cell Number 7
Lucas or LUCAS may refer to: People * Lucas (surname) * Lucas (given name) Arts and entertainment * Luca Family Singers, or the Lucas, a 19th-century African-American singing group * Lucas, a 1960s Swedish pop group formed by Janne Lucas Persson * ''Lucas'' (album) (2007), an album by Skeletons and the Kings of All Cities * ''Lucas'' (1986 film), an American romantic-comedy teen film * ''Lucas'' (2021 film), a Spanish thriller drama film * ''Lucas'' (novel) (2003), by Kevin Brooks Organisations * Lucas Industries, a former British manufacturer of motor industry and aerospace industry components * Lucasfilm, an American film and television production company * LucasVarity, a defunct British automotive parts manufacturer, successor to Lucas Industries Places Australia * Lucas, Victoria Mexico * Cabo San Lucas, Baja California United States * Lucas, Illinois * Lucas, Iowa * Lucas County, Iowa * Lucas, Kansas * Lucas, Kentucky * Lucas, Michigan * Lucas, Missouri * Luc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Scintillation Counter
A scintillation counter is an instrument for detecting and measuring ionizing radiation by using the Electron excitation, excitation effect of incident radiation on a Scintillation (physics), scintillating material, and detecting the resultant light pulses. It consists of a scintillator which generates photons in response to incident radiation, a sensitive photodetector (usually a photomultiplier tube (PMT), a charge-coupled device (CCD) camera, or a photodiode), which converts the light to an electrical signal and electronics to process this signal. Scintillation counters are widely used in radiation protection, assay of radioactive materials and physics research because they can be made inexpensively yet with good quantum efficiency, and can measure both the intensity and the Electronvolt, energy of incident radiation. History The first electronic scintillation counter was invented in 1944 by Samuel Curran, Sir Samuel Curran whilst he was working on the Manhattan Project at th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
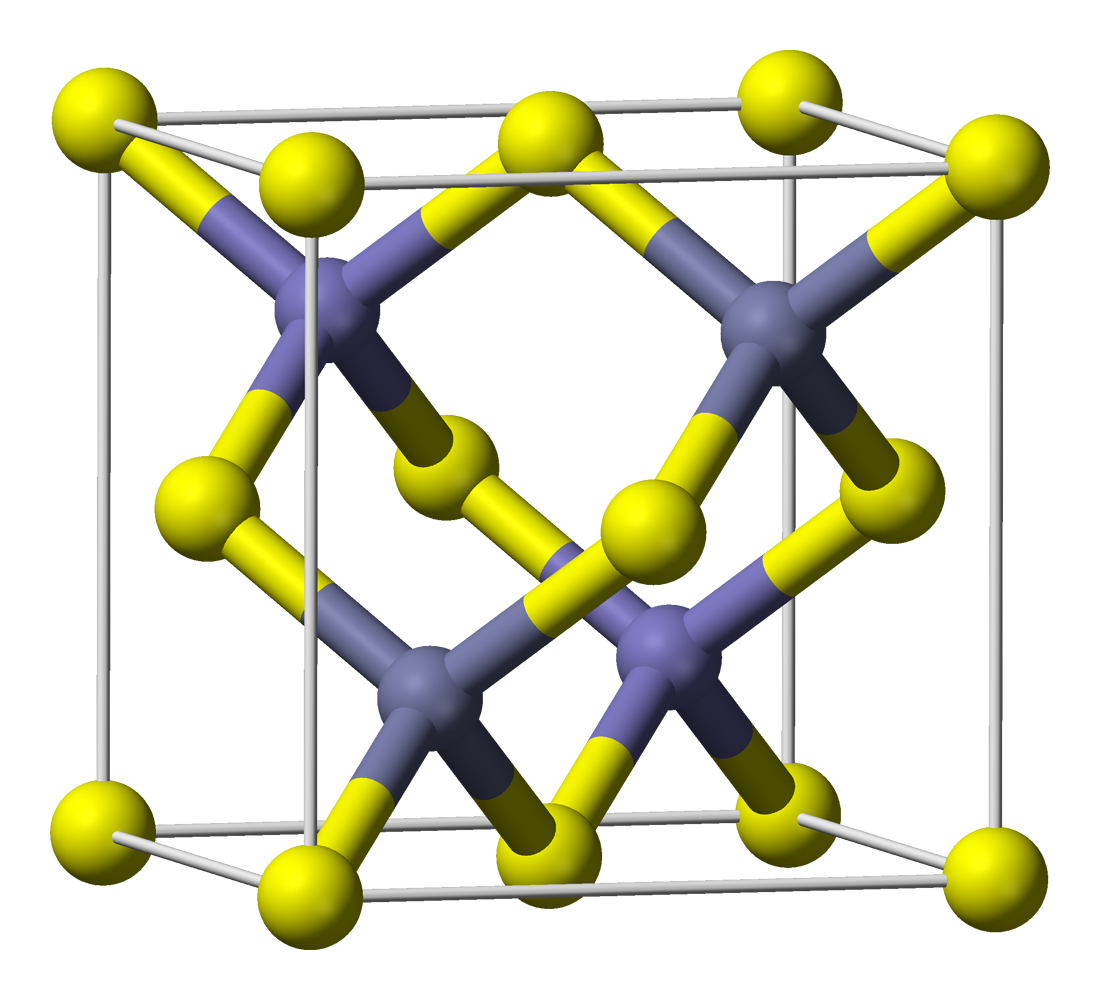
Zinc Sulfide
Zinc sulfide (or zinc sulphide) is an inorganic compound with the chemical formula of ZnS. This is the main form of zinc found in nature, where it mainly occurs as the mineral sphalerite. Although this mineral is usually black because of various impurities, the pure material is white, and it is widely used as a pigment. In its dense synthetic form, zinc sulfide can be transparency and translucency, transparent, and it is used as a window for visible light, visible optics and infrared optics. Structure ZnS exists in two main crystalline forms. This dualism is an example of polymorphism (materials science), polymorphism. In each form, the coordination geometry at Zn and S is tetrahedral. The more stable cubic form is known also as zinc blende or sphalerite. The hexagonal form is known as the mineral wurtzite, although it also can be produced synthetically.. The transition from the sphalerite form to the wurtzite form occurs at around 1020 °C. Applications Luminescent mate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silver
Silver is a chemical element; it has Symbol (chemistry), symbol Ag () and atomic number 47. A soft, whitish-gray, lustrous transition metal, it exhibits the highest electrical conductivity, thermal conductivity, and reflectivity of any metal. Silver is found in the Earth's crust in the pure, free elemental form ("native metal, native silver"), as an alloy with gold and other metals, and in minerals such as argentite and chlorargyrite. Most silver is produced as a byproduct of copper, gold, lead, and zinc Refining (metallurgy), refining. Silver has long been valued as a precious metal. Silver metal is used in many bullion coins, sometimes bimetallism, alongside gold: while it is more abundant than gold, it is much less abundant as a native metal. Its purity is typically measured on a per-mille basis; a 94%-pure alloy is described as "0.940 fine". As one of the seven metals of antiquity, silver has had an enduring role in most human cultures. Other than in currency and as an in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alpha Particles
Alpha particles, also called alpha rays or alpha radiation, consist of two protons and two neutrons bound together into a particle identical to a helium-4 nucleus. They are generally produced in the process of alpha decay but may also be produced in different ways. Alpha particles are named after the first letter in the Greek alphabet, α. The symbol for the alpha particle is α or α2+. Because they are identical to helium nuclei, they are also sometimes written as He2+ or 2+ indicating a helium ion with a +2 charge (missing its two electrons). Once the ion gains electrons from its environment, the alpha particle becomes a normal (electrically neutral) helium atom . Alpha particles have a net spin of zero. When produced in standard alpha radioactive decay, alpha particles generally have a kinetic energy of about 5 MeV and a velocity in the vicinity of 4% of the speed of light. They are a highly ionizing form of particle radiation, with low penetration depth (stopped by a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Photomultiplier
A photomultiplier is a device that converts incident photons into an electrical signal. Kinds of photomultiplier include: * Photomultiplier tube, a vacuum tube converting incident photons into an electric signal. Photomultiplier tubes (PMTs for short) are members of the class of vacuum tubes, and more specifically vacuum phototubes, which are extremely sensitive detectors of light in the ultraviolet, visible, and near-infrared ranges of the electromagnetic spectrum. ** Magnetic photomultiplier, developed by the Soviets in the 1930s. ** Electrostatic photomultiplier, a kind of photomultiplier tube demonstrated by Jan Rajchman of RCA Laboratories in Princeton, NJ in the late 1930s which became the standard for all future commercial photomultipliers. The first mass-produced photomultiplier, the Type 931, was of this design and is still commercially produced today. * Silicon photomultiplier, a solid-state device converting incident photons into an electric signal. Silicon photomul ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Photons
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless particles that can move no faster than the speed of light measured in vacuum. The photon belongs to the class of boson particles. As with other elementary particles, photons are best explained by quantum mechanics and exhibit wave–particle duality, their behavior featuring properties of both waves and particles. The modern photon concept originated during the first two decades of the 20th century with the work of Albert Einstein, who built upon the research of Max Planck. While Planck was trying to explain how matter and electromagnetic radiation could be in thermal equilibrium with one another, he proposed that the energy stored within a material object should be regarded as composed of an integer number of discrete, equal-sized parts. To explain the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radon
Radon is a chemical element; it has symbol Rn and atomic number 86. It is a radioactive noble gas and is colorless and odorless. Of the three naturally occurring radon isotopes, only Rn has a sufficiently long half-life (3.825 days) for it to be released from the soil and rock where it is generated. Radon isotopes are the immediate decay products of radium isotopes. The instability of Rn, its most stable isotope, makes radon one of the rarest elements. Radon will be present on Earth for several billion more years despite its short half-life, because it is constantly being produced as a step in the decay chains of U and Th, both of which are abundant radioactive nuclides with half-lives of at least several billion years. The decay of radon produces many other short-lived nuclides, known as "radon daughters", ending at stable isotopes of lead. Rn occurs in significant quantities as a step in the normal radioactive decay chain of U, also known as the uranium series, which slo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Inert Gas
An inert gas is a gas that does not readily undergo chemical reactions with other chemical substances and therefore does not readily form chemical compounds. Though inert gases have a variety of applications, they are generally used to prevent unwanted chemical reactions with the oxygen (oxidation) and moisture (hydrolysis) in the air from degrading a sample. Generally, all noble gases except oganesson (helium, neon, argon, krypton, xenon, and radon), nitrogen, and carbon dioxide are considered inert gases. The term ''inert gas'' is context-dependent because several of the inert gases, including nitrogen and carbon dioxide, can be made to react under certain conditions. Purified argon gas is the most commonly used inert gas due to its high natural abundance (78.3% N2, 1% Ar in air) and low relative cost. Unlike noble gases, an inert gas is not necessarily elemental and is often a compound gas. Like the noble gases, the tendency for non-reactivity is due to the valence, the ou ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radioactive Decay
Radioactive decay (also known as nuclear decay, radioactivity, radioactive disintegration, or nuclear disintegration) is the process by which an unstable atomic nucleus loses energy by radiation. A material containing unstable nuclei is considered ''radioactive''. Three of the most common types of decay are Alpha decay, alpha, Beta decay, beta, and Gamma ray, gamma decay. The weak force is the Fundamental interactions, mechanism that is responsible for beta decay, while the other two are governed by the electromagnetic force, electromagnetic and nuclear forces. Radioactive decay is a randomness, random process at the level of single atoms. According to quantum mechanics, quantum theory, it is impossible to predict when a particular atom will decay, regardless of how long the atom has existed. However, for a significant number of identical atoms, the overall decay rate can be expressed as a decay constant or as a half-life. The half-lives of radioactive atoms have a huge range: f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alpha Radiation
Alpha decay or α-decay is a type of radioactive decay in which an atomic nucleus emits an alpha particle (helium nucleus). The parent nucleus transforms or "decays" into a daughter product, with a mass number that is reduced by four and an atomic number that is reduced by two. An alpha particle is identical to the nucleus of a helium-4 atom, which consists of two protons and two neutrons. It has a charge of and a mass of , and is represented as ^_\alpha. For example, uranium-238 undergoes alpha decay to form thorium-234. While alpha particles have a charge , this is not usually shown because a nuclear equation describes a nuclear reaction without considering the electrons – a convention that does not imply that the nuclei necessarily occur in neutral atoms. Alpha decay typically occurs in the heaviest nuclides. Theoretically, it can occur only in nuclei somewhat heavier than nickel (element 28), where the overall binding energy per nucleon is no longer a maximum and the nuc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Beta Radiation
A beta particle, also called beta ray or beta radiation (symbol β), is a high-energy, high-speed electron or positron emitted by the radioactive decay of an atomic nucleus, known as beta decay. There are two forms of beta decay, β− decay and β+ decay, which produce electrons and positrons, respectively. Beta particles with an energy of 0.5 MeV have a range of about one metre in the air; the distance is dependent on the particle's energy and the air's density and composition. Beta particles are a type of ionizing radiation, and for radiation protection purposes, they are regarded as being more ionising than gamma rays, but less ionising than alpha particles. The higher the ionising effect, the greater the damage to living tissue, but also the lower the penetrating power of the radiation through matter. Beta decay modes β− decay (electron emission) An unstable atomic nucleus with an excess of neutrons may undergo β− decay, where a neutron is converted into a prot ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |