Natural gas on:
[Wikipedia]
[Google]
[Amazon]
 Natural gas (also called fossil gas or simply gas) is a naturally occurring mixture of gaseous hydrocarbons consisting primarily of methane in addition to various smaller amounts of other higher alkanes. Low levels of trace gases like carbon dioxide,
Natural gas (also called fossil gas or simply gas) is a naturally occurring mixture of gaseous hydrocarbons consisting primarily of methane in addition to various smaller amounts of other higher alkanes. Low levels of trace gases like carbon dioxide,
 In the 19th century, natural gas was primarily obtained as a by-product of producing oil. The small, light gas carbon chains came out of solution as the extracted fluids underwent pressure reduction from the reservoir to the surface, similar to uncapping a soft drink bottle where the carbon dioxide
In the 19th century, natural gas was primarily obtained as a by-product of producing oil. The small, light gas carbon chains came out of solution as the extracted fluids underwent pressure reduction from the reservoir to the surface, similar to uncapping a soft drink bottle where the carbon dioxide  It is estimated that there are about 900,000 km3 of "unconventional" gas such as shale gas, of which 180,000 km3 may be recoverable. In turn, many studies from
It is estimated that there are about 900,000 km3 of "unconventional" gas such as shale gas, of which 180,000 km3 may be recoverable. In turn, many studies from
 Shale gas is natural gas produced from shale. Because shale has matrix permeability too low to allow gas to flow in economical quantities, shale gas wells depend on fractures to allow the gas to flow. Early shale gas wells depended on natural fractures through which gas flowed; almost all shale gas wells today require fractures artificially created by hydraulic fracturing. Since 2000, shale gas has become a major source of natural gas in the United States and Canada. Because of increased shale gas production the United States was in 2014 the number one natural gas producer in the world. The production of shale gas in the United States has been described as a "shale gas revolution" and as "one of the landmark events in the 21st century."
Following the increased production in the United States, shale gas exploration is beginning in countries such as Poland, China, and South Africa. Chinese geologists have identified the Sichuan Basin as a promising target for shale gas drilling, because of the similarity of shales to those that have proven productive in the United States. Production from the Wei-201 well is between 10,000 and 20,000 m3 per day. In late 2020, China National Petroleum Corporation claimed daily production of 20 million cubic meters of gas from its Changning-Weiyuan demonstration zone.
Shale gas is natural gas produced from shale. Because shale has matrix permeability too low to allow gas to flow in economical quantities, shale gas wells depend on fractures to allow the gas to flow. Early shale gas wells depended on natural fractures through which gas flowed; almost all shale gas wells today require fractures artificially created by hydraulic fracturing. Since 2000, shale gas has become a major source of natural gas in the United States and Canada. Because of increased shale gas production the United States was in 2014 the number one natural gas producer in the world. The production of shale gas in the United States has been described as a "shale gas revolution" and as "one of the landmark events in the 21st century."
Following the increased production in the United States, shale gas exploration is beginning in countries such as Poland, China, and South Africa. Chinese geologists have identified the Sichuan Basin as a promising target for shale gas drilling, because of the similarity of shales to those that have proven productive in the United States. Production from the Wei-201 well is between 10,000 and 20,000 m3 per day. In late 2020, China National Petroleum Corporation claimed daily production of 20 million cubic meters of gas from its Changning-Weiyuan demonstration zone.
 The image below is a schematic block flow diagram of a typical natural gas processing plant. It shows the various unit processes used to convert raw natural gas into sales gas pipelined to the end user markets.
The block flow diagram also shows how processing of the raw natural gas yields byproduct sulfur, byproduct ethane, and natural gas liquids (NGL) propane, butanes and natural gasoline (denoted as
The image below is a schematic block flow diagram of a typical natural gas processing plant. It shows the various unit processes used to convert raw natural gas into sales gas pipelined to the end user markets.
The block flow diagram also shows how processing of the raw natural gas yields byproduct sulfur, byproduct ethane, and natural gas liquids (NGL) propane, butanes and natural gasoline (denoted as 
 As of mid-2020, natural gas production in the US had peaked three times, with current levels exceeding both previous peaks. It reached 24.1 trillion cubic feet per year in 1973, followed by a decline, and reached 24.5 trillion cubic feet in 2001. After a brief drop, withdrawals increased nearly every year since 2006 (owing to the shale gas boom), with 2017 production at 33.4 trillion cubic feet and 2019 production at 40.7 trillion cubic feet. After the third peak in December 2019, extraction continued to fall from March onward due to decreased demand caused by the
As of mid-2020, natural gas production in the US had peaked three times, with current levels exceeding both previous peaks. It reached 24.1 trillion cubic feet per year in 1973, followed by a decline, and reached 24.5 trillion cubic feet in 2001. After a brief drop, withdrawals increased nearly every year since 2006 (owing to the shale gas boom), with 2017 production at 33.4 trillion cubic feet and 2019 production at 40.7 trillion cubic feet. After the third peak in December 2019, extraction continued to fall from March onward due to decreased demand caused by the

 Because of its low density, it is not easy to store natural gas or to transport it by vehicle. Natural gas pipelines are impractical across oceans, since the gas needs to be cooled down and compressed, as the friction in the pipeline causes the gas to heat up. Many existing pipelines in the US are close to reaching their capacity, prompting some politicians representing northern states to speak of potential shortages. The large trade cost implies that natural gas markets are globally much less integrated, causing significant price differences across countries. In Western Europe, the gas pipeline network is already dense. New pipelines are planned or under construction in Eastern Europe and between gas fields in Russia, Near East and
Because of its low density, it is not easy to store natural gas or to transport it by vehicle. Natural gas pipelines are impractical across oceans, since the gas needs to be cooled down and compressed, as the friction in the pipeline causes the gas to heat up. Many existing pipelines in the US are close to reaching their capacity, prompting some politicians representing northern states to speak of potential shortages. The large trade cost implies that natural gas markets are globally much less integrated, causing significant price differences across countries. In Western Europe, the gas pipeline network is already dense. New pipelines are planned or under construction in Eastern Europe and between gas fields in Russia, Near East and  In the past, the natural gas which was recovered in the course of recovering petroleum could not be profitably sold, and was simply burned at the oil field in a process known as flaring. Flaring is now illegal in many countries. Additionally, higher demand in the last 20–30 years has made production of gas associated with oil economically viable. As a further option, the gas is now sometimes re- injected into the formation for enhanced oil recovery by pressure maintenance as well as miscible or immiscible flooding. Conservation, re-injection, or flaring of natural gas associated with oil is primarily dependent on proximity to markets (pipelines), and regulatory restrictions.
Natural gas can be indirectly exported through the absorption in other physical output. A recent study suggests that the expansion of shale gas production in the US has caused prices to drop relative to other countries. This has caused a boom in energy intensive manufacturing sector exports, whereby the average dollar unit of US manufacturing exports has almost tripled its energy content between 1996 and 2012.
A "master gas system" was invented in Saudi Arabia in the late 1970s, ending any necessity for flaring. Satellite and nearby infra-red camera observations, however, shows that flaring and venting are still happening in some countries.
Natural gas is used to generate electricity and heat for desalination. Similarly, some landfills that also discharge methane gases have been set up to capture the methane and generate electricity.
Natural gas is often stored underground eferences about geological storage needednside depleted gas reservoirs from previous gas wells,
In the past, the natural gas which was recovered in the course of recovering petroleum could not be profitably sold, and was simply burned at the oil field in a process known as flaring. Flaring is now illegal in many countries. Additionally, higher demand in the last 20–30 years has made production of gas associated with oil economically viable. As a further option, the gas is now sometimes re- injected into the formation for enhanced oil recovery by pressure maintenance as well as miscible or immiscible flooding. Conservation, re-injection, or flaring of natural gas associated with oil is primarily dependent on proximity to markets (pipelines), and regulatory restrictions.
Natural gas can be indirectly exported through the absorption in other physical output. A recent study suggests that the expansion of shale gas production in the US has caused prices to drop relative to other countries. This has caused a boom in energy intensive manufacturing sector exports, whereby the average dollar unit of US manufacturing exports has almost tripled its energy content between 1996 and 2012.
A "master gas system" was invented in Saudi Arabia in the late 1970s, ending any necessity for flaring. Satellite and nearby infra-red camera observations, however, shows that flaring and venting are still happening in some countries.
Natural gas is used to generate electricity and heat for desalination. Similarly, some landfills that also discharge methane gases have been set up to capture the methane and generate electricity.
Natural gas is often stored underground eferences about geological storage needednside depleted gas reservoirs from previous gas wells,
 Natural gas dispensed in a residential setting can generate temperatures in excess of making it a powerful domestic cooking and heating fuel. In much of the developed world it is supplied through pipes to homes, where it is used for many purposes including ranges and ovens,
Natural gas dispensed in a residential setting can generate temperatures in excess of making it a powerful domestic cooking and heating fuel. In much of the developed world it is supplied through pipes to homes, where it is used for many purposes including ranges and ovens, Fuel Gas Code, except when approved by the Public Safety Authority or when insurance companies have more stringent requirements.{{
Generally, natural gas system pressures are not allowed to exceed 5 psig (34.5 kPa) unless all of the following conditions are met:
* The AHJ will allow a higher pressure.
* The distribution pipe is welded. (Note: 2. Some jurisdictions may also require that welded joints be radiographed to verify continuity).
* The pipes are closed for protection and placed in a ventilated area that does not allow gas accumulation.
* The pipe is installed in the areas used for industrial processes, research, storage or mechanical equipment rooms.
Generally, a maximum liquefied petroleum gas pressure of 20 psig (138 kPa) is allowed, provided the building is used specifically for industrial or research purposes and is constructed in accordance with NFPA 58: Liquefied Petroleum Gas Code, Chapter 7.
A seismic earthquake valve operating at a pressure of 55 psig (3.7 bar) can stop the flow of natural gas into the site wide natural gas distribution piping network (that runs (outdoors underground, above building roofs, and or within the upper supports of a canopy roof). Seismic earthquake valves are designed for use at a maximum of 60 psig.
In Australia, natural gas is transported from gas processing facilities to regulator stations via Transmission pipelines. Gas is then regulated down to distributed pressures and the gas is distributed around a gas network via gas mains. Small branches from the network, called services, connect individual domestic dwellings, or multi-dwelling buildings to the network. The networks typically range in pressures from 7 kPa (low pressure) to 515 kPa (high pressure). Gas is then regulated down to 1.1 kPa or 2.75 kPa, before being metered and passed to the consumer for domestic use. Natural gas mains are made from a variety of materials: historically cast iron, though more modern mains are made from steel or polyethylene.
In the US compressed natural gas (CNG) is available in some rural areas as an alternative to less expensive and more abundant LPG ( liquefied petroleum gas), the dominant source of rural gas. It is used in homes lacking direct connections to 
 Human activity is responsible for about 60% of all
Human activity is responsible for about 60% of all
"Anthropogenic and Natural Radiative Forcing"
Table 8.7 on page 714. In: ''Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change''. Stocker, T.F., D. Qin, G.-K. Plattner, M. Tignor, S.K. Allen, J. Boschung, A. Nauels, Y. Xia, V. Bex and P.M. Midgley (eds.). Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA
Anthropogenic and Natural Radiative Forcing
/ref> Natural gas is intentionally released or is otherwise known to leak during the extraction, storage, transportation, and distribution of fossil fuels. Globally, methane accounts for an estimated 33% of anthropogenic greenhouse gas warming.{{Cite web , url=https://www.globalmethane.org/documents/gmi-mitigation-factsheet.pdf , title=Global Methane Emissions and Mitigation Opportunities , publisher= Global Methane Initiative , access-date=2020-04-24 The decomposition of municipal solid waste (a source of landfill gas) and wastewater account for an additional 18% of such emissions. These estimates include substantial uncertainties which should be reduced in the near future with improved satellite measurements, such as those planned for
 The natural gas extraction workforce face unique health and safety challenges.
The natural gas extraction workforce face unique health and safety challenges.
 Explosions caused by natural
Explosions caused by natural
 {{Update section, date=October 2022
Quantities of natural gas are measured in standard cubic meters (cubic meter of gas at temperature {{convert, 15, °C, °F, abbr=on and pressure {{convert, 101.325, kPa, psi, abbr=on) or
{{Update section, date=October 2022
Quantities of natural gas are measured in standard cubic meters (cubic meter of gas at temperature {{convert, 15, °C, °F, abbr=on and pressure {{convert, 101.325, kPa, psi, abbr=on) or

 In
In
online review
Global Fossil Infrastructure Tracker
Carbon Mapper Data Portal featuring methane point source data
{{Fuel gas {{Authority control {{DEFAULTSORT:Natural Gas Chinese inventions Fuel gas Fossil fuels
 Natural gas (also called fossil gas or simply gas) is a naturally occurring mixture of gaseous hydrocarbons consisting primarily of methane in addition to various smaller amounts of other higher alkanes. Low levels of trace gases like carbon dioxide,
Natural gas (also called fossil gas or simply gas) is a naturally occurring mixture of gaseous hydrocarbons consisting primarily of methane in addition to various smaller amounts of other higher alkanes. Low levels of trace gases like carbon dioxide, nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
, hydrogen sulfide, and helium are also usually present. Natural gas is colorless and odorless, so odorizers such as mercaptan (which smells like sulfur or rotten eggs) are commonly added to natural gas supplies for safety so that leaks can be readily detected.
Natural gas is a fossil fuel and non-renewable resource that is formed when layers of organic matter (primarily marine microorganisms) decompose under anaerobic
Anaerobic means "living, active, occurring, or existing in the absence of free oxygen", as opposed to aerobic which means "living, active, or occurring only in the presence of oxygen." Anaerobic may also refer to:
* Anaerobic adhesive, a bonding a ...
conditions and are subjected to intense heat and pressure underground over millions of years. The energy that the decayed organisms originally obtained from the sun via photosynthesis is stored as chemical energy within the molecules of methane and other hydrocarbons.
Natural gas can be burned for heating, cooking, and electricity generation. It is also used as a chemical feedstock in the manufacture of plastics and other commercially important organic chemicals
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. The s ...
and less commonly used as a fuel for vehicles.
The extraction and consumption of natural gas is a major and growing contributor to climate change. Both the gas itself (specifically methane) and carbon dioxide, which is released when natural gas is burned, are greenhouse gases. When burned for heat or electricity, natural gas emits fewer toxic air pollutants, less carbon dioxide, and almost no particulate matter compared to other fossil and biomass fuels. However, gas venting and unintended fugitive emissions throughout the supply chain can result in natural gas having a similar carbon footprint to other fossil fuels overall.
Natural gas can be found in underground geological formations, often alongside other fossil fuels like coal and oil
An oil is any nonpolar chemical substance that is composed primarily of hydrocarbons and is hydrophobic (does not mix with water) & lipophilic (mixes with other oils). Oils are usually flammable and surface active. Most oils are unsaturated ...
(petroleum). Most natural gas has been created through either biogenic or thermogenic processes. Biogenic gas is formed when methanogenic organisms in marshes, bogs, landfills, and shallow sediments anaerobically decompose but are not subjected to high temperatures and pressures. Thermogenic gas takes a much longer period of time to form and is created when organic matter is heated and compressed deep underground.
During petroleum production, natural gas is sometimes flared rather than being collected and used. Before natural gas can be burned as a fuel or used in manufacturing processes, it almost always has to be processed to remove impurities such as water. The byproducts of this processing include ethane
Ethane ( , ) is an organic chemical compound with chemical formula . At standard temperature and pressure, ethane is a colorless, odorless gas. Like many hydrocarbons, ethane is isolated on an industrial scale from natural gas and as a petroc ...
, propane, butanes, pentanes, and higher molecular weight hydrocarbons. Hydrogen sulfide (which may be converted into pure sulfur), carbon dioxide, water vapor, and sometimes helium and nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
must also be removed.
Natural gas is sometimes informally referred to simply as "gas", especially when it is being compared to other energy sources, such as oil or coal. However, it is not to be confused with gasoline, which is often shortened in colloquial usage to "gas", especially in North America.
Natural gas is measured in standard cubic meters or standard cubic feet
A standard cubic foot (scf) is a unit used both in the natural gas industry to represent an amount of natural gas and in other industries where other gases are used. It is the unit commonly used when following the customary system, a collection ...
. The density compared to air ranges from 0.58 (16.8 g/mole, 0.71 kg per standard cubic meter) to as high as 0.79 (22.9 g/mole, 0.97 kg per scm), but generally less than 0.64 (18.5 g/mole, 0.78 kg per scm). For comparison, pure methane (16.0425 g/mole) has a density 0.5539 times that of air (0.678 kg per standard cubic meter).
History
Natural gas can come out of the ground and cause a long-burning fire. In ancient Greece, the gas flames atMount Chimaera
Mount Chimaera was the name of a place in ancient Lycia, notable for constantly burning fires. It is thought to be the area called Yanartaş in Turkey, where methane and other gases emerge from the rock and burn. Some ancient sources considered i ...
contributed to the legend of the fire-breathing creature Chimera
Chimera, Chimaera, or Chimaira (Greek for " she-goat") originally referred to:
* Chimera (mythology), a fire-breathing monster of Ancient Lycia said to combine parts from multiple animals
* Mount Chimaera, a fire-spewing region of Lycia or Cilici ...
. In ancient China, gas resulting from the drilling for brines was first used by about 400 BC. The Chinese transported gas seeping from the ground in crude pipelines of bamboo to where it was used to boil salt water to extract the salt in the Ziliujing District of Sichuan.
The discovery and identification of natural gas in the Americas happened in 1626. In 1821, William Hart successfully dug the first natural gas well at Fredonia, New York, United States, which led to the formation of the Fredonia Gas Light Company. The city of Philadelphia
Philadelphia, often called Philly, is the List of municipalities in Pennsylvania#Municipalities, largest city in the Commonwealth (U.S. state), Commonwealth of Pennsylvania, the List of United States cities by population, sixth-largest city i ...
created the first municipally owned natural gas distribution venture in 1836. By 2009, 66,000 km3 (16,000 mi3) (or 8%) had been used out of the total 850,000 km3 (200,000 mi3) of estimated remaining recoverable reserves of natural gas.
Sources
Natural gas
 In the 19th century, natural gas was primarily obtained as a by-product of producing oil. The small, light gas carbon chains came out of solution as the extracted fluids underwent pressure reduction from the reservoir to the surface, similar to uncapping a soft drink bottle where the carbon dioxide
In the 19th century, natural gas was primarily obtained as a by-product of producing oil. The small, light gas carbon chains came out of solution as the extracted fluids underwent pressure reduction from the reservoir to the surface, similar to uncapping a soft drink bottle where the carbon dioxide effervesce
Effervescence is the escape of gas from an aqueous solution and the foaming or fizzing that results from that release. The word effervescence is derived from the Latin verb ''fervere'' (to boil), preceded by the adverb ''ex''. It has the same lin ...
s. The gas was often viewed as a by-product, a hazard, and a disposal problem in active oil fields. The large volumes produced could not be used until relatively expensive pipeline and storage facilities were constructed to deliver the gas to consumer markets.
Until the early part of the 20th century, most natural gas associated with oil was either simply released or burned off at oil fields. Gas venting and production flaring are still practised in modern times, but efforts are ongoing around the world to retire them, and to replace them with other commercially viable and useful alternatives. Unwanted gas (or stranded gas without a market) is often returned to the reservoir with 'injection' wells while awaiting a possible future market or to re-pressurize the formation, which can enhance oil extraction rates from other wells. In regions with a high natural gas demand (such as the US), pipelines are constructed when it is economically feasible to transport gas from a wellsite to an end consumer.
In addition to transporting gas via pipelines for use in power generation, other end uses for natural gas include export as liquefied natural gas
Liquefied natural gas (LNG) is natural gas (predominantly methane, CH4, with some mixture of ethane, C2H6) that has been cooled down to liquid form for ease and safety of non-pressurized storage or transport. It takes up about 1/600th the vol ...
(LNG) or conversion of natural gas into other liquid products via gas to liquids
Gas to liquids (GTL) is a refinery process to convert natural gas or other gaseous hydrocarbons into longer-chain hydrocarbons, such as gasoline or diesel fuel. Methane-rich gases are converted into liquid synthetic fuels. Two general strategies ...
(GTL) technologies. GTL technologies can convert natural gas into liquids products such as gasoline, diesel or jet fuel. A variety of GTL technologies have been developed, including Fischer–Tropsch (F–T), methanol to gasoline (MTG) and syngas to gasoline plus (STG+). F–T produces a synthetic crude that can be further refined into finished products, while MTG can produce synthetic gasoline from natural gas. STG+ can produce drop-in gasoline, diesel, jet fuel and aromatic chemicals directly from natural gas via a single-loop process. In 2011, Royal Dutch Shell's per day F–T plant went into operation in Qatar.
Natural gas can be "associated" (found in oil fields), or "non-associated" (isolated in natural gas field
A petroleum reservoir or oil and gas reservoir is a subsurface accumulation of hydrocarbons contained in porous or fractured rock formations.
Such reservoirs form when kerogen (ancient plant matter) is created in surrounding rock by the presence ...
s), and is also found in coal beds (as coalbed methane). It sometimes contains a significant amount of ethane
Ethane ( , ) is an organic chemical compound with chemical formula . At standard temperature and pressure, ethane is a colorless, odorless gas. Like many hydrocarbons, ethane is isolated on an industrial scale from natural gas and as a petroc ...
, propane, butane, and pentane—heavier hydrocarbons removed for commercial use prior to the methane being sold as a consumer fuel or chemical plant feedstock. Non-hydrocarbons such as carbon dioxide, nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
, helium (rarely), and hydrogen sulfide must also be removed before the natural gas can be transported.
Natural gas extracted from oil wells is called casinghead gas (whether or not truly produced up the annulus and through a casinghead outlet) or associated gas. The natural gas industry is extracting an increasing quantity of gas from challenging, unconventional resource types: sour gas, tight gas
Tight gas is natural gas produced from reservoir rocks with such low permeability that massive hydraulic fracturing is necessary to produce the well at economic rates. This natural gas is trapped within rocks with very low permeability, in other ...
, shale gas
Shale gas is an unconventional natural gas that is found trapped within shale formations. Since the 1990s a combination of horizontal drilling and hydraulic fracturing has made large volumes of shale gas more economical to produce, and some ...
, and coalbed methane.
There is some disagreement on which country has the largest proven gas reserves. Sources that consider that Russia has by far the largest proven reserves include the US Central Intelligence Agency (47,600 km3) and Energy Information Administration (47,800 km3), as well as the Organization of Petroleum Exporting Countries
The Organization of the Petroleum Exporting Countries (OPEC, ) is a cartel of countries. Founded on 14 September 1960 in Baghdad by the first five members (Iran, Iraq, Kuwait, Saudi Arabia, and Venezuela), it has, since 1965, been headquart ...
(48,700 km3). Contrarily, BP credits Russia with only 32,900 km3, which would place it in second, slightly behind Iran (33,100 to 33,800 km3, depending on the source).
MIT
The Massachusetts Institute of Technology (MIT) is a private land-grant research university in Cambridge, Massachusetts. Established in 1861, MIT has played a key role in the development of modern technology and science, and is one of the m ...
, Black & Veatch and the US Department of Energy predict that natural gas will account for a larger portion of electricity generation and heat in the future.
The world's largest gas field is the offshore South Pars / North Dome Gas-Condensate field, shared between Iran and Qatar. It is estimated to have of natural gas and of natural gas condensates.
Because natural gas is not a pure product, as the reservoir pressure drops when non-associated gas is extracted from a field under supercritical (pressure/temperature) conditions, the higher molecular weight components may partially condense upon isothermic depressurizing—an effect called retrograde condensation Retrograde condensation occurs when gas in a tube is compressed beyond the point of condensation with the effect that the liquid evaporates again. This is the opposite of condensation: the so-called retrograde condensation.
Description
If the volu ...
. The liquid thus formed may get trapped as the pores of the gas reservoir get depleted. One method to deal with this problem is to re-inject dried gas free of condensate to maintain the underground pressure and to allow re-evaporation and extraction of condensates. More frequently, the liquid condenses at the surface, and one of the tasks of the gas plant is to collect this condensate. The resulting liquid is called natural gas liquid (NGL) and has commercial value.
Shale gas
 Shale gas is natural gas produced from shale. Because shale has matrix permeability too low to allow gas to flow in economical quantities, shale gas wells depend on fractures to allow the gas to flow. Early shale gas wells depended on natural fractures through which gas flowed; almost all shale gas wells today require fractures artificially created by hydraulic fracturing. Since 2000, shale gas has become a major source of natural gas in the United States and Canada. Because of increased shale gas production the United States was in 2014 the number one natural gas producer in the world. The production of shale gas in the United States has been described as a "shale gas revolution" and as "one of the landmark events in the 21st century."
Following the increased production in the United States, shale gas exploration is beginning in countries such as Poland, China, and South Africa. Chinese geologists have identified the Sichuan Basin as a promising target for shale gas drilling, because of the similarity of shales to those that have proven productive in the United States. Production from the Wei-201 well is between 10,000 and 20,000 m3 per day. In late 2020, China National Petroleum Corporation claimed daily production of 20 million cubic meters of gas from its Changning-Weiyuan demonstration zone.
Shale gas is natural gas produced from shale. Because shale has matrix permeability too low to allow gas to flow in economical quantities, shale gas wells depend on fractures to allow the gas to flow. Early shale gas wells depended on natural fractures through which gas flowed; almost all shale gas wells today require fractures artificially created by hydraulic fracturing. Since 2000, shale gas has become a major source of natural gas in the United States and Canada. Because of increased shale gas production the United States was in 2014 the number one natural gas producer in the world. The production of shale gas in the United States has been described as a "shale gas revolution" and as "one of the landmark events in the 21st century."
Following the increased production in the United States, shale gas exploration is beginning in countries such as Poland, China, and South Africa. Chinese geologists have identified the Sichuan Basin as a promising target for shale gas drilling, because of the similarity of shales to those that have proven productive in the United States. Production from the Wei-201 well is between 10,000 and 20,000 m3 per day. In late 2020, China National Petroleum Corporation claimed daily production of 20 million cubic meters of gas from its Changning-Weiyuan demonstration zone.
Town gas
Town gas is a flammable gaseous fuel made by the destructive distillation of coal. It contains a variety of calorific gases includinghydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic ...
, carbon monoxide, methane, and other volatile hydrocarbons, together with small quantities of non-calorific gases such as carbon dioxide and nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
, and was used in a similar way to natural gas. This is a historical technology and is not usually economically competitive with other sources of fuel gas today.
Most town "gashouses" located in the eastern US in the late 19th and early 20th centuries were simple by-product coke ovens that heated bituminous coal in air-tight chambers. The gas driven off from the coal was collected and distributed through networks of pipes to residences and other buildings where it was used for cooking and lighting. (Gas heating did not come into widespread use until the last half of the 20th century.) The coal tar (or asphalt) that collected in the bottoms of the gashouse ovens was often used for roofing and other waterproofing purposes, and when mixed with sand and gravel was used for paving streets.
Crystallized natural gas – clathrates
Huge quantities of natural gas (primarily methane) exist in the form of clathrates under sediment on offshore continental shelves and on land in arctic regions that experience permafrost, such as those in Siberia. Hydrates require a combination of high pressure and low temperature to form. In 2013, Japan Oil, Gas and Metals National Corporation (JOGMEC) announced that they had recovered commercially relevant quantities of natural gas from methane hydrate.Processing
pentanes
The pentanes are a group of alkanes with five carbon atoms with the formula
In science, a formula is a concise way of expressing information symbolically, as in a mathematical formula or a ''chemical formula''. The informal use of the term '' ...
+).
Demand
 As of mid-2020, natural gas production in the US had peaked three times, with current levels exceeding both previous peaks. It reached 24.1 trillion cubic feet per year in 1973, followed by a decline, and reached 24.5 trillion cubic feet in 2001. After a brief drop, withdrawals increased nearly every year since 2006 (owing to the shale gas boom), with 2017 production at 33.4 trillion cubic feet and 2019 production at 40.7 trillion cubic feet. After the third peak in December 2019, extraction continued to fall from March onward due to decreased demand caused by the
As of mid-2020, natural gas production in the US had peaked three times, with current levels exceeding both previous peaks. It reached 24.1 trillion cubic feet per year in 1973, followed by a decline, and reached 24.5 trillion cubic feet in 2001. After a brief drop, withdrawals increased nearly every year since 2006 (owing to the shale gas boom), with 2017 production at 33.4 trillion cubic feet and 2019 production at 40.7 trillion cubic feet. After the third peak in December 2019, extraction continued to fall from March onward due to decreased demand caused by the COVID-19 pandemic in the US
The COVID-19 pandemic in the United States is a part of the worldwide pandemic of coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). In the United States, it has resulted in confirme ...
.
The 2021 global energy crisis The 2021 global energy crisis is an ongoing shortage of energy across the world, affecting countries such as the United Kingdom and China, among others.
Background
In December 2020, after months of restrictions, China fully blocked coal import ...
was driven by a global surge in demand as the world quit the economic recession caused by COVID-19, particularly due to strong energy demand in Asia.
Storage and transport

 Because of its low density, it is not easy to store natural gas or to transport it by vehicle. Natural gas pipelines are impractical across oceans, since the gas needs to be cooled down and compressed, as the friction in the pipeline causes the gas to heat up. Many existing pipelines in the US are close to reaching their capacity, prompting some politicians representing northern states to speak of potential shortages. The large trade cost implies that natural gas markets are globally much less integrated, causing significant price differences across countries. In Western Europe, the gas pipeline network is already dense. New pipelines are planned or under construction in Eastern Europe and between gas fields in Russia, Near East and
Because of its low density, it is not easy to store natural gas or to transport it by vehicle. Natural gas pipelines are impractical across oceans, since the gas needs to be cooled down and compressed, as the friction in the pipeline causes the gas to heat up. Many existing pipelines in the US are close to reaching their capacity, prompting some politicians representing northern states to speak of potential shortages. The large trade cost implies that natural gas markets are globally much less integrated, causing significant price differences across countries. In Western Europe, the gas pipeline network is already dense. New pipelines are planned or under construction in Eastern Europe and between gas fields in Russia, Near East and Northern Africa
North Africa, or Northern Africa is a region encompassing the northern portion of the African continent. There is no singularly accepted scope for the region, and it is sometimes defined as stretching from the Atlantic shores of Mauritania in t ...
and Western Europe.
Whenever gas is bought or sold at custody transfer points, rules and agreements are made regarding the gas quality. These may include the maximum allowable concentration of , and . Usually sales quality gas that has been treated to remove contamination is traded on a "dry gas" basis and is required to be commercially free from objectionable odours, materials, and dust or other solid or liquid matter, waxes, gums and gum forming constituents, which might damage or adversely affect operation of equipment downstream of the custody transfer point.
LNG carrier
An LNG carrier is a tank ship designed for transporting liquefied natural gas (LNG).
History
The first LNG carrier '' Methane Pioneer'' () carrying , classed by Bureau Veritas, left the Calcasieu River on the Louisiana Gulf coast on 25 January ...
s transport liquefied natural gas
Liquefied natural gas (LNG) is natural gas (predominantly methane, CH4, with some mixture of ethane, C2H6) that has been cooled down to liquid form for ease and safety of non-pressurized storage or transport. It takes up about 1/600th the vol ...
(LNG) across oceans, while tank trucks can carry liquefied or compressed natural gas (CNG) over shorter distances. Sea transport using CNG carrier ships that are now under development may be competitive with LNG transport in specific conditions.
Gas is turned into liquid at a liquefaction plant, and is returned to gas form at regasification
Regasification is a process of converting liquefied natural gas (LNG) at −162 °C (−260 °F) temperature back to natural gas at atmospheric temperature. LNG gasification plants can be located on land as well as on floating barges, i.e. a Float ...
plant at the terminal. Shipborne regasification equipment is also used. LNG is the preferred form for long distance, high volume transportation of natural gas, whereas pipeline is preferred for transport for distances up to over land and approximately half that distance offshore.
CNG is transported at high pressure, typically above . Compressors and decompression equipment are less capital intensive and may be economical in smaller unit sizes than liquefaction/regasification plants. Natural gas trucks and carriers may transport natural gas directly to end-users, or to distribution points such as pipelines.
 In the past, the natural gas which was recovered in the course of recovering petroleum could not be profitably sold, and was simply burned at the oil field in a process known as flaring. Flaring is now illegal in many countries. Additionally, higher demand in the last 20–30 years has made production of gas associated with oil economically viable. As a further option, the gas is now sometimes re- injected into the formation for enhanced oil recovery by pressure maintenance as well as miscible or immiscible flooding. Conservation, re-injection, or flaring of natural gas associated with oil is primarily dependent on proximity to markets (pipelines), and regulatory restrictions.
Natural gas can be indirectly exported through the absorption in other physical output. A recent study suggests that the expansion of shale gas production in the US has caused prices to drop relative to other countries. This has caused a boom in energy intensive manufacturing sector exports, whereby the average dollar unit of US manufacturing exports has almost tripled its energy content between 1996 and 2012.
A "master gas system" was invented in Saudi Arabia in the late 1970s, ending any necessity for flaring. Satellite and nearby infra-red camera observations, however, shows that flaring and venting are still happening in some countries.
Natural gas is used to generate electricity and heat for desalination. Similarly, some landfills that also discharge methane gases have been set up to capture the methane and generate electricity.
Natural gas is often stored underground eferences about geological storage needednside depleted gas reservoirs from previous gas wells,
In the past, the natural gas which was recovered in the course of recovering petroleum could not be profitably sold, and was simply burned at the oil field in a process known as flaring. Flaring is now illegal in many countries. Additionally, higher demand in the last 20–30 years has made production of gas associated with oil economically viable. As a further option, the gas is now sometimes re- injected into the formation for enhanced oil recovery by pressure maintenance as well as miscible or immiscible flooding. Conservation, re-injection, or flaring of natural gas associated with oil is primarily dependent on proximity to markets (pipelines), and regulatory restrictions.
Natural gas can be indirectly exported through the absorption in other physical output. A recent study suggests that the expansion of shale gas production in the US has caused prices to drop relative to other countries. This has caused a boom in energy intensive manufacturing sector exports, whereby the average dollar unit of US manufacturing exports has almost tripled its energy content between 1996 and 2012.
A "master gas system" was invented in Saudi Arabia in the late 1970s, ending any necessity for flaring. Satellite and nearby infra-red camera observations, however, shows that flaring and venting are still happening in some countries.
Natural gas is used to generate electricity and heat for desalination. Similarly, some landfills that also discharge methane gases have been set up to capture the methane and generate electricity.
Natural gas is often stored underground eferences about geological storage needednside depleted gas reservoirs from previous gas wells, salt domes
A salt dome is a type of structural dome formed when salt (or other evaporite minerals) intrudes into overlying rocks in a process known as diapirism. Salt domes can have unique surface and subsurface structures, and they can be discovered using ...
, or in tanks as liquefied natural gas. The gas is injected in a time of low demand and extracted when demand picks up. Storage nearby end users helps to meet volatile demands, but such storage may not always be practicable.
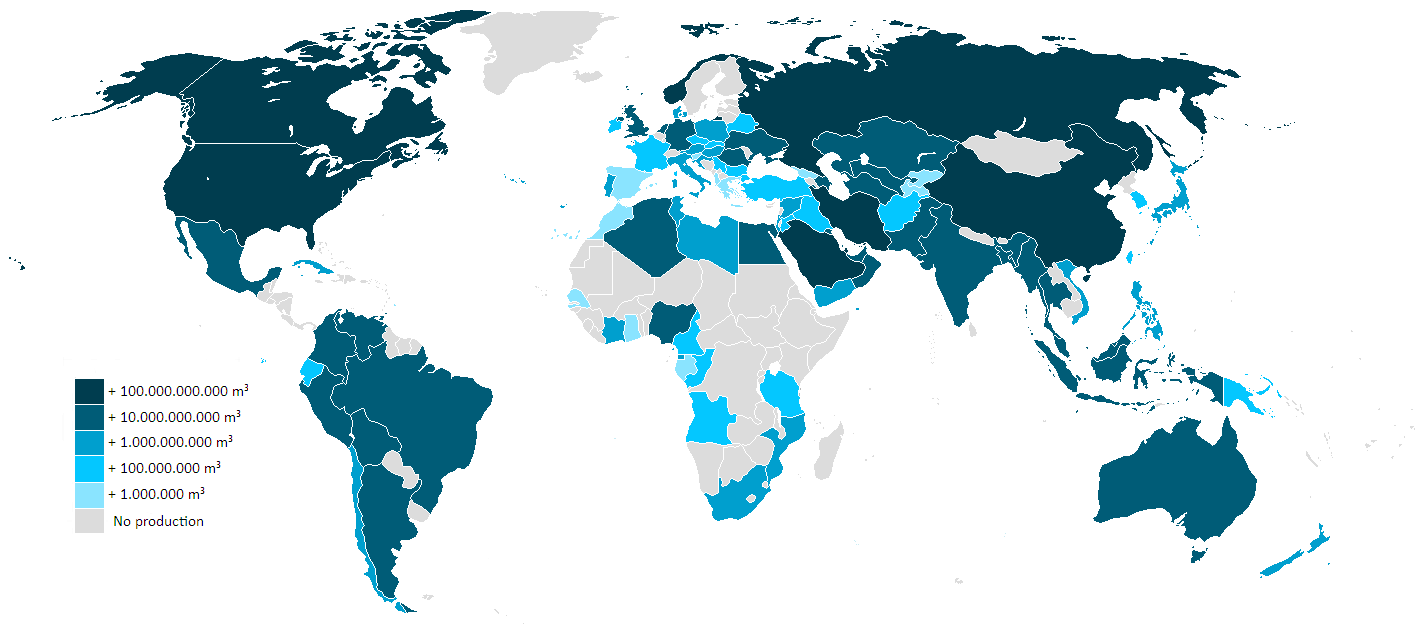
With 15 countries accounting for 84% of the worldwide extraction, access to natural gas has become an important issue in international politics, and countries vie for control of pipelines. In the first decade of the 21st century, Gazprom, the state-owned energy company in Russia, engaged in disputes with Ukraine
Ukraine ( uk, Україна, Ukraïna, ) is a country in Eastern Europe. It is the second-largest European country after Russia, which it borders to the east and northeast. Ukraine covers approximately . Prior to the ongoing Russian inv ...
and Belarus
Belarus,, , ; alternatively and formerly known as Byelorussia (from Russian ). officially the Republic of Belarus,; rus, Республика Беларусь, Respublika Belarus. is a landlocked country in Eastern Europe. It is bordered by ...
over the price of natural gas, which have created concerns that gas deliveries to parts of Europe could be cut off for political reasons. The United States is preparing to export natural gas.
Floating liquefied natural gas
Floating liquefied natural gas (FLNG) is an innovative technology designed to enable the development of offshore gas resources that would otherwise remain untapped due to environmental or economic factors which currently make them impractical to develop via a land-based LNG operation. FLNG technology also provides a number of environmental and economic advantages: * Environmental – Because all processing is done at the gas field, there is no requirement for long pipelines to shore, compression units to pump the gas to shore, dredging and jetty construction, and onshore construction of an LNG processing plant, which significantly reduces the environmental footprint. Avoiding construction also helps preserve marine and coastal environments. In addition, environmental disturbance will be minimised during decommissioning because the facility can easily be disconnected and removed before being refurbished and re-deployed elsewhere. * Economic – Where pumping gas to shore can be prohibitively expensive, FLNG makes development economically viable. As a result, it will open up new business opportunities for countries to develop offshore gas fields that would otherwise remain stranded, such as those offshore East Africa. Many gas and oil companies are considering the economic and environmental benefits of floating liquefied natural gas (FLNG). There are currently projects underway to construct five FLNG facilities. Petronas is close to completion on their FLNG-1 atDaewoo Shipbuilding and Marine Engineering
Hanwha Ocean Co., Ltd. (), formerly known as Daewoo Shipbuilding & Marine Engineering Co., Ltd. (DSME; ), is one of the "Big Three" shipbuilders of South Korea, along with Hyundai and Samsung.
History
On 21 February 2011, the A. P. Moller-Ma ...
and are underway on their FLNG-2 project at Samsung Heavy Industries. Shell Prelude is due to start production 2017. The Browse LNG project will commence FEED in 2019.
Uses
Natural gas is primarily used in the northern hemisphere. North America and Europe are major consumers. Often well head gases require removal of various hydrocarbon molecules contained within the gas. Some of these gases includeheptane
Heptane or ''n''-heptane is the straight-chain alkane with the chemical formula H3C(CH2)5CH3 or C7H16. When used as a test fuel component in anti-knock test engines, a 100% heptane fuel is the zero point of the octane rating scale (the 100 poin ...
, pentane, propane and other hydrocarbons with molecular weights above methane (). The natural gas transmission lines extend to the natural gas processing plant or unit which removes the higher-molecular weight hydrocarbons to produce natural gas with energy content between . The processed natural gas may then be used for residential, commercial and industrial uses.
Mid-stream natural gas
Natural gas flowing in the distribution lines is called mid-stream natural gas and is often used to power engines which rotate compressors. These compressors are required in the transmission line to pressurize and repressurize the mid-stream natural gas as the gas travels. Typically, natural gas powered engines require natural gas to operate at the rotational name plate specifications. Several methods are used to remove these higher molecular weighted gases for use by the natural gas engine. A few technologies are as follows: * Joule–Thomson skid * Cryogenic orchiller
A chiller is a machine that removes heat from a liquid coolant via a vapor-compression, adsorption refrigeration, or absorption refrigeration cycles. This liquid can then be circulated through a heat exchanger to cool equipment, or another p ...
system
* Chemical
A chemical substance is a form of matter having constant chemical composition and characteristic properties. Some references add that chemical substance cannot be separated into its constituent elements by physical separation methods, i.e., w ...
enzymology system
Power generation
Domestic use
 Natural gas dispensed in a residential setting can generate temperatures in excess of making it a powerful domestic cooking and heating fuel. In much of the developed world it is supplied through pipes to homes, where it is used for many purposes including ranges and ovens,
Natural gas dispensed in a residential setting can generate temperatures in excess of making it a powerful domestic cooking and heating fuel. In much of the developed world it is supplied through pipes to homes, where it is used for many purposes including ranges and ovens, heating
A central heating system provides warmth to a number of spaces within a building from one main source of heat. It is a component of heating, ventilation, and air conditioning (short: HVAC) systems, which can both cool and warm interior spaces.
...
/cooling
Cooling is removal of heat, usually resulting in a lower temperature and/or phase change. Temperature lowering achieved by any other means may also be called cooling.ASHRAE Terminology, https://www.ashrae.org/technical-resources/free-resources/as ...
, and central heating. Heaters in homes and other buildings may include boilers, furnaces, and water heaters. Both North America and Europe are major consumers of natural gas.
Domestic appliances, furnaces, and boilers use low pressure, usually with a standard preassure around over atmospheric preassure. The pressures in the supply lines vary, either the standard utilization pressure (UP) mentioned above or elevated pressure (EP), which may be anywhere from over atmospheric preassure. Systems using EP have a regulator at the service entrance to step down the pressure to UP.
Natural Gas Piping Systems inside buildings are often designed with pressures of , and have downstream pressure regulators to reduce pressure as needed. The maximum allowable operating pressure for natural gas piping systems within a building is based on NFPA 54: Nationalpublic utility
A public utility company (usually just utility) is an organization that maintains the infrastructure for a public service (often also providing a service using that infrastructure). Public utilities are subject to forms of public control and r ...
provided gas, or to fuel portable grills. Natural gas is also supplied by independent natural gas suppliers through Natural Gas Choice
Natural Gas Choice programs in United States of America allow residential consumers and other small volume gas users to purchase natural gas from someone other than their traditional utility company. With Natural Gas Choice programs, customers c ...
programs{{clarify, date=May 2019 throughout the United States.

Transportation
CNG is a cleaner and also cheaper alternative to other automobile fuels such as gasoline (petrol). By the end of 2014, there were over 20 million natural gas vehicles worldwide, led byIran
Iran, officially the Islamic Republic of Iran, and also called Persia, is a country located in Western Asia. It is bordered by Iraq and Turkey to the west, by Azerbaijan and Armenia to the northwest, by the Caspian Sea and Turkmeni ...
(3.5 million), China (3.3 million), Pakistan (2.8 million), Argentina (2.5 million), India
India, officially the Republic of India (Hindi: ), is a country in South Asia. It is the seventh-largest country by area, the second-most populous country, and the most populous democracy in the world. Bounded by the Indian Ocean on the so ...
(1.8 million), and Brazil (1.8 million). The energy efficiency is generally equal to that of gasoline engines, but lower compared with modern diesel engines. Gasoline/petrol vehicles converted to run on natural gas suffer because of the low compression ratio of their engines, resulting in a cropping of delivered power while running on natural gas (10–15%). CNG-specific engines, however, use a higher compression ratio due to this fuel's higher octane number of 120–130.
Besides use in road vehicles, CNG can also be used in aircraft. Compressed natural gas has been used in some aircraft like the Aviat Aircraft
Aviat Aircraft Inc. is an American manufacturer of sport and utility aircraft based in Afton, Wyoming.Bayerl, Robby; Martin Berkemeier; et al: ''World Directory of Leisure Aviation 2011-12'', page 94. WDLA UK, Lancaster UK, 2011. ISSN 1368-485X
...
Husky 200 CNG and the Chromarat VX-1 KittyHawk
LNG is also being used in aircraft. Russian aircraft manufacturer Tupolev for instance is running a development program to produce LNG- and hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic ...
-powered aircraft. The program has been running since the mid-1970s, and seeks to develop LNG and hydrogen variants of the Tu-204 and Tu-334
The Tupolev Tu-334 (russian: Туполев Ту-334) was a Russian short-to-medium range airliner project that was developed to replace the ageing Tu-134s and Yak-42s in service around the world. The airframe was based on a shortened Tu-2 ...
passenger aircraft, and also the Tu-330 cargo aircraft. Depending on the current market price for jet fuel and LNG, fuel for an LNG-powered aircraft could cost 5,000 rubles (US$100) less per tonne, roughly 60%, with considerable reductions to carbon monoxide, hydrocarbon and nitrogen oxide emissions.{{Cn, date=October 2022
The advantages of liquid methane as a jet engine fuel are that it has more specific energy than the standard kerosene mixes do and that its low temperature can help cool the air which the engine compresses for greater volumetric efficiency, in effect replacing an intercooler. Alternatively, it can be used to lower the temperature of the exhaust.{{Cn, date=October 2022
Fertilizers
{{Seealso, 2007–2008 world food price crisis Natural gas is a major feedstock for the production of ammonia, via theHaber process
The Haber process, also called the Haber–Bosch process, is an artificial nitrogen fixation process and is the main industrial procedure for the production of ammonia today. It is named after its inventors, the German chemists Fritz Haber and ...
, for use in fertilizer production. The development of synthetic nitrogen fertilizer has significantly supported global population growth — it has been estimated that almost half the people on the Earth are currently fed as a result of synthetic nitrogen fertilizer use.
Hydrogen
{{See also, Industrial gas Natural gas can be used to producehydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic ...
, with one common method being the hydrogen reformer. Hydrogen has many applications: it is a primary feedstock for the chemical industry, a hydrogenating agent, an important commodity for oil refineries, and the fuel source in hydrogen vehicles.
Animal and fish feed
Protein rich animal and fish feed is produced by feeding natural gas to Methylococcus capsulatus bacteria on commercial scale.Other
Natural gas is also used in the manufacture offabrics
Textile is an umbrella term that includes various fiber-based materials, including fibers, yarns, filaments, threads, different fabric types, etc. At first, the word "textiles" only referred to woven fabrics. However, weaving is not the ...
, glass
Glass is a non-crystalline, often transparent, amorphous solid that has widespread practical, technological, and decorative use in, for example, window panes, tableware, and optics. Glass is most often formed by rapid cooling ( quenching ...
, steel, plastics, paint, synthetic oil, and other products. The first step in the valorization of natural gas components is usually the {{what, date=January 2021 of the alkane into olefin. The oxidative dehydrogenation of ethane leads to ethylene which can be converted forther to ethylene epoxide, ethylene glycol, acetaldehyde or other olefins. Propane can be converted to propylene or can be oxidized to acrylic acid and acrylonitrile.
Environmental effects
{{See also, Environmental impact of the energy industryGreenhouse effect and natural gas release
{{See also, Greenhouse effect, Atmospheric methane, Gas venting, Fugitive gas emissionsmethane emissions
Increasing methane emissions are a major contributor to the rising concentration of greenhouse gases in Earth's atmosphere, and are responsible for up to one-third of near-term global heating. During 2019, about 60% (360 million tons) of methane r ...
and for most of the resulting increase in atmospheric methane.{{Cite web, url=https://www.nationalgeographic.com/environment/global-warming/methane/, title=Methane, explained, date=2019-01-23, website=National Geographic, publisher=nationalgeographic.com, access-date=2020-04-24Myhre, G., D. Shindell, F.-M. Bréon, W. Collins, J. Fuglestvedt, J. Huang, D. Koch, J.-F. Lamarque, D. Lee, B. Mendoza, T. Nakajima, A. Robock, G. Stephens, T. Takemura and H. Zhang (2013"Anthropogenic and Natural Radiative Forcing"
Table 8.7 on page 714. In: ''Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change''. Stocker, T.F., D. Qin, G.-K. Plattner, M. Tignor, S.K. Allen, J. Boschung, A. Nauels, Y. Xia, V. Bex and P.M. Midgley (eds.). Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA
Anthropogenic and Natural Radiative Forcing
/ref> Natural gas is intentionally released or is otherwise known to leak during the extraction, storage, transportation, and distribution of fossil fuels. Globally, methane accounts for an estimated 33% of anthropogenic greenhouse gas warming.{{Cite web , url=https://www.globalmethane.org/documents/gmi-mitigation-factsheet.pdf , title=Global Methane Emissions and Mitigation Opportunities , publisher= Global Methane Initiative , access-date=2020-04-24 The decomposition of municipal solid waste (a source of landfill gas) and wastewater account for an additional 18% of such emissions. These estimates include substantial uncertainties which should be reduced in the near future with improved satellite measurements, such as those planned for
MethaneSAT
MethaneSAT is a planned American-New Zealand space mission currently scheduled for launch in October 2023. The mission is planned to be an Earth observation satellite that will monitor and study global methane emissions in order to combat clim ...
.
After release to the atmosphere, methane is removed by gradual oxidation to carbon dioxide and water by hydroxyl radicals ({{chem, O, H, -) formed in the troposphere or stratosphere, giving the overall chemical reaction {{chem, C, H, 4 + 2{{chem, O, 2 → {{chem, C, O, 2 + 2{{chem, H, 2, O.{{cite book , title=Environmental Chemistry , edition= 9th , publisher=CRC press , year=2010 , isbn=978-1420059205 , author=Stanley Manahan{{cite web , title=Methane: A Scientific Journey from Obscurity to Climate Super-Stardom , author=Gavin Schmidt , url=http://www.giss.nasa.gov/research/features/200409_methane/ , date=September 2004 , website=National Aeronautics and Space Administration. Goddard Institute for Space Studies , access-date=11 June 2013 While the lifetime of atmospheric methane is relatively short when compared to carbon dioxide,{{cite news , title=Curbing Emissions by Sealing Gas Leaks , url=https://www.nytimes.com/2009/10/15/business/energy-environment/15degrees.html?_r=3&hpw& , newspaper=The New York Times, date=14 October 2009 , access-date=11 June 2013 with a half-life of about 7 years, it is more efficient at trapping heat in the atmosphere, so that a given quantity of methane has 84 times the global-warming potential of carbon dioxide over a 20-year period and 28 times over a 100-year period. Natural gas is thus a potent greenhouse gas due to the strong radiative forcing of methane in the short term, and the continuing effects of carbon dioxide in the longer term.
Targeted efforts to reduce warming quickly by reducing anthropogenic methane emissions is a climate change mitigation strategy supported by the Global Methane Initiative.
Greenhouse gas emissions
When refined and burned, natural gas can produce 25–30% less carbon dioxide per joule delivered than oil, and 40–45% less than coal.{{cite web , title=Natural Gas and the Environment , url=http://www.naturalgas.org/environment/naturalgas.asp , url-status=dead , archive-url=https://web.archive.org/web/20090503132200/http://www.naturalgas.org/environment/naturalgas.asp , archive-date=3 May 2009 , access-date=11 June 2013 , publisher=NaturalGas.org It can also produce potentially fewer toxicpollutant
A pollutant or novel entity is a substance or energy introduced into the environment that has undesired effects, or adversely affects the usefulness of a resource. These can be both naturally forming (i.e. minerals or extracted compounds like o ...
s than other hydrocarbon fuels.{{cite web , url=http://www.nbr.org/downloads/pdfs/eta/PES_2011_Herberg.pdf , title=Natural Gas in Asia: History and Prospects , author=Mikkal Herberg , others=(written for 2011 Pacific Energy Summit) , website=The National Bureau of Asian Research However, compared to other major fossil fuels, natural gas causes more emissions in relative terms during the production and transportation of the fuel, meaning that the life cycle greenhouse gas emissions are about 50% higher than the direct emissions from the site of consumption.
In terms of the warming effect over 100 years, natural gas production and use comprises about one fifth of human greenhouse gas emissions, and this contribution is growing rapidly. Globally, natural gas use emitted about 7.8 billion tons of {{chem, C, O, 2 in 2020 (including flaring), while coal and oil use emitted 14.4 and 12 billion tons, respectively. The IEA estimates the energy sector (oil, natural gas, coal and bioenergy) to be responsible for about 40% of human methane emissions. According to the IPCC Sixth Assessment Report, natural gas consumption grew by 15% between 2015 and 2019, compared to a 5% increase in oil and oil product consumption.
The continued financing and construction of new gas pipelines indicates that huge emissions of fossil greenhouse gases could be locked-in for 40 to 50 years into the future.{{cite news , title=North American drilling boom threatens big blow to climate efforts, study finds , url=https://www.theguardian.com/environment/2019/apr/25/us-oil-gas-boom-climate-change-report , work=The Guardian
''The Guardian'' is a British daily newspaper. It was founded in 1821 as ''The Manchester Guardian'', and changed its name in 1959. Along with its sister papers ''The Observer'' and ''The Guardian Weekly'', ''The Guardian'' is part of the Gu ...
, date=2019-04-25 In the U.S. state of Texas
Texas (, ; Spanish: ''Texas'', ''Tejas'') is a state in the South Central region of the United States. At 268,596 square miles (695,662 km2), and with more than 29.1 million residents in 2020, it is the second-largest U.S. state by ...
alone, five new long-distance gas pipelines have been under construction, with the first entering service in 2019, and the others scheduled to come online during 2020–2022.{{cite web , title=Natural Gas Flaring and Venting: State and Federal Regulatory Overview, Trends, and Impacts , url=https://www.energy.gov/sites/prod/files/2019/08/f65/Natural%20Gas%20Flaring%20and%20Venting%20Report.pdf
, publisher=U.S. Department of Energy , access-date=2019-12-29 , date=2019-06-01{{rp, 23
To reduce its greenhouse emissions, the Netherlands
)
, anthem = ( en, "William of Nassau")
, image_map =
, map_caption =
, subdivision_type = Sovereign state
, subdivision_name = Kingdom of the Netherlands
, established_title = Before independence
, established_date = Spanish Netherl ...
is subsidizing a transition away from natural gas for all homes in the country by 2050. In Amsterdam
Amsterdam ( , , , lit. ''The Dam on the River Amstel'') is the Capital of the Netherlands, capital and Municipalities of the Netherlands, most populous city of the Netherlands, with The Hague being the seat of government. It has a population ...
, no new residential gas accounts have been allowed since 2018, and all homes in the city are expected to be converted by 2040 to use the excess heat from adjacent industrial buildings and operations.
Some cities in the United States have started prohibiting gas hookups for new houses, with state laws passed and under consideration to either require electrification or prohibit local requirements. The UK government is also experimenting with alternative home heating technologies to meet its climate goals. To preserve their businesses, natural gas utilities in the United States have been lobbying for laws preventing local electrification ordinances, and are promoting renewable natural gas
Renewable natural gas (RNG), also known as sustainable natural gas (SNG) or biomethane, is a biogas which has been upgraded to a quality similar to fossil natural gas and having a methane concentration of 90% or greater. By increasing the concentr ...
and hydrogen fuel
Hydrogen fuel refers to hydrogen which is burned as fuel with oxygen. It is zero-carbon, provided that it is created in a process that does not involve carbon. It can be used in fuel cells or internal combustion engines (see HICEV). Regarding hydr ...
.
Other pollutants
Although natural gas produces far lower amounts ofsulfur dioxide
Sulfur dioxide (IUPAC-recommended spelling) or sulphur dioxide (traditional Commonwealth English) is the chemical compound with the formula . It is a toxic gas responsible for the odor of burnt matches. It is released naturally by volcanic activ ...
and nitrogen oxides Nitrogen oxide may refer to a binary compound of oxygen and nitrogen, or a mixture of such compounds:
Charge-neutral
*Nitric oxide (NO), nitrogen(II) oxide, or nitrogen monoxide
*Nitrogen dioxide (), nitrogen(IV) oxide
* Nitrogen trioxide (), or ...
(NOx) than other fossil fuels, NOx from burning natural gas in homes can be a health hazard.Radionuclides
Natural gas extraction also produces radioactive isotopes ofpolonium
Polonium is a chemical element with the symbol Po and atomic number 84. Polonium is a chalcogen. A rare and highly radioactive metal with no stable isotopes, polonium is chemically similar to selenium and tellurium, though its metallic character ...
(Po-210), lead
Lead is a chemical element with the symbol Pb (from the Latin ) and atomic number 82. It is a heavy metal that is denser than most common materials. Lead is soft and malleable, and also has a relatively low melting point. When freshly cu ...
(Pb-210) and radon
Radon is a chemical element with the symbol Rn and atomic number 86. It is a radioactive, colourless, odourless, tasteless noble gas. It occurs naturally in minute quantities as an intermediate step in the normal radioactive decay chains through ...
(Rn-220). Radon is a gas with initial activity from 5 to 200,000 becquerels per cubic meter of gas. It decays rapidly to Pb-210 which can build up as a thin film in gas extraction equipment.
Safety concerns
Production
Some gas fields yield sour gas containing hydrogen sulfide ({{chem, H, 2, S), atoxic
Toxicity is the degree to which a chemical substance or a particular mixture of substances can damage an organism. Toxicity can refer to the effect on a whole organism, such as an animal, bacterium, or plant, as well as the effect on a subs ...
compound when inhaled. Amine gas treating
Amine gas treating, also known as amine scrubbing, gas sweetening and acid gas removal, refers to a group of processes that use aqueous solutions of various alkylamines (commonly referred to simply as amines) to remove hydrogen sulfide (H2S) and c ...
, an industrial scale process which removes acidic
In computer science, ACID ( atomicity, consistency, isolation, durability) is a set of properties of database transactions intended to guarantee data validity despite errors, power failures, and other mishaps. In the context of databases, a ...
gas
Gas is one of the four fundamental states of matter (the others being solid, liquid, and plasma).
A pure gas may be made up of individual atoms (e.g. a noble gas like neon), elemental molecules made from one type of atom (e.g. oxygen), or ...
eous components, is often used to remove hydrogen sulfide from natural gas.{{cite web, url=http://www.naturalgas.org/naturalgas/processing_ng.asp , title=Processing Natural Gas , publisher=NaturalGas.org , access-date=2011-02-06 , url-status=dead , archive-url=https://web.archive.org/web/20110101053431/http://naturalgas.org/naturalgas/processing_ng.asp , archive-date=1 January 2011
Extraction of natural gas (or oil) leads to decrease in pressure in the reservoir
A reservoir (; from French ''réservoir'' ) is an enlarged lake behind a dam. Such a dam may be either artificial, built to store fresh water or it may be a natural formation.
Reservoirs can be created in a number of ways, including contro ...
. Such decrease in pressure in turn may result in subsidence, sinking of the ground above. Subsidence may affect ecosystems, waterways, sewer and water supply systems, foundations, and so on.
Fracking
{{Main, Environmental impact of hydraulic fracturing Releasing natural gas from subsurface porous rock formations may be accomplished by a process called hydraulic fracturing or "fracking". Since the first commercial hydraulic fracturing operation in 1949, approximately one million wells have been hydraulically fractured in the United States. The production of natural gas from hydraulically fractured wells has used the technological developments of directional and horizontal drilling, which improved access to natural gas in tight rock formations. Strong growth in the production of unconventional gas from hydraulically fractured wells occurred between 2000 and 2012. In hydraulic fracturing, well operators force water mixed with a variety of chemicals through the wellbore casing into the rock. The high pressure water breaks up or "fracks" the rock, which releases gas from the rock formation. Sand and other particles are added to the water as aproppant
A proppant is a solid material, typically sand, treated sand or man-made ceramic materials, designed to keep an hydraulic fracturing, induced hydraulic fracture open, during or following a fracturing treatment, most commonly for Unconventional (oi ...
to keep the fractures in the rock open, thus enabling the gas to flow into the casing and then to the surface. Chemicals are added to the fluid to perform such functions as reducing friction and inhibiting corrosion. After the "frack", oil or gas is extracted and 30–70% of the frack fluid, i.e. the mixture of water, chemicals, sand, etc., flows back to the surface. Many gas-bearing formations also contain water, which will flow up the wellbore to the surface along with the gas, in both hydraulically fractured and non-hydraulically fractured wells. This produced water
Produced water is a term used in the oil industry or geothermal industry to describe water that is produced as a byproduct during the extraction of oil and natural gas, or used as a medium for heat extraction. Produced water is the kind of brackis ...
often has a high content of salt and other dissolved minerals that occur in the formation.
The volume of water used to hydraulically fracture wells varies according to the hydraulic fracturing technique. In the United States, the average volume of water used per hydraulic fracture has been reported as nearly 7,375 gallons for vertical oil and gas wells prior to 1953, nearly 197,000 gallons for vertical oil and gas wells between 2000 and 2010, and nearly 3 million gallons for horizontal gas wells between 2000 and 2010.
Determining which fracking technique is appropriate for well productivity depends largely on the properties of the reservoir rock from which to extract oil or gas. If the rock is characterized by low-permeability – which refers to its ability to let substances, i.e. gas, pass through it, then the rock may be considered a source of tight gas
Tight gas is natural gas produced from reservoir rocks with such low permeability that massive hydraulic fracturing is necessary to produce the well at economic rates. This natural gas is trapped within rocks with very low permeability, in other ...
. Fracking for shale gas, which is currently also known as a source of unconventional gas
Unconventional gas is natural gas obtained from sources of production that are, in a given era and location, considered to be new and different when compared with conventional gas. Sources that are at times considered to be unconventional include ...
, involves drilling a borehole vertically until it reaches a lateral shale rock formation, at which point the drill turns to follow the rock for hundreds or thousands of feet horizontally. In contrast, conventional oil and gas sources are characterized by higher rock permeability, which naturally enables the flow of oil or gas into the wellbore with less intensive hydraulic fracturing techniques than the production of tight gas has required. The decades in development of drilling technology for conventional and unconventional oil and gas production have not only improved access to natural gas in low-permeability reservoir rocks, but also posed significant adverse impacts on environmental and public health.
The US EPA has acknowledged that toxic, carcinogenic chemicals, i.e. benzene and ethylbenzene, have been used as gelling agents in water and chemical mixtures for high volume horizontal fracturing (HVHF). Following the hydraulic fracture in HVHF, the water, chemicals, and frack fluid that return to the well's surface, called flowback or produced water, may contain radioactive materials, heavy metals, natural salts, and hydrocarbons which exist naturally in shale rock formations. Fracking chemicals, radioactive materials, heavy metals, and salts that are removed from the HVHF well by well operators are so difficult to remove from the water they are mixed with, and would so heavily pollute the water cycle
The water cycle, also known as the hydrologic cycle or the hydrological cycle, is a biogeochemical cycle that describes the continuous movement of water on, above and below the surface of the Earth. The mass of water on Earth remains fairly cons ...
, that most of the flowback is either recycled into other fracking operations or injected into deep underground wells, eliminating the water that HVHF required from the hydrologic cycle.
Historically low gas prices have delayed the nuclear renaissance
Since about 2001 the term nuclear renaissance has been used to refer to a possible nuclear power industry revival, driven by rising fossil fuel prices and new concerns about meeting greenhouse gas emission limits.
In the 2009 ''World Energy ...
, as well as the development of solar thermal energy
Solar thermal energy (STE) is a form of energy and a technology for harnessing solar energy to generate thermal energy for use in industry, and in the residential and commercial sectors.
Solar thermal collectors are classified by the United St ...
.
Added odor
Natural gas in its native state is colorless and almostodorless
The sense of smell, or olfaction, is the special sense through which smells (or odors) are perceived. The sense of smell has many functions, including detecting desirable foods, hazards, and pheromones, and plays a role in taste.
In humans, it ...
. In order to assist consumers in detecting leak
A leak is a way (usually an opening) for fluid to escape a container or fluid-containing system, such as a tank or a ship's hull, through which the contents of the container can escape or outside matter can enter the container. Leaks are usually ...
s, an odorizer with a scent similar to rotten eggs, tert-Butylthiol
''tert''-Butylthiol, also known as 2-methylpropane-2-thiol, 2-methyl-2-propanethiol, ''tert''-butyl mercaptan (TBM), and ''t''-BuSH, is an organosulfur compound with the formula (CH3)3CSH. This thiol is used as an odorant for natural gas, which ...
(t-butyl mercaptan), is added. Sometimes a related compound, thiophane, may be used in the mixture. Situations in which an odorant that is added to natural gas can be detected by analytical instrumentation, but cannot be properly detected by an observer with a normal sense of smell, have occurred in the natural gas industry. This is caused by odor masking, when one odorant overpowers the sensation of another. As of 2011, the industry is conducting research on the causes of odor masking.{{Update inline, date=October 2022
Risk of explosion
gas leak
A gas leak refers to a leak of natural gas or another gaseous product from a pipeline or other containment into any area where the gas should not be present. Gas leaks can be hazardous to health as well as the environment. Even a small leak in ...
s occur a few times each year. Individual homes, small businesses and other structures are most frequently affected when an internal leak builds up gas inside the structure. Leaks often result from excavation work, such as when contractors dig and strike pipelines, sometimes without knowing any damage resulted. Frequently, the blast is powerful enough to significantly damage a building but leave it standing. In these cases, the people inside tend to have minor to moderate injuries. Occasionally, the gas can collect in high enough quantities to cause a deadly explosion, destroying one or more buildings in the process. Many building codes now forbid the installation of gas pipes inside cavity walls or below floor boards to mitigate against this risk. Gas usually dissipates readily outdoors, but can sometimes collect in dangerous quantities if flow rates are high enough. However, considering the tens of millions of structures that use the fuel, the individual risk of using natural gas is low.
Risk of carbon monoxide inhalation
Natural gas heating systems may causecarbon monoxide poisoning
Carbon monoxide poisoning typically occurs from breathing in carbon monoxide (CO) at excessive levels. Symptoms are often described as "flu-like" and commonly include headache, dizziness, weakness, vomiting, chest pain, and confusion. Large e ...
if unvented or poorly vented. Improvements in natural gas furnace designs have greatly reduced CO poisoning concerns. Detectors
A sensor is a device that produces an output signal for the purpose of sensing a physical phenomenon.
In the broadest definition, a sensor is a device, module, machine, or subsystem that detects events or changes in its environment and sends ...
are also available that warn of carbon monoxide or explosive gases such as methane and propane.
Energy content, statistics, and pricing
{{Main, Natural gas prices {{see also, Billion cubic metres of natural gas {{Update section, date=October 2022
Quantities of natural gas are measured in standard cubic meters (cubic meter of gas at temperature {{convert, 15, °C, °F, abbr=on and pressure {{convert, 101.325, kPa, psi, abbr=on) or
{{Update section, date=October 2022
Quantities of natural gas are measured in standard cubic meters (cubic meter of gas at temperature {{convert, 15, °C, °F, abbr=on and pressure {{convert, 101.325, kPa, psi, abbr=on) or standard cubic feet
A standard cubic foot (scf) is a unit used both in the natural gas industry to represent an amount of natural gas and in other industries where other gases are used. It is the unit commonly used when following the customary system, a collection ...
(cubic foot of gas at temperature 60.0 °F and pressure {{convert, 14.73, psi, kPa, abbr=on), 1 standard cubic meter = 35.301 standard cubic feet. The gross heat of combustion of commercial quality natural gas is around {{convert, 39, MJ/m3, kWh/cuft, abbr=on, but this can vary by several percent. This is about 50 to 54 MJ/kg depending on the density. For comparison, the heat of combustion of pure methane is 37.7 MJ per standard cubic metre, or 55.5 MJ/kg.
Except in the European Union, the U.S., and Canada, natural gas is sold in gigajoule retail units. LNG (liquefied natural gas) and LPG ( liquefied petroleum gas) are traded in metric tonnes (1,000 kg) or million BTU as spot deliveries. Long term natural gas distribution contracts are signed in cubic meters, and LNG contracts are in metric tonnes. The LNG and LPG is transported by specialized transport ships, as the gas is liquified at cryogenic temperatures. The specification of each LNG/LPG cargo will usually contain the energy content, but this information is in general not available to the public. The European Union aims to cut its gas dependency on Russia by two-thirds in 2022.
In August 2015, possibly the largest natural gas discovery in history was made and notified by an Italian gas company ENI. The energy company indicated that it has unearthed a "supergiant" gas field in the Mediterranean Sea covering about {{convert, 40, sqmi. This was named the Zohr gas field and could hold a potential {{convert, 30, e12cuft, abbr=off, sp=us of natural gas. ENI said that the energy is about {{convert, 5.5, e9BOE, abbr=~. The Zohr field was found in the deep waters off the northern coast of Egypt and ENI claims that it will be the largest ever in the Mediterranean and even the world.
European Union
Gas prices for end users vary greatly across the EU. A single European energy market, one of the key objectives of the EU, should level the prices of gas in all EU member states. Moreover, it would help to resolve supply andglobal warming
In common usage, climate change describes global warming—the ongoing increase in global average temperature—and its effects on Earth's climate system. Climate change in a broader sense also includes previous long-term changes to E ...
issues, as well as strengthen relations with other Mediterranean countries and foster investments in the region. Qatar has been asked by the US to supply emergency gas to the EU in case of supply disruptions in the Russo-Ukrainian crisis.
United States
 In
In US units
United States customary units form a system of Units of measurement, measurement units commonly used in the United States and Territories of the United States, U.S. territories since being standardized and adopted in 1832. The United States cust ...
, {{convert, 1, cuft, L , adj=pre, standard , spell=in of natural gas produces around {{convert , 1028, btu , kJ, lk=in. The actual heating value when the water formed does not condense is the net heat of combustion and can be as much as 10% less.
In the United States, retail sales are often in units of therm
The therm (symbol, thm) is a non- SI unit of heat energy equal to 100,000 British thermal units (BTU), and approximately megajoules, kilowatt-hours, kilocalories and thermies. One therm is the energy content of approximately of natural gas a ...
s (th); 1 therm = 100,000 BTU. Gas sales to domestic consumers are often in units of 100 standard cubic feet
A standard cubic foot (scf) is a unit used both in the natural gas industry to represent an amount of natural gas and in other industries where other gases are used. It is the unit commonly used when following the customary system, a collection ...
(scf). Gas meter
A gas meter is a specialized flow meter, used to measure the volume of fuel gases such as natural gas and liquefied petroleum gas. Gas meters are used at residential, commercial, and industrial buildings that consume fuel gas supplied by a ga ...
s measure the volume of gas used, and this is converted to therms by multiplying the volume by the energy content of the gas used during that period, which varies slightly over time. The typical annual consumption of a single family residence is 1,000 therms or one Residential Customer Equivalent Residential Customer Equivalent (RCE) is a unit of measures used by the energy industry to denote the typical annual commodity consumption by a single-family residential customer. Also known as "RCE" for short, a single RCE represents 1,000 therms ...
(RCE). Wholesale transactions are generally done in decatherm
The therm (symbol, thm) is a non-International System of Units, SI Units of energy, unit of heat energy equal to 100,000 British thermal units (BTU), and approximately joule, megajoules, kilowatt-hours, kilocalories and thermies. One therm is the ...
s (Dth), thousand decatherms (MDth), or million decatherms (MMDth). A million decatherms is a trillion BTU, roughly a billion cubic feet of natural gas.
The price of natural gas varies greatly depending on location and type of consumer. The typical caloric value of natural gas is roughly 1,000 BTU per cubic foot, depending on gas composition. Natural gas in the United States is traded as a futures contract
In finance, a futures contract (sometimes called a futures) is a standardized legal contract to buy or sell something at a predetermined price for delivery at a specified time in the future, between parties not yet known to each other. The asset ...
on the New York Mercantile Exchange
The New York Mercantile Exchange (NYMEX) is a commodity futures exchange owned and operated by CME Group of Chicago. NYMEX is located at One North End Avenue in Brookfield Place in the Battery Park City section of Manhattan, New York City.
T ...
. Each contract is for 10,000 million BTU or {{convert, 10, e9BTU, GJ, 0, abbr=unit. Thus, if the price of gas is $10/million BTU on the NYMEX, the contract is worth $100,000.
Canada
{{Unreferenced section, date=October 2022 Canada usesmetric
Metric or metrical may refer to:
* Metric system, an internationally adopted decimal system of measurement
* An adjective indicating relation to measurement in general, or a noun describing a specific type of measurement
Mathematics
In mathem ...
measure for internal trade of petrochemical products. Consequently, natural gas is sold by the gigajoule (GJ), cubic meter (m3) or thousand cubic meters (E3m3). Distribution infrastructure and meters almost always meter volume (cubic foot or cubic meter). Some jurisdictions, such as Saskatchewan, sell gas by volume only. Other jurisdictions, such as Alberta, gas is sold by the energy content (GJ). In these areas, almost all meters for residential and small commercial customers measure volume (m3 or ft3), and billing statements include a multiplier to convert the volume to energy content of the local gas supply.
A gigajoule
The joule ( , ; symbol: J) is the unit of energy in the International System of Units (SI). It is equal to the amount of work done when a force of 1 newton displaces a mass through a distance of 1 metre in the direction of the force applied. ...
(GJ) is a measure approximately equal to {{convert, 0.5, oilbbl, L, order=flip, sigfig=1, abbr=off of oil, or {{convert, 1000, ft3, m3, disp=or, abbr=on, order=flip or 1 million BTUs of gas. The energy content of gas supply in Canada can vary from {{convert, 37, to, 43, MJ/m3, abbr=on depending on gas supply and processing between the wellhead and the customer.
Adsorbed natural gas (ANG)
Natural gas may be stored by adsorbing it to the porous solids called sorbents. The optimal condition for methane storage is at room temperature and atmospheric pressure. Pressures up to 4 MPa (about 40 times atmospheric pressure) will yield greater storage capacity. The most common sorbent used for ANG is activated carbon (AC), primarily in three forms: Activated Carbon Fiber (ACF), Powdered Activated Carbon (PAC), and activated carbon monolith.{{cite web, url=http://scopewe.com/category/chemical-engineering/ang, title=Adsorbed Natural Gas, work=scopeWe – a Virtual Engineer, access-date=11 June 2015, archive-url=https://web.archive.org/web/20131109181802/http://scopewe.com/category/chemical-engineering/ang/, archive-date=9 November 2013, url-status=deadSee also
{{Portal, Energy, Renewable energy *Associated petroleum gas
Associated petroleum gas (APG), or associated gas, is a form of natural gas which is found with deposits of petroleum, either dissolved in the oil or as a free "gas cap" above the oil in the reservoir. The gas can be utilized in a number of ways a ...
* Energy transition
* Gas oil ratio
When oil is produced to surface temperature and pressure it is usual for some natural gas to come out of solution. The gas/oil ratio (GOR) is the ratio of the volume of gas ("scf") that comes out of solution to the volume of oil — at standard ...
* Natural gas by country
This article includes a chart representing proven reserves, production, consumption, exports and imports of natural gas by country. Below the numbers there is specified which position a country holds by the corresponding parameter. Dependent te ...
* Peak gas
Peak gas is the year in which the maximum global natural gas (fossil gas) production rate will be reached, after which the rate of production will enter its terminal decline. Although demand is peaking in the United States and Europe, it continu ...
* Power to gas
Power-to-gas (often abbreviated P2G) is a technology that uses electric power to produce a gaseous fuel. When using surplus power from wind generation, the concept is sometimes called windgas.
Most P2G systems use electrolysis to produce hydrogen ...
* Renewable natural gas
Renewable natural gas (RNG), also known as sustainable natural gas (SNG) or biomethane, is a biogas which has been upgraded to a quality similar to fossil natural gas and having a methane concentration of 90% or greater. By increasing the concentr ...
* World energy resources and consumption
World energy supply and consumption is global production and preparation of fuel, generation of electricity, energy transport, and energy consumption. It is a basic part of economic activity. It includes heat, but not energy from food.
This art ...
References
{{ReflistFurther reading
* Blanchard, Charles (2020). ''The Extraction State: A History of Natural Gas in America''. U of Pittsburgh Pressonline review
External links
Global Fossil Infrastructure Tracker
Carbon Mapper Data Portal featuring methane point source data
{{Fuel gas {{Authority control {{DEFAULTSORT:Natural Gas Chinese inventions Fuel gas Fossil fuels