|
Estrone
Estrone (E1), also spelled oestrone, is a steroid, a weak estrogen, and a minor female sex hormone. It is one of three major endogenous estrogens, the others being estradiol and estriol. Estrone, as well as the other estrogens, are synthesized from cholesterol and secreted mainly from the gonads, though they can also be formed from adrenal androgens in adipose tissue. Relative to estradiol, both estrone and estriol have far weaker activity as estrogens. Estrone can be converted into estradiol, and serves mainly as a precursor or metabolic intermediate of estradiol. It is both a precursor and metabolite of estradiol. In addition to its role as a natural hormone, estrone has been used as a medication, for instance in menopausal hormone therapy; for information on estrone as a medication, see the estrone (medication) article. Biological activity Estrone is an estrogen, specifically an agonist of the estrogen receptors ERα and ERβ. It is a far less potent estrogen than is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Estrone (medication)
Estrone (E1), sold under the brand names Estragyn, Kestrin, and Theelin among many others, is an estrogen medication and naturally occurring steroid hormone which has been used in menopausal hormone therapy and for other indications. It has been provided as an aqueous suspension or oil solution given by injection into muscle and as a vaginal cream applied inside of the vagina. It can also be taken by mouth as estradiol/estrone/estriol (brand name Hormonin) and in the form of prodrugs like estropipate (estrone sulfate; brand name Ogen) and conjugated estrogens (mostly estrone sulfate; brand name Premarin). Side effects of estrogens like estrone include breast tenderness, breast enlargement, headache, nausea, fluid retention, and edema, among others. Estrone is a naturally occurring and bioidentical estrogen, or an agonist of the estrogen receptor, the biological target of estrogens like endogenous estradiol. It is a relatively weak estrogen, with much lower activity tha ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Estradiol
Estradiol (E2), also called oestrogen, oestradiol, is an estrogen steroid hormone and the major female sex hormone. It is involved in the regulation of female reproductive cycles such as estrous and menstrual cycles. Estradiol is responsible for the development of female secondary sexual characteristics such as the breasts, widening of the hips and a female pattern of fat distribution. It is also important in the development and maintenance of female reproductive tissues such as the mammary glands, uterus and vagina during puberty, adulthood and pregnancy. It also has important effects in many other tissues including bone, fat, skin, liver, and the brain. Though estradiol levels in males are much lower than in females, estradiol has important roles in males as well. Apart from humans and other mammals, estradiol is also found in most vertebrates and crustaceans, insects, fish, and other animal species. Estradiol is produced within the follicles of the ovaries and in oth ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
G Protein-coupled Estrogen Receptor
G protein-coupled estrogen receptor 1 (GPER), also known as G protein-coupled receptor 30 (GPR30), is a protein that in humans is encoded by the ''GPER'' gene. GPER binds to and is activated by the female sex hormone estradiol and is responsible for some of the rapid effects that estradiol has on cells. Discovery The classical estrogen receptors first characterized in 1958 are water-soluble proteins located in the interior of cells that are activated by estrogenenic hormones such as estradiol and several of its metabolites such as estrone or estriol. These proteins belong to the nuclear hormone receptor class of transcription factors that regulate gene transcription. Since it takes time for genes to be transcribed into RNA and translated into protein, the effects of estrogens binding to these classical estrogen receptors is delayed. However, estrogens are also known to have effects that are too fast to be caused by regulation of gene transcription. In 2005, it was discovere ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Androgen
An androgen (from Greek ''andr-'', the stem of the word meaning ) is any natural or synthetic steroid hormone that regulates the development and maintenance of male characteristics in vertebrates by binding to androgen receptors. This includes the embryological development of the primary male sex organs, and the development of male secondary sex characteristics at puberty. Androgens are synthesized in the testes, the ovaries, and the adrenal glands. Androgens increase in both males and females during puberty. The major androgen in males is testosterone. Dihydrotestosterone (DHT) and androstenedione are of equal importance in male development. DHT ''in utero'' causes differentiation of the penis, scrotum and prostate. In adulthood, DHT contributes to balding, prostate growth, and sebaceous gland activity. Although androgens are commonly thought of only as male Sex steroids, sex hormones, females also have them, but at lower levels: they function in libido and sexual arousal. An ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sex Hormone
Sex hormones, also known as sex steroids, gonadocorticoids and gonadal steroids, are steroid hormones that interact with vertebrate steroid hormone receptors. The sex hormones include the androgens, estrogens, and progestogens. Their effects are mediated by slow genomic mechanisms through nuclear receptors as well as by fast nongenomic mechanisms through membrane-associated receptors and signaling cascades. Certain polypeptide hormones including the luteinizing hormone, follicle-stimulating hormone, and gonadotropin-releasing hormone – each associated with the gonadotropin axis – are usually not regarded as sex hormones, although they play major sex-related roles. Production Natural sex hormones are made by the gonads (ovaries or testicles), by adrenal glands, or by conversion from other sex steroids in other tissue such as liver or fat. Image:Steroidogenesis.svg Image:Biosinthesis of steroid hormones (simplified version).jpg Image:Biosinthesis of steroid hormones ( ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Estrogen
Estrogen (also spelled oestrogen in British English; see spelling differences) is a category of sex hormone responsible for the development and regulation of the female reproductive system and secondary sex characteristics. There are three major endogenous estrogens that have estrogenic hormonal activity: estrone (E1), estradiol (E2), and estriol (E3). Estradiol, an estrane, is the most potent and prevalent. Another estrogen called estetrol (E4) is produced only during pregnancy. Estrogens are synthesized in all vertebrates and some insects. Quantitatively, estrogens circulate at lower levels than androgens in both men and women. While estrogen levels are significantly lower in males than in females, estrogens nevertheless have important physiological roles in males. Like all steroid hormones, estrogens readily diffuse across the cell membrane. Once inside the cell, they bind to and activate estrogen receptors (ERs) which in turn modulate the expression of many ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Estriol
Estriol (E3), also spelled oestriol, is a steroid, a weak estrogen, and a minor female sex hormone. It is one of three major endogenous estrogens, the others being estradiol and estrone. Levels of estriol in women who are not pregnant are almost undetectable. However, during pregnancy, estriol is synthesized in very high quantities by the placenta and is the most produced estrogen in the body by far, although circulating levels of estriol are similar to those of other estrogens due to a relatively high rate of metabolism and excretion. Relative to estradiol, both estriol and estrone have far weaker activity as estrogens. In addition to its role as a natural hormone, estriol is used as a medication, for instance in menopausal hormone therapy; for information on estriol as a medication, see the estriol (medication) article. Biological activity Estriol is an estrogen, specifically an agonist of the estrogen receptors ERα and ERβ. It is a far less potent estrogen than is estra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
ERα
Estrogen receptor alpha (ERα), also known as NR3A1 (nuclear receptor subfamily 3, group A, member 1), is one of two main types of estrogen receptor, a nuclear receptor (mainly found as a chromatin-binding protein) that is activated by the sex hormone estrogen. In humans, ERα is encoded by the gene ''ESR1'' (EStrogen Receptor 1). Structure The estrogen receptor (ER) is a ligand-activated transcription factor composed of several domains important for hormone binding, DNA binding, and activation of transcription. Alternative splicing results in several ESR1 mRNA transcripts, which differ primarily in their 5-prime untranslated regions. The translated receptors show less variability. Ligands Agonists Non-selective * Endogenous estrogens (e.g., estradiol, estrone, estriol, estetrol) * Natural estrogens (e.g., conjugated equine estrogens) * Synthetic estrogens (e.g., ethinylestradiol, diethylstilbestrol) Selective Agonists of ERα selective over ERβ in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
ERβ
Estrogen receptor beta (ERβ) also known as NR3A2 (nuclear receptor subfamily 3, group A, member 2) is one of two main types of estrogen receptor—a nuclear receptor which is activated by the sex hormone estrogen. In humans ERβ is encoded by the ''ESR2'' gene. Function ERβ is a member of the family of estrogen receptors and the superfamily of nuclear receptor transcription factors. The gene product contains an N-terminus, N-terminal DNA binding domain and C-terminus, C-terminal ligand binding domain and is localized to the nucleus, cytoplasm, and mitochondria. Upon binding to 17-β-estradiol, estriol or related ligands, the encoded protein forms homo-dimers or hetero-dimers with estrogen receptor alpha, estrogen receptor α that interact with specific DNA sequences to activate transcription. Some isoforms dominantly inhibit the activity of other estrogen receptor family members. Several alternatively spliced transcript variants of this gene have been described, but the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potency (pharmacology)
In pharmacology, potency or biological potency is a measure of a drug's biological activity expressed in terms of the dose required to produce a pharmacological effect of given intensity. A highly potent drug (e.g., fentanyl, clonazepam, risperidone, benperidol, bumetanide) evokes a given response at low concentrations, while a drug of lower potency (e.g. morphine, alprazolam, ziprasidone, haloperidol, furosemide) evokes the same response only at higher concentrations. Higher potency does not necessarily mean greater effectiveness nor more side effects nor less side effects. Types of potency The International Union of Basic and Clinical Pharmacology (IUPHAR) has stated that "potency is an imprecise term that should always be further defined", and lists of types of potency as follows: Miscellaneous Lysergic acid diethylamide (LSD) is one of the most potent psychoactive drug A psychoactive drug, psychopharmaceutical, mind-altering drug, consciousness-altering drug, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Subcutaneous Injection
Subcutaneous administration is the insertion of medications beneath the skin either by injection or infusion. A subcutaneous injection is administered as a bolus (medicine), bolus into the subcutis, the layer of skin directly below the dermis and Epidermis (skin), epidermis, collectively referred to as the Cutis (anatomy), cutis. The instruments are usually a hypodermic needle and a syringe. Subcutaneous injections are highly effective in administering medications such as insulin, morphine, heroin, diacetylmorphine and goserelin. Subcutaneous administration may be List of medical abbreviations, abbreviated as SC, SQ, subcu, sub-Q, SubQ, or subcut. Subcut is the preferred abbreviation to reduce the risk of misunderstanding and potential errors. Subcutaneous tissue has few blood vessels and so drugs injected into it are intended for slow, sustained rates of absorption, often with some amount of depot injection, depot effect. Compared with other route of administration, routes of ad ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
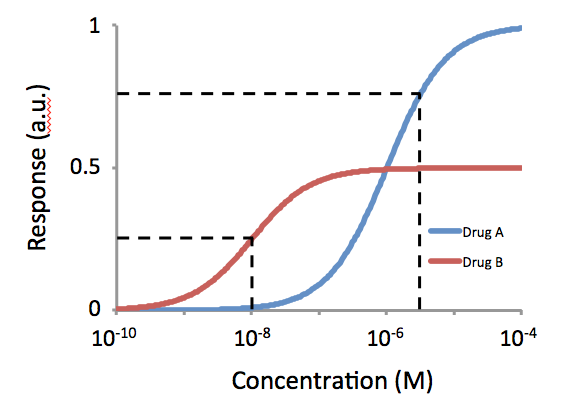
Relative Transactivational Capacities
Intrinsic activity (IA) and efficacy (Emax) refer to the relative ability of a drug-receptor complex to produce a maximum functional response. This must be distinguished from the affinity, which is a measure of the ability of the drug to bind to its molecular target, and the EC50, which is a measure of the potency of the drug and which is proportional to both efficacy and affinity. This use of the word "efficacy" was introduced by Stephenson (1956) to describe the way in which agonists vary in the response they produce, even when they occupy the same number of receptors. High efficacy agonists can produce the maximal response of the receptor system while occupying a relatively low proportion of the receptors in that system. There is a distinction between efficacy and intrinsic activity. Mechanism of efficacy Agonists of lower efficacy are not as efficient at producing a response from the drug-bound receptor, by stabilizing the active form of the drug-bound receptor. Therefore, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |