|
Aziridine
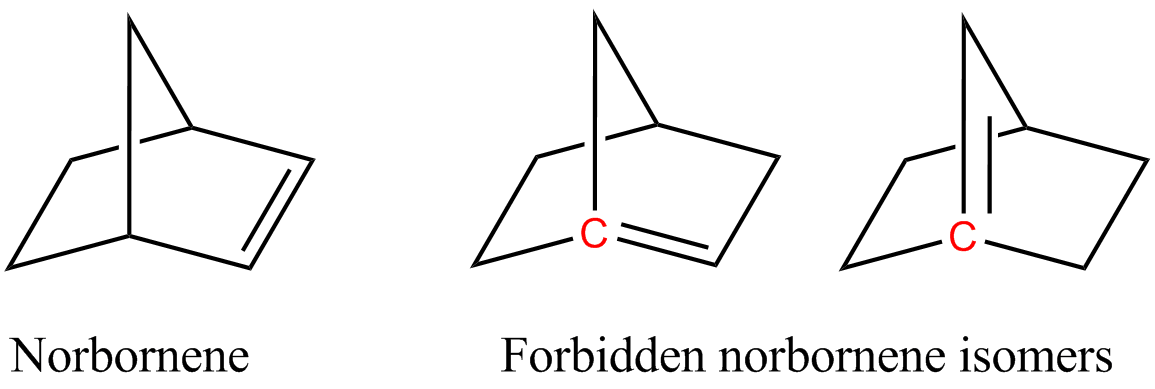
Aziridine is an organic compound consisting of the three-membered heterocycle . It is a colorless, toxic, volatile liquid that is of significant practical interest. Aziridine was discovered in 1888 by the chemist Siegmund Gabriel. Its derivatives, also referred to as aziridines, are of broader interest in medicinal chemistry. Structure The bond angles in aziridine are approximately 60°, considerably less than the normal hydrocarbon bond angle of 109.5°, which results in angle strain as in the comparable cyclopropane and ethylene oxide molecules. A banana bond model explains bonding in such compounds. Aziridine is less basic than acyclic aliphatic amines, with a pKa of 7.9 for the conjugate acid, due to increased s character of the nitrogen free electron pair. Angle strain in aziridine also increases the barrier to nitrogen inversion. This barrier height permits the isolation of separate ''invertomers'', for example the ''cis'' and ''trans'' invertomers of ''N''-chlo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aziridines
220 px, chemotherapeutic agent by virtue of its antitumour activity. Aziridines are organic compounds containing the aziridine functional group, a three-membered heterocycle with one amine (-NR-) and two methylene bridges (--). The parent compound is aziridine (or ethylene imine), with molecular formula . Several drugs feature aziridine rings, including mitomycin C, porfiromycin, and azinomycin B (carzinophilin). Structure The bond angles in aziridine are approximately 60°, considerably less than the normal hydrocarbon bond angle of 109.5°, which results in angle strain as in the comparable cyclopropane and ethylene oxide molecules. A banana bond model explains bonding in such compounds. Aziridine is less basic than acyclic aliphatic amines, with a pKa of 7.9 for the conjugate acid, due to increased s character of the nitrogen free electron pair. Angle strain in aziridine also increases the barrier to nitrogen inversion. This barrier height permits the isolation of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Wenker Synthesis
The Wenker synthesis is an organic reaction converting a beta amino alcohol to an aziridine with the help of sulfuric acid. It is used industrially for the synthesis of aziridine itself. The original Wenker synthesis of aziridine itself takes place in two steps. In the first step ethanolamine is reacted with sulfuric acid at high temperatures (250 °C) to form the sulfate monoester. This salt is then reacted with sodium hydroxide in the second step forming aziridine. The base abstracts an amine proton enabling it to displace the sulfate group. A modification of this reaction involving lower reaction temperatures (140–180 °C) and therefore reduced charring increases the yield of the intermediate. The Wenker synthesis protocol using ''trans''-2-aminocyclooctanol, available from reaction of ammonia with the epoxide of cyclooctene, gives a mixture of cyclooctenimine (the Wenker aziridine product) and cyclooctanone (a competing Hofmann elimination Hofmann elimination is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polyethylenimine
Polyethylenimine (PEI) or polyaziridine is a polymer with repeating units composed of the amine group and two carbon aliphatic ''CHCH'' spacers. Linear polyethyleneimines contain all secondary amines, in contrast to branched PEIs which contain primary, secondary and tertiary amino groups. Totally branched, dendrimeric forms were also reported. PEI is produced on an industrial scale and finds many applications usually derived from its polycationic character. Properties The linear PEI is a semi-crystalline solid at room temperature while branched PEI is a fully amorphous polymer existing as a liquid at all molecular weights. Linear polyethyleneimine is soluble in hot water, at low pH, in methanol, ethanol, or chloroform. It is insoluble in cold water, benzene, ethyl ether, and acetone. Linear polyethyleneimine has a melting point of around 67 °C. Both linear and branched polyethyleneimine can be stored at room temperature. Linear polyethyleneimine is able to form cryogels up ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Borirane
Borirane is a heterocyclic organic compound with the formula C2 H4 BH. This colourless, flammable gas is the simplest borirane, a three-membered ring consisting of two carbon and one boron atom. It can be viewed as a structural analog of aziridine, with boron replacing the nitrogen atom of aziridine. Borirane is isomer In chemistry, isomers are molecules or polyatomic ions with identical molecular formulae – that is, same number of atoms of each element – but distinct arrangements of atoms in space. Isomerism is existence or possibility of isomers. Is ...ic with ethylideneborane. This compound has five isomers. References {{Reflist Boron heterocycles Gases Three-membered rings ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ring Strain
In organic chemistry, ring strain is a type of instability that exists when bonds in a molecule form angles that are abnormal. Strain is most commonly discussed for small rings such as cyclopropanes and cyclobutanes, whose internal angles are substantially smaller than the idealized value of approximately 109°. Because of their high strain, the heat of combustion for these small rings is elevated. Ring strain results from a combination of angle strain, conformational strain or Pitzer strain (torsional eclipsing interactions), and transannular strain, also known as van der Waals strain or Prelog strain. The simplest examples of angle strain are small cycloalkanes such as cyclopropane and cyclobutane. Ring strain energy can be attributed to the energy required for the distortion of bond and bond angles in order to close a ring. Ring strain energy is believed to be the cause of accelerated rates in altering ring reactions. Its interactions with traditional bond energies ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Angle Strain
In organic chemistry, ring strain is a type of instability that exists when bonds in a molecule form angles that are abnormal. Strain is most commonly discussed for small rings such as cyclopropanes and cyclobutanes, whose internal angles are substantially smaller than the idealized value of approximately 109°. Because of their high strain, the heat of combustion for these small rings is elevated. Ring strain results from a combination of angle strain, conformational strain or Pitzer strain (torsional eclipsing interactions), and transannular strain, also known as van der Waals strain or Prelog strain. The simplest examples of angle strain are small cycloalkanes such as cyclopropane and cyclobutane. Ring strain energy can be attributed to the energy required for the distortion of bond and bond angles in order to close a ring. Ring strain energy is believed to be the cause of accelerated rates in altering ring reactions. Its interactions with traditional bond energies ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heterocycle
A heterocyclic compound or ring structure is a cyclic compound that has atoms of at least two different elements as members of its ring(s). Heterocyclic chemistry is the branch of organic chemistry dealing with the synthesis, properties, and applications of these heterocycles. Examples of heterocyclic compounds include all of the nucleic acids, the majority of drugs, most biomass (cellulose and related materials), and many natural and synthetic dyes. More than half of known compounds are heterocycles. 59% of US FDA-approved drugs contain nitrogen heterocycles. Classification The study of heterocyclic chemistry focuses especially on unsaturated derivatives, and the preponderance of work and applications involves unstrained 5- and 6-membered rings. Included are pyridine, thiophene, pyrrole, and furan. Another large class of heterocycles refers to those fused to benzene rings. For example, the fused benzene derivatives of pyridine, thiophene, pyrrole, and furan are quino ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrogen Inversion
In chemistry, pyramidal inversion (also umbrella inversion) is a fluxional process in compounds with a pyramidal molecule, such as ammonia (NH3) "turns inside out". It is a rapid oscillation of the atom and substituents, the molecule or ion passing through a planar transition state. For a compound that would otherwise be chiral due to a stereocenter, pyramidal inversion allows its enantiomers to racemize. The general phenomenon of pyramidal inversion applies to many types of molecules, including carbanions, amines, phosphines, arsines, stibines, and sulfoxides. Energy barrier The identity of the inverting atom has a dominating influence on the barrier. Inversion of ammonia is rapid at room temperature. In contrast, phosphine (PH3) inverts very slowly at room temperature (energy barrier: 132 kJ/mol). Consequently, amines of the type RR′R"N usually are not optically stable (enantiomers racemize rapidly at room temperature), but ''P''-chiral phosphines are. Appropriately ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Banana Bond
In organic chemistry, a bent bond, also known as a banana bond, is a type of covalent chemical bond with a geometry somewhat reminiscent of a banana. The term itself is a general representation of electron density or configuration resembling a similar "bent" structure within small ring molecules, such as cyclopropane (C3H6) or as a representation of double or triple bonds within a compound that is an alternative to the sigma and pi bond model. Small cyclic molecules Bent bonds are a special type of chemical bonding in which the ordinary hybridization state of two atoms making up a chemical bond are modified with increased or decreased s-orbital character in order to accommodate a particular molecular geometry. Bent bonds are found in strained organic compounds such as cyclopropane, oxirane and aziridine. In these compounds, it is not possible for the carbon atoms to assume the 109.5° bond angles with standard sp3 hybridization. Increasing the p-character to sp5 (i.e. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cis Isomer
Cis or cis- may refer to: Places * Cis, Trentino, in Italy * In Poland: ** Cis, Świętokrzyskie Voivodeship, south-central ** Cis, Warmian-Masurian Voivodeship, north Math, science and biology * cis (mathematics) (cis(''θ'')), a trigonometric mathematical function related to Euler's formula * ''Cis'' (beetle), genus * Cis–trans isomerism, in chemistry * cis-regulatory element, regions of non-coding DNA which regulate the transcription of nearby genes Other uses * Cisgender, in contrast with transgender * C♯ (musical note), known as cis See also * CIS (other) * * Ciss (other) Ciss (pronounced SIHS) is a Senegalese surname. Notable people with the surname include: * Amadou Ciss (born 1999), Senegalese footballer who plays for Fortuna Sittard * Elhadji Ciss (born 1994), Senegalese footballer who plays for Sion * Khadij ... * Csi (other) {{disambiguation, geo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aminoethanol
Aminoethanol may refer to: * 1-Aminoethanol * Ethanolamine (2-aminoethanol, ETA, or MEA) {{Short pages monitor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trans Isomer
Trans- is a Latin prefix meaning "across", "beyond", or "on the other side of". Used alone, trans may refer to: Arts, entertainment, and media * Trans (festival), a former festival in Belfast, Northern Ireland, United Kingdom * ''Trans'' (film), a 1998 American film * Trans Corp, an Indonesian business unit of CT Corp in the fields of media, lifestyle, and entertainment ** Trans Media, a media subsidiary of Trans Corp *** Trans TV, an Indonesian television network *** Trans7, an Indonesian television network Literature * '' Trans: Gender and Race in an Age of Unsettled Identities'', a 2016 book by Rogers Brubaker * '' Trans: When Ideology Meets Reality'', a 2021 book by Helen Joyce Music * ''Trans'' (album), by Neil Young * ''Trans'' (Stockhausen), a 1971 orchestral composition Places * Trans, Mayenne, France, a commune * Trans, Switzerland, a village Science and technology * Trans effect in inorganic chemistry, the increased lability of ligands that are trans to c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |