|
Disilene
Disilene is an inorganic compound with the chemical formula . The name ''disilene'', referring to the structure of a particular prototropic tautomer of the molecule. It is the simplest silene. Properties and bonding Disilene is a molecule with one Si=Si bond, and four equivalent Si-H bonds. Unlike ethylene, disilene is kinetically unstable with respect to tautomerisation. Disilene has two other tautomers, that are very close in energy: (μ2-''H'')disilene, and disilanylidene. Organodisilenes Disilenes bearing sterically bulky substituents are isolable and have been well characterized although they remain mainly of academic interest. The first stabilised disilene was tetramesityldisilene, (C6Me3H2)4Si2. The Si=Si distance in this molecule is 2.15 Å, about 10% shorter than a typical Si–Si single bond. The Si2C4 core is roughly planar. Such species are typically prepared by reduction of organosilicon halides: :2 R2SiCl2 + 4 Na → R2Si=SiR2 + 4 NaCl. An alternative synthesi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silenes
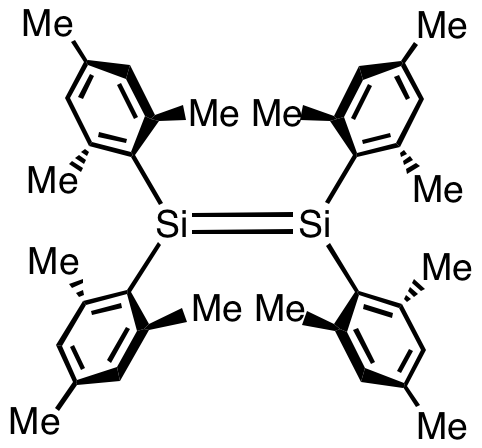
Silene, or disilalkenes,Philip P. Power "pi-Bonding and the Lone Pair Effect in Multiple Bonds between Heavier Main Group Elements" Chemical Reviews, 1999, 99, 3462. are silicon compounds that contain Si=Si double bonds. The parent silene is (the parent molecule) is disilene, . Structure The first transient disilene was reported in 1972 by D. N. Roark and Garry J. D. Peddle. Simple disilenes easily polymerize. To suppress this tendency, bulky substituents are used. Indeed the first isolable disilene, tetramesityldisilene, was described in 1981 by West, Fink, and Michl. It was prepared by UV-photolysis of the related cyclic trisilane: : 2 i(mesityl)2sub>3 → 3 (mesityl)2Si=Si(mesityl)2 Structure of tetramesityldisilene tetramesityldisilene is a yellow-orange solid. The Si=Si double bond lengths of disilenes vary between 2.14 and 2.29 Å and are nearly 5 to 10% shorter than the Si-Si single bond lengths of corresponding disilanes. A peculiarity of disilenes is the ''trans' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |