|
Antifebrin
Acetanilide is an odourless solid chemical of leaf or flake-like appearance. It is also known as ''N''-phenylacetamide, acetanil, or acetanilid, and was formerly known by the trade name Antifebrin. Preparation and properties Acetanilide can be produced by reacting acetic anhydride with aniline: :C6H5NH2 + (CH3CO)2O → C6H5NHCOCH3 + CH3COOH The preparation used to be a traditional experiment in introductory organic chemistry lab classes, but it has now been widely replaced by the preparation of either paracetamol or aspirin, both of which teach the same practical techniques (especially recrystallization of the product) but which avoid the use of aniline, a suspected carcinogen. Acetanilide is slightly soluble in water, and stable under most conditions. Pure crystals are plate shaped and appear colorless, white, or in between. Applications Acetanilide is used as an inhibitor of hydrogen peroxide decomposition and is used to stabilize cellulose ester varnishes. It has al ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Paracetamol
Paracetamol, also known as acetaminophen, is a medication used to treat fever and mild to moderate pain. Common brand names include Tylenol and Panadol. At a standard dose, paracetamol only slightly decreases body temperature; it is inferior to ibuprofen in that respect, and the benefits of its use for fever are unclear. Paracetamol may relieve pain in acute mild migraine but only slightly in episodic tension headache. However, the aspirin/paracetamol/caffeine combination helps with both conditions where the pain is mild and is recommended as a first-line treatment for them. Paracetamol is effective for post-surgical pain, but it is inferior to ibuprofen. The paracetamol/ibuprofen combination provides further increase in potency and is superior to either drug alone. The pain relief paracetamol provides in osteoarthritis is small and clinically insignificant. The evidence in its favor for the use in low back pain, cancer pain, and neuropathic pain is insufficient. In the sho ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetanilide Crystals
Acetanilide is an odourless solid chemical of leaf or flake-like appearance. It is also known as ''N''-phenylacetamide, acetanil, or acetanilid, and was formerly known by the trade name Antifebrin. Preparation and properties Acetanilide can be produced by reacting acetic anhydride with aniline: :C6H5NH2 + (CH3CO)2O → C6H5NHCOCH3 + CH3COOH The preparation used to be a traditional experiment in introductory organic chemistry lab classes, but it has now been widely replaced by the preparation of either paracetamol or aspirin, both of which teach the same practical techniques (especially recrystallization of the product) but which avoid the use of aniline, a suspected carcinogen. Acetanilide is slightly soluble in water, and stable under most conditions. Pure crystals are plate shaped and appear colorless, white, or in between. Applications Acetanilide is used as an inhibitor of hydrogen peroxide decomposition and is used to stabilize cellulose ester varnishes. It has also ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Royal Society Of Chemistry
The Royal Society of Chemistry (RSC) is a learned society (professional association) in the United Kingdom with the goal of "advancing the chemistry, chemical sciences". It was formed in 1980 from the amalgamation of the Chemical Society, the Royal Institute of Chemistry, the Faraday Society, and the Society for Analytical Chemistry with a new Royal Charter and the dual role of learned society and professional body. At its inception, the Society had a combined membership of 34,000 in the UK and a further 8,000 abroad. The headquarters of the Society are at Burlington House, Piccadilly, London. It also has offices in Thomas Graham House in Cambridge (named after Thomas Graham (chemist), Thomas Graham, the first president of the Chemical Society) where ''RSC Publishing'' is based. The Society has offices in the United States, on the campuses of The University of Pennsylvania and Drexel University, at the University City Science Center in Philadelphia, Pennsylvania, in both Beijing a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Peroxide
Hydrogen peroxide is a chemical compound with the formula . In its pure form, it is a very pale blue liquid that is slightly more viscous than water. It is used as an oxidizer, bleaching agent, and antiseptic, usually as a dilute solution (3%–6% by weight) in water for consumer use, and in higher concentrations for industrial use. Concentrated hydrogen peroxide, or " high-test peroxide", decomposes explosively when heated and has been used as a propellant in rocketry. Hydrogen peroxide is a reactive oxygen species and the simplest peroxide, a compound having an oxygen–oxygen single bond. It decomposes slowly when exposed to light, and rapidly in the presence of organic or reactive compounds. It is typically stored with a stabilizer in a weakly acidic solution in a dark bottle to block light. Hydrogen peroxide is found in biological systems including the human body. Enzymes that use or decompose hydrogen peroxide are classified as peroxidases. Properties The boiling poi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phenacetin
Phenacetin (acetophenetidin, ''N''-(4-ethoxyphenyl)acetamide) is a pain-relieving and fever-reducing drug, which was widely used following its introduction in 1887. It was withdrawn from medicinal use as dangerous from the 1970s (e.g., withdrawn in Canada in 1973, and by the U.S. Food and Drug Administration in 1983). History Phenacetin was introduced in 1887 in Elberfeld, Germany by German company Bayer, and was used principally as an analgesic; it was one of the first synthetic fever reducers to go on the market. It is also known historically to be one of the first non-opioid analgesics without anti-inflammatory properties. Although paracetamol was produced earlier, a historical accident saw it ignored after Joseph von Mering's assessment. Prior to World War One, Britain imported phenacetin from Germany. During the war, a team including Jocelyn Field Thorpe and Martha Annie Whiteley developed a synthesis in Britain. Known mechanism of action Phenacetin's analgesic effects ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methemoglobinemia
Methemoglobinemia, or methaemoglobinaemia, is a condition of elevated methemoglobin in the blood. Symptoms may include headache, dizziness, shortness of breath, nausea, poor muscle coordination, and blue-colored skin (cyanosis). Complications may include seizures and heart arrhythmias. Methemoglobinemia can be due to certain medications, chemicals, or food or it can be inherited from a person's parents. Substances involved may include benzocaine, nitrates, or dapsone. The underlying mechanism involves some of the iron in hemoglobin being converted from the ferrous e2+to the ferric e3+form. The diagnosis is often suspected based on symptoms and a low blood oxygen that does not improve with oxygen therapy. Diagnosis is confirmed by a blood gas. Treatment is generally with oxygen therapy and methylene blue. Other treatments may include vitamin C, exchange transfusion, and hyperbaric oxygen therapy. Outcomes are generally good with treatment. Methemoglobinemia is relatively u ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyanosis
Cyanosis is the change of body tissue color to a bluish-purple hue as a result of having decreased amounts of oxygen bound to the hemoglobin in the red blood cells of the capillary bed. Body tissues that show cyanosis are usually in locations where the skin is thinner, including the mucous membranes, lips, nail beds, and ear lobes. Some medications containing amiodarone or silver, Mongolian spots, large birth marks, and the consumption of food products with blue or purple dyes can also result in the bluish skin tissue discoloration and may be mistaken for cyanosis. Cyanosis is further classified into central cyanosis vs. peripheral cyanosis. Pathophysiology The mechanism behind cyanosis is different depending on whether it is central or peripheral. Central cyanosis Central cyanosis is caused by a decrease in arterial oxygen saturation (SaO2) and begins to show once the concentration of deoxyhemoglobin in the blood reaches a concentration of ≥ 5.0 g/dL (≥ 3.1 mmol/L ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Antipyretic
An antipyretic (, from ''anti-'' 'against' and ' 'feverish') is a substance that reduces fever. Antipyretics cause the hypothalamus to override a prostaglandin-induced increase in temperature. The body then works to lower the temperature, which results in a reduction in fever. Most antipyretic medications have other purposes. The most common antipyretics in the US are usually ibuprofen and aspirin, which are nonsteroidal anti-inflammatory drugs (NSAIDs) used primarily as anti-inflammatories and analgesics (pain relievers), but which also have antipyretic properties; and paracetamol (acetaminophen), an analgesic without anti-inflammatory properties. There is some debate over the appropriate use of such medications, since fever is part of the body's immune response to infection. A study published by the Royal Society claims that fever suppression causes at least 1% more influenza deaths in the United States, or 700 extra deaths per year. Non-pharmacological treatment Bathing or ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Analgesic
An analgesic drug, also called simply an analgesic (American English), analgaesic (British English), pain reliever, or painkiller, is any member of the group of drugs used to achieve relief from pain (that is, analgesia or pain management). It is typically used to induce cooperation with a medical procedure. Analgesics are conceptually distinct from anesthetics, which temporarily reduce, and in some instances eliminate, sensation, although analgesia and anesthesia are neurophysiologically overlapping and thus various drugs have both analgesic and anesthetic effects. Analgesic choice is also determined by the type of pain: For neuropathic pain, traditional analgesics are less effective, and there is often benefit from classes of drugs that are not normally considered analgesics, such as tricyclic antidepressants and anticonvulsants. Various analgesics, such as many NSAIDs, are available over the counter in most countries, whereas various others are prescription drugs owing ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Photographic Developer
In the processing of photographic films, plates or papers, the photographic developer (or just developer) is one or more chemicals that convert the latent image to a visible image. Developing agents achieve this conversion by reducing the silver halides, which are pale-colored, into silver metal, which is black (when a fine particle).Karlheinz Keller et al. ''Photography'' in ''Ullmann's Encyclopedia of Industrial Chemistry'', 2005, Wiley-VCH, Weinheim. The conversion occurs within the gelatine matrix. The special feature of photography is that the developer acts more quickly on those particles of silver halides that have been exposed to light. Paper left in developer will eventually reduce all the silver halides and turn black. Generally, the longer a developer is allowed to work, the darker the image. Chemical composition of developers The developer typically consists of a mixture of chemical compounds prepared as an aqueous solution. For black-and-white photography, three ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
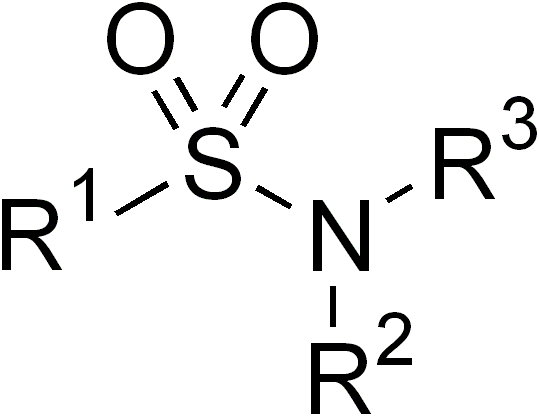
Sulfonamide (medicine)
Sulfonamide is a functional group (a part of a molecule) that is the basis of several groups of drugs, which are called sulphonamides, sulfa drugs or sulpha drugs. The original antibacterial sulfonamides are synthetic (nonantibiotic) antimicrobial agents that contain the sulfonamide group. Some sulfonamides are also devoid of antibacterial activity, e.g., the anticonvulsant sultiame. The sulfonylureas and thiazide diuretics are newer drug groups based upon the antibacterial sulfonamides. Allergies to sulfonamides are common. The overall incidence of adverse drug reactions to sulfa antibiotics is approximately 3%, close to penicillin; hence medications containing sulfonamides are prescribed carefully. Sulfonamide drugs were the first broadly effective antibacterials to be used systemically, and paved the way for the antibiotic revolution in medicine. Function In bacteria, antibacterial sulfonamides act as competitive inhibitors of the enzyme dihydropteroate synthase (DHP ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Camphor
Camphor () is a waxy, colorless solid with a strong aroma. It is classified as a terpenoid and a cyclic ketone. It is found in the wood of the camphor laurel ('' Cinnamomum camphora''), a large evergreen tree found in East Asia; and in the kapur tree ( ''Dryobalanops'' sp.), a tall timber tree from South East Asia. It also occurs in some other related trees in the laurel family, notably '' Ocotea usambarensis''. Rosemary leaves (''Rosmarinus officinalis'') contain 0.05 to 0.5% camphor, while camphorweed (''Heterotheca'') contains some 5%. A major source of camphor in Asia is camphor basil (the parent of African blue basil). Camphor can also be synthetically produced from oil of turpentine. The compound is chiral, existing in two possible enantiomers as shown in the structural diagrams. The structure on the left is the naturally occurring (+)-camphor ((1''R'',4''R'')-bornan-2-one), while its mirror image shown on the right is the (−)-camphor ((1''S'',4''S'')-bornan-2-one). ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |




2.jpg)