|
Carbon Fusion
The carbon-burning process or carbon fusion is a set of nuclear fusion reactions that take place in the cores of massive stars (at least 4 \beginM_\odot\end at birth) that combines carbon into other elements. It requires high temperatures (> 5×108 K or 50 keV) and densities (> 3×109 kg/m3). These figures for temperature and density are only a guide. More massive stars burn their nuclear fuel more quickly, since they have to offset greater gravitational forces to stay in (approximate) hydrostatic equilibrium. That generally means higher temperatures, although lower densities, than for less massive stars. To get the right figures for a particular mass, and a particular stage of evolution, it is necessary to use a numerical stellar model computed with computer algorithms. Such models are continually being refined based on nuclear physics experiments (which measure nuclear reaction rates) and astronomical observations (which include direct observation of mass loss, dete ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nuclear Fusion
Nuclear fusion is a nuclear reaction, reaction in which two or more atomic nuclei combine to form a larger nuclei, nuclei/neutrons, neutron by-products. The difference in mass between the reactants and products is manifested as either the release or absorption (electromagnetic radiation), absorption of energy. This difference in mass arises as a result of the difference in nuclear binding energy between the atomic nuclei before and after the fusion reaction. Nuclear fusion is the process that powers all active stars, via many Stellar nucleosynthesis, reaction pathways. Fusion processes require an extremely large Lawson criterion, triple product of temperature, density, and confinement time. These conditions occur only in Stellar core, stellar cores, advanced Nuclear weapon design, nuclear weapons, and are approached in List of fusion experiments, fusion power experiments. A nuclear fusion process that produces atomic nuclei lighter than nickel-62 is generally exothermic, due t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen
Hydrogen is a chemical element; it has chemical symbol, symbol H and atomic number 1. It is the lightest and abundance of the chemical elements, most abundant chemical element in the universe, constituting about 75% of all baryon, normal matter. Under standard conditions, hydrogen is a gas of diatomic molecules with the chemical formula, formula , called dihydrogen, or sometimes hydrogen gas, molecular hydrogen, or simply hydrogen. Dihydrogen is colorless, odorless, non-toxic, and highly combustible. Stars, including the Sun, mainly consist of hydrogen in a plasma state, while on Earth, hydrogen is found as the gas (dihydrogen) and in molecular forms, such as in water and organic compounds. The most common isotope of hydrogen (H) consists of one proton, one electron, and no neutrons. Hydrogen gas was first produced artificially in the 17th century by the reaction of acids with metals. Henry Cavendish, in 1766–1781, identified hydrogen gas as a distinct substance and discovere ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pair Production
Pair production is the creation of a subatomic particle and its antiparticle from a neutral boson. Examples include creating an electron and a positron, a muon and an antimuon, or a proton and an antiproton. Pair production often refers specifically to a photon creating an electron–positron pair near a nucleus. As energy must be conserved, for pair production to occur, the incoming energy of the photon must be above a threshold of at least the total rest mass energy of the two particles created. (As the electron is the lightest, hence, lowest mass/energy, elementary particle, it requires the least energetic photons of all possible pair-production processes.) Conservation of energy and momentum are the principal constraints on the process. All other conserved quantum numbers ( angular momentum, electric charge, lepton number) of the produced particles must sum to zero thus the created particles shall have opposite values of each other. For instance, if one partic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neutrino
A neutrino ( ; denoted by the Greek letter ) is an elementary particle that interacts via the weak interaction and gravity. The neutrino is so named because it is electrically neutral and because its rest mass is so small ('' -ino'') that it was long thought to be zero. The rest mass of the neutrino is much smaller than that of the other known elementary particles (excluding massless particles). The weak force has a very short range, the gravitational interaction is extremely weak due to the very small mass of the neutrino, and neutrinos do not participate in the electromagnetic interaction or the strong interaction. Consequently, neutrinos typically pass through normal matter unimpeded and with no detectable effect. Weak interactions create neutrinos in one of three leptonic flavors: # electron neutrino, # muon neutrino, # tau neutrino, Each flavor is associated with the correspondingly named charged lepton. Although neutrinos were long believed to be mas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Triple-alpha Process
The triple-alpha process is a set of nuclear fusion reactions by which three helium-4 nuclei (alpha particles) are transformed into carbon. In stars Helium accumulates in the cores of stars as a result of the proton–proton chain reaction and the carbon–nitrogen–oxygen cycle. Nuclear fusion reaction of two helium-4 nuclei produces beryllium-8, which is highly unstable, and decays back into smaller nuclei with a half-life of , unless within that time a third alpha particle fuses with the beryllium-8 nucleus to produce an excited resonance state of carbon-12, called the Hoyle state, which nearly always decays back into three alpha particles, but once in about 2421.3 times releases energy and changes into the stable base form of carbon-12. When a star runs out of hydrogen to fuse in its core, it begins to contract and heat up. If the central temperature rises to 108 K, six times hotter than the Sun's core, alpha particles can fuse fast enough to get past the beryllium-8 b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solar Mass
The solar mass () is a frequently used unit of mass in astronomy, equal to approximately . It is approximately equal to the mass of the Sun. It is often used to indicate the masses of other stars, as well as stellar clusters, nebulae, galaxies and black holes. More precisely, the mass of the Sun is The solar mass is about times the mass of Earth (), or times the mass of Jupiter (). History of measurement The value of the gravitational constant was first derived from measurements that were made by Henry Cavendish in 1798 with a torsion balance. The value he obtained differs by only 1% from the modern value, but was not as precise. The diurnal parallax of the Sun was accurately measured during the transits of Venus in 1761 and 1769, yielding a value of (9 arcseconds, compared to the present value of ). From the value of the diurnal parallax, one can determine the distance to the Sun from the geometry of Earth. The first known estimate of the solar mass was by ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
CNO Cycle
In astrophysics, the carbon–nitrogen–oxygen (CNO) cycle, sometimes called Bethe–Weizsäcker cycle, after Hans Albrecht Bethe and Carl Friedrich von Weizsäcker, is one of the two known sets of fusion reactions by which stars convert hydrogen to helium, the other being the proton–proton chain reaction (p–p cycle), which is more efficient at the Sun's core temperature. The CNO cycle is hypothesized to be dominant in stars that are more than 1.3 times as massive as the Sun. Unlike the proton-proton reaction, which consumes all its constituents, the CNO cycle is a catalytic cycle. In the CNO cycle, four protons fuse, using carbon, nitrogen, and oxygen isotopes as catalysts, each of which is consumed at one step of the CNO cycle, but re-generated in a later step. The end product is one alpha particle (a stable helium nucleus), two positrons, and two electron neutrinos. There are various alternative paths and catalysts involved in the CNO cycles, but all thes ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
S-process
The slow neutron-capture process, or ''s''-process, is a series of nuclear reactions, reactions in nuclear astrophysics that occur in stars, particularly asymptotic giant branch stars. The ''s''-process is responsible for the creation (nucleosynthesis) of approximately half the Atomic nucleus, atomic nuclei Heavy metal (chemical element), heavier than iron. In the ''s''-process, a seed nucleus undergoes neutron capture to form an isotope with one higher atomic mass. If the new isotope is stable nuclide, stable, a series of increases in mass can occur, but if it is unstable nucleus, unstable, then beta decay will occur, producing an element of the next higher atomic number. The process is ''slow'' (hence the name) in the sense that there is sufficient time for this radioactive decay to occur before another neutron is captured. A series of these reactions produces stable isotopes by moving along the valley of stability, valley of beta-decay stable isobars in the table of nuclides. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
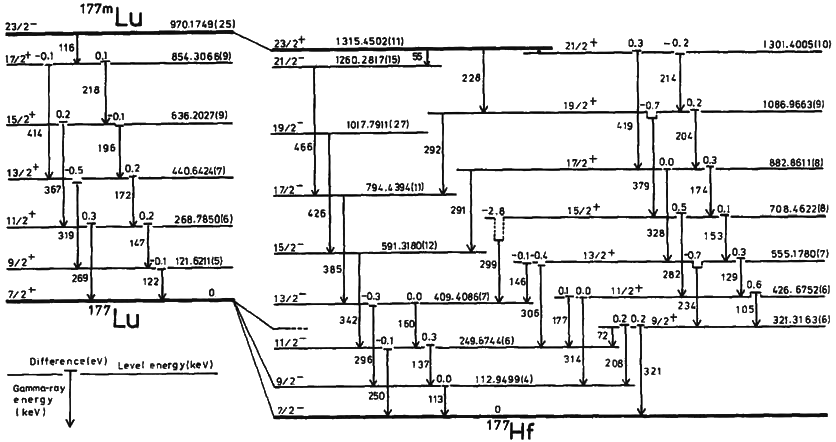
Nuclear Isomer
A nuclear isomer is a metastable state of an atomic nucleus, in which one or more nucleons (protons or neutrons) occupy excited state levels (higher energy levels). "Metastable" describes nuclei whose excited states have Half-life, half-lives of 10−9 seconds or longer, 100 to 1000 times longer than the half-lives of the excited nuclear states that decay with a "prompt" half life (ordinarily on the order of 10−12 seconds). Some references recommend seconds to distinguish the metastable half life from the normal "prompt" Induced gamma emission, gamma-emission half-life. Occasionally the half-lives are far longer than this and can last minutes, hours, or years. For example, the Isotopes of tantalum#Tantalum-180m, nuclear isomer survives so long (at least years) that it has never been observed to decay spontaneously. The half-life of a nuclear isomer can even exceed that of the ground state of the same nuclide, as shown by as well as isotopes of rhenium, , isotopes of iridium, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxygen-16
Oxygen-16 (symbol: 16O or ) is a nuclide. It is a stable isotope of oxygen, with 8 neutrons and 8 protons in its nucleus, and when not ionized, 8 electrons orbiting the nucleus. Oxygen-16 has a mass of . It is the most abundant isotope of oxygen and accounts for 99.757% of oxygen's natural abundance. The relative and absolute abundances of oxygen-16 are high because it is a principal product of stellar evolution and because it is a primordial isotope, meaning it can be made by stars that were initially made exclusively of hydrogen. Most oxygen-16 is synthesized at the end of the helium fusion process in stars; the triple-alpha process creates carbon-12, which captures an additional helium-4 to make oxygen-16. It is also created by the neon-burning process. Oxygen-16 is doubly magic. Solid samples (organic and inorganic) for oxygen-16 studies are usually stored in silver cups and measured with pyrolysis and mass spectrometry. Researchers need to avoid improper or prolo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |