|
9-Anthracenecarboxaldehyde
Anthracene-9-carbaldehyde is the most common monoaldehyde derivative of anthracene. It is a yellow solid that is soluble in common organic solvents. It is prepared by Vilsmeier formylation of anthracene. The compound is also used as a building block for supramolecular assemblies. Hydrogenation Hydrogenation is a chemical reaction between molecular hydrogen (H2) and another compound or element, usually in the presence of a Catalysis, catalyst such as nickel, palladium or platinum. The process is commonly employed to redox, reduce or S ... of 9-anthracenecarboxaldehyde gives 9-anthracenemethanol. References Aromatic aldehydes Anthracenes {{Hydrocarbon-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Anthracene
Anthracene is a solid polycyclic aromatic hydrocarbon (PAH) of formula C14H10, consisting of three fused benzene rings. It is a component of coal tar. Anthracene is used in the Economic production, production of the red dye alizarin and other dyes. Anthracene is colorless but exhibits a blue (400–500 nm peak) fluorescence under ultraviolet radiation. Occurrence and production Coal tar, which contains around 1.5% anthracene, remains a major source of this material. Common impurities are phenanthrene and carbazole. The mineral form of anthracene is called freitalite and is related to a coal deposit. A classic laboratory method for the preparation of anthracene is by cyclodehydration of o-methyl- or o-methylene-substituted diarylketones in the so-called Elbs reaction, for example from ''o''-tolyl phenyl ketone. Reactions Reduction Reduction of anthracene with alkali metals yields the deeply colored radical anion salts M+[anthracene]− (M = Li, Na, K). Hydrogenation gives 9 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
9-Anthracenemethanol
9-Anthracenemethanol is the derivative of anthracene with a hydroxymethyl group (CH2OH) attached to the 9-position. It is a colorless solid that is soluble in ordinary organic solvents. The compound can be prepared by hydrogenation of 9-anthracenecarboxaldehyde Anthracene-9-carbaldehyde is the most common monoaldehyde derivative of anthracene. It is a yellow solid that is soluble in common organic solvents. It is prepared by Vilsmeier formylation of anthracene. The compound is also used as a building b .... It is a versatile precursor to supramolecular assemblies. References {{DEFAULTSORT:Anthracenemethanol, 9- Alcohols Anthracenes ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
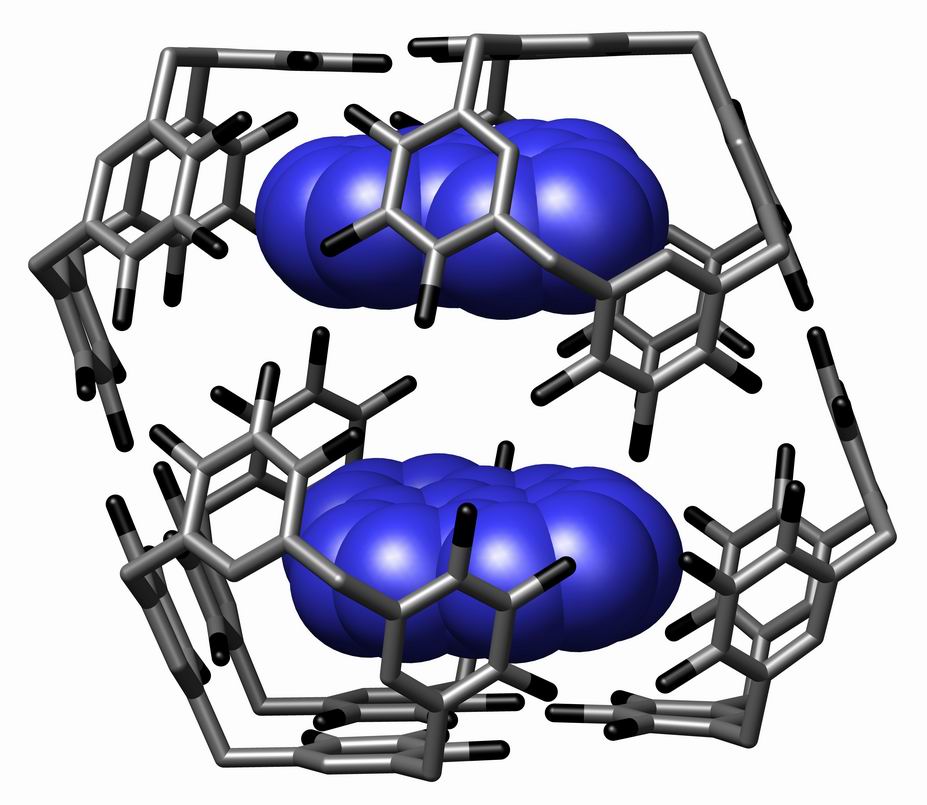
Supramolecular Assemblies
In chemistry, a supramolecular assembly is a complex of molecules held together by noncovalent bonds. While a supramolecular assembly can be simply composed of two molecules (e.g., a DNA double helix or an inclusion compound), or a defined number of stoichiometrically interacting molecules within a quaternary complex, it is more often used to denote larger complexes composed of indefinite numbers of molecules that form sphere-, rod-, or sheet-like species. Colloids, liquid crystals, biomolecular condensates, micelles, liposomes and biological membranes are examples of supramolecular assemblies, and their realm of study is known as supramolecular chemistry. The dimensions of supramolecular assemblies can range from nanometers to micrometers. Thus they allow access to nanoscale objects using a bottom-up approach in far fewer steps than a single molecule of similar dimensions. The process by which a supramolecular assembly forms is called molecular self-assembly. Some try to dist ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogenation
Hydrogenation is a chemical reaction between molecular hydrogen (H2) and another compound or element, usually in the presence of a Catalysis, catalyst such as nickel, palladium or platinum. The process is commonly employed to redox, reduce or Saturated and unsaturated compounds, saturate organic compounds. Hydrogenation typically constitutes the addition of pairs of hydrogen atoms to a molecule, often an alkene. Catalysts are required for the reaction to be usable; non-catalytic hydrogenation takes place only at very high temperatures. Hydrogenation reduces Double bond, double and Triple bond, triple bonds in hydrocarbons. Process Hydrogenation has three components, the Saturated and unsaturated compounds, unsaturated substrate, the hydrogen (or hydrogen source) and, invariably, a catalyst. The redox, reduction reaction is carried out at different temperatures and pressures depending upon the substrate and the activity of the catalyst. Related or competing reactions The same ca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aromatic Aldehydes
In chemistry, aromaticity is a chemical property of cyclic (ring-shaped), ''typically'' planar (flat) molecular structures with pi bonds in resonance (those containing delocalized electrons) that gives increased stability compared to saturated compounds having single bonds, and other geometric or connective non-cyclic arrangements with the same set of atoms. Aromatic rings are very stable and do not break apart easily. Organic compounds that are not aromatic are classified as aliphatic compounds—they might be cyclic, but only aromatic rings have enhanced stability. The term ''aromaticity'' with this meaning is historically related to the concept of having an aroma, but is a distinct property from that meaning. Since the most common aromatic compounds are derivatives of benzene (an aromatic hydrocarbon common in petroleum and its distillates), the word ''aromatic'' occasionally refers informally to benzene derivatives, and so it was first defined. Nevertheless, many non-benz ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

.png)