|
Benzotriazole
Benzotriazole (BTA) is a heterocyclic compound with the chemical formula . It can be viewed as the fusion of a benzene and triazole rings. It is a white solid, although impure samples can appear tan. It is used as a corrosion inhibitor for copper. Structure and synthesis : Benzotriazole features two fused rings. It can in principle exist as tautomers, but X-ray crystallography establishes the depicted structure. The N=N and HN-N distances are 1.306 and 1.340 Å. Benzotriazole can be prepared by the monodiazotization of o-Phenylenediamine, ''o''-phenylenediamine using sodium nitrite and acetic acid.Robert A. Smiley "Phenylene- and Toluenediamines" in ''Ullmann's Encyclopedia of Industrial Chemistry'', 2002, Wiley-VCH, Weinheim. Reactions Acid-base behavior BTA is a weak Bronsted acid with a pKa = 8.2. It is a weak Brønsted base, as indicated by the low pKa 0.1 μg/L. One source of this pollution is their use as anti-icing/deicing agents in airports. Benzotriazo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tolyltriazole
Tolyltriazole is a mixture of isomers or congeners that differ from benzotriazole by the addition of one methyl group attached somewhere on the benzene ring. "The term tolyltriazole (CAS 29385-43-1) generally efers tothe commercial mixture composed of approximately equal amounts of 4- and 5-methylbenzotriazole, with small quantities of heir respective 7- and 6-methyl tautomers. Structure Synthesis and reactions Synthesis is much like that of benzotriazole Benzotriazole (BTA) is a heterocyclic compound with the chemical formula . It can be viewed as the fusion of a benzene and triazole rings. It is a white solid, although impure samples can appear tan. It is used as a corrosion inhibitor for copper. ..., but starting with methyl-''o''-phenylenediamine instead of o-Phenylenediamine">''o''-phenylenediamine. Isomers of methyl-''o''-phenylenediamine include 3-methyl-''o''-phenylenediamine, 4-methyl-''o''-phenylenediamine, and N-methyl-''o''-phenylenediamine (not involved here). A ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Corrosion Inhibitor
A corrosion inhibitor or anti-corrosive is a chemical compound added to a liquid or gas to decrease the corrosion rate of a metal that comes into contact with the fluid. The effectiveness of a corrosion inhibitor depends on fluid composition and dynamics. Corrosion inhibitors are common in industry, and also found in over-the-counter products, typically in spray form in combination with a lubricant and sometimes a penetrating oil. They may be added to water to prevent leaching of lead or copper from pipes. A common mechanism for inhibiting corrosion involves formation of a coating, often a passivation layer, which prevents access of the corrosive substance to the metal. Permanent treatments such as chrome plating are not generally considered inhibitors, however: corrosion inhibitors are additives to the fluids that surround the metal or related object. Types The nature of the corrosive inhibitor depends on (i) the material being protected, which are most commonly metal object ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
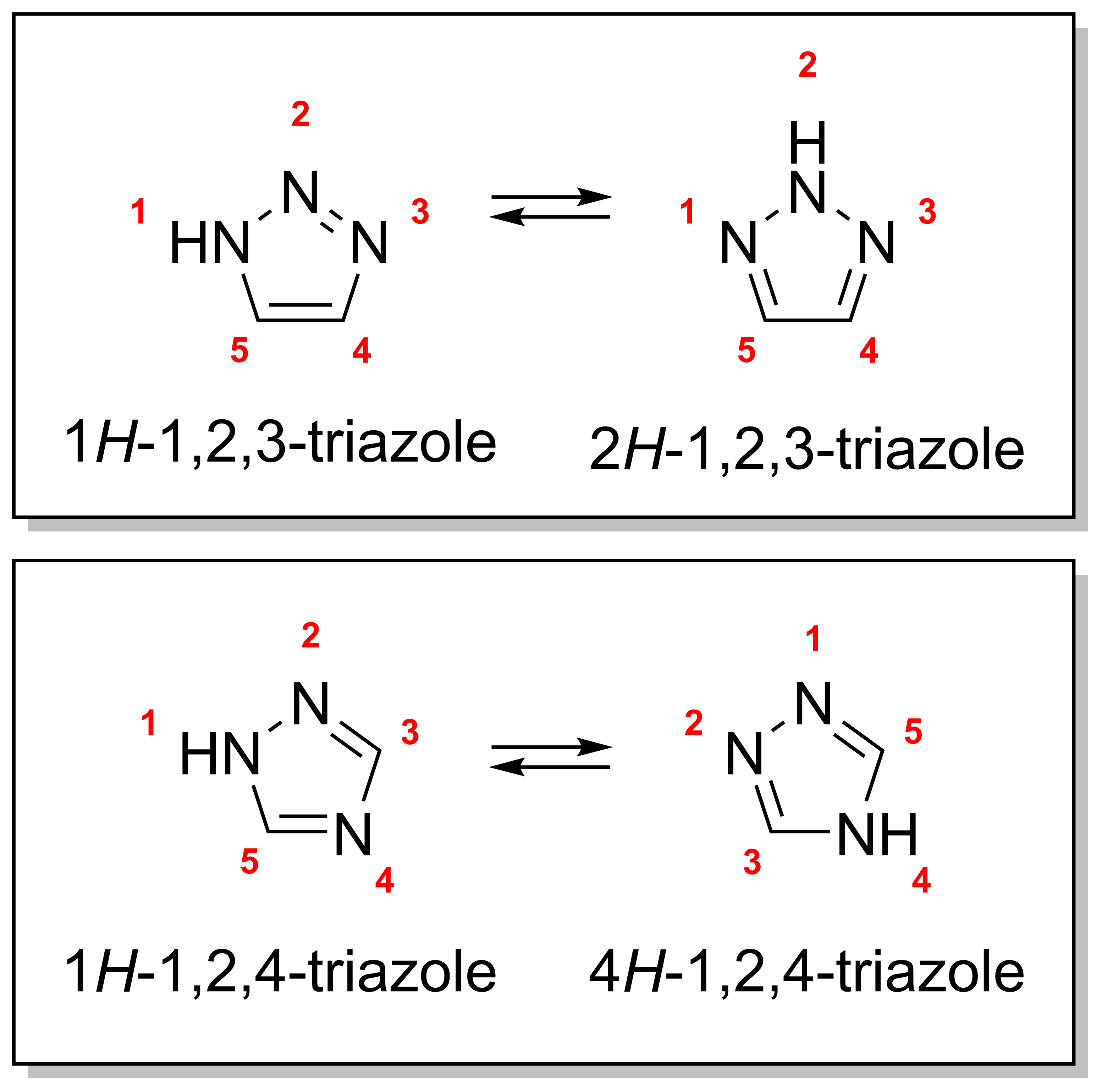
Triazole
A triazole is a heterocyclic compound featuring a five-membered ring of two carbon atoms and three nitrogen atoms with molecular formula C2H3N3. Triazoles exhibit substantial Isomer, isomerism, depending on the positioning of the nitrogen atoms within the ring. Many triazoles are versatile, biologically active compounds commonly used as fungicides and plant retardants. However, triazoles are also useful in bioorthogonal chemistry, because the large number of nitrogen atoms causes triazoles to react similar to Azide, azides. Lastly, the many free lone pairs in triazoles make them useful as coordination compounds, although not typically as Piano stool complex, haptic ligands. Isomerism There are four triazole isomers, which are conventionally divided into two pairs of Tautomer, tautomers. In the 1,2,3-Triazole, 1,2,3-triazoles, the three nitrogen atoms are adjacent; in the 1,2,4-Triazole, 1,2,4-triazoles, an interstitial carbon separates out one nitrogen atom. Each category ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Biphenylene
Biphenylene is an organic compound with the formula (C6H4)2. It is a pale, yellowish solid with a hay-like odor. Despite its unusual structure, it behaves like a traditional polycyclic aromatic hydrocarbon. Bonding Biphenylene is a polycyclic hydrocarbon, composed of two benzene rings joined by two bridging bonds (as opposed to a normal ring fusion), thus forming a 6-4-6 arene system. The resulting planar structure was one of the first π-electronic hydrocarbon systems discovered to show evidence of antiaromaticity. The spectral and chemical properties show the influence of the central nring, leading to considerable interest in the system in terms of its degree of lessened aromaticity. Questions of bond alternation and ring currents have been investigated repeatedly. Both X-ray diffraction and electron diffraction studies show a considerable alternation of bond lengths, with the bridging bonds between the benzenoid rings having the unusually great length of 1.524 Å. The s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
O-Phenylenediamine
''o''-Phenylenediamine (OPD) is an organic compound with the formula C6H4(NH2)2. This aromatic diamine is an important precursor to many heterocyclic compounds. OPD is a white compound although samples appear darker owing to oxidation by air. It is isomeric with ''m''-phenylenediamine and ''p''-phenylenediamine. Preparation Commonly, 2-nitrochlorobenzene is treated with ammonia to generate 2-nitroaniline, whose nitro group is then reduced: :ClC6H4NO2 + 2 NH3 → H2NC6H4NO2 + NH4Cl :H2NC6H4NO2 + 3 H2 → H2NC6H4NH2 + 2 H2O In the laboratory, the reduction of the nitroaniline is effected with zinc powder in ethanol, followed by purification of the diamine as the hydrochloride salt. Darkened impure samples can be purified by treatment of its aqueous solution with sodium dithionite and activated carbon. Reactions and uses ''o''-Phenylenediamine condenses with ketones and aldehydes to give rise to various valuable products. Its reactions with formic acids to produce benz ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzimidazole
Benzimidazole is a heterocyclic aromatic organic compound. This bicyclic compound may be viewed as fused rings of the aromatic compounds benzene and imidazole. It is a white solid that appears in form of tabular crystals. Preparation Benzimidazole was discovered during research on vitamin B12. The benzimidazole nucleus was found to be a stable platform on which drugs could be developed. Benzimidazole is produced by Condensation reaction, condensation of o-phenylenediamine with formic acid, or the equivalent trimethyl orthoformate: :C6H4(NH2)2 + HC(OCH3)3 → C6H4N(NH)CH + 3 CH3OH 2-Substituted derivatives are obtained when the condensation is conducted with aldehydes in place of formic acid, followed by oxidation. Reactions Benzimidazole is a Base (chemistry), base: :C6H4N(NH)CH + H+ → [C6H4(NH)2CH]+ It can also be deprotonated with stronger bases: :C6H4N(NH)CH + LiH → Li [C6H4N2CH] + H2 The imine can be alkylated and also serves as a ligand in coordinati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lead(IV) Acetate
Lead(IV) acetate or lead tetraacetate is an metalorganic compound with chemical formula , often abbreviated as , where Ac is acyl. It is a colorless solid that is soluble in nonpolar, organic solvents, indicating that it is not a salt. It is degraded by moisture and is typically stored with additional acetic acid. The compound is used in organic synthesis. Structure In the solid state the lead(IV) centers are coordinated by four acetate ions, which are bidentate, each coordinating via two oxygen atoms. The lead atom is 8 coordinate and the O atoms form a flattened trigonal dodecahedron. Preparation It is typically prepared by treating of red lead with acetic acid and acetic anhydride (), which absorbs water. The net reaction is shown: : The remaining lead(II) acetate can be partially oxidized to the tetraacetate by Cl2, with a by-product: : Reagent in organic chemistry Lead tetraacetate is a strong oxidizing agent, a source of acetyloxy groups, and a general reagent for t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acylsilane
Acylsilanes are a group of chemical compounds sharing a common functional group with the general structure RC(O)-SiR3. Synthesis Acylsilanes can be synthesized by treating acyl anion equivalents with silyl halides (typically trimethylsilyl chloride, tmsCl). Silylation of 2-lithio- 1,3-dithiane, followed by hydrolysis of the dithioacetal group with mercury(II) chloride. Analogous methods has also been used to produce acylgermanes. Several approaches to acylsilanes start with carboxylic acid derivatives. Esters undergo reductive silylation en route to acylsilanes: : : Tertiary amides react with silyl lithium reagents: : Acid chlorides are converted using hexamethyldisilane: : Some acyl silanes are prepared by oxidation of a suitable silanes. Reactions Acylsilanes are starting compounds in the Brook rearrangement with vinyl lithium compounds to silyl enol ethers. Acyl silanes and aryl bromides are coupling partners in Pd-catalyzed cross coupling reactions: Further rea ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydroxylamine-O-sulfonic Acid
Hydroxylamine-''O''-sulfonic acid (HOSA) or aminosulfuric acid is the inorganic compound with molecular formula H3NO4S that is formed by the sulfonation of hydroxylamine with oleum. It is a white, water-soluble and hygroscopic, solid, commonly represented by the condensed structural formula H2NOSO3H, though it actually exists as a zwitterion and thus is more accurately represented as +H3NOSO3−. It is used as a reagent for the introduction of amine groups (–NH2), for the conversion of aldehydes into nitriles and alicyclic compound, alicyclic ketones into lactams (cyclic amides), and for the synthesis of variety of nitrogen-containing heterocycles. Preparation According to a laboratory procedure hydroxylamine-''O''-sulfonic acid can be prepared by treating hydroxylamine sulfate with fuming sulfuric acid (oleum). The industrial process is similar. :(NH3OH)2SO4 + 2SO3 → 2H2NOSO3H + H2SO4 The sulfonation of hydroxylamine can also be effected with chlorosulfonic acid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimer (chemistry)
In chemistry, dimerization is the process of joining two identical or similar Molecular entity, molecular entities by Chemical bond, bonds. The resulting bonds can be either strong or weak. Many symmetrical chemical species are described as dimers, even when the monomer is unknown or highly unstable. The term ''homodimer'' is used when the two subunits are identical (e.g. A–A) and ''heterodimer'' when they are not (e.g. A–B). The reverse of dimerization is often called Dissociation (chemistry), dissociation. When two oppositely-charged ions associate into dimers, they are referred to as ''Bjerrum pairs'', after Danish chemist Niels Bjerrum. Noncovalent dimers Anhydrous carboxylic acids form dimers by hydrogen bonding of the acidic hydrogen and the carbonyl oxygen. For example, acetic acid forms a dimer in the gas phase, where the monomer units are held together by hydrogen bonds. Many OH-containing molecules form dimers, e.g. the water dimer. Dimers that form based on w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |