Inflammation on:
[Wikipedia]
[Google]
[Amazon]
Inflammation (from la, inflammatio) is part of the complex biological response of body tissues to harmful stimuli, such as
 Acute inflammation is characterized by marked vascular changes, including
Acute inflammation is characterized by marked vascular changes, including

 Various
Various
 Inflammatory abnormalities are a large group of disorders that underlie a vast variety of human diseases. The immune system is often involved with inflammatory disorders, as demonstrated in both
Inflammatory abnormalities are a large group of disorders that underlie a vast variety of human diseases. The immune system is often involved with inflammatory disorders, as demonstrated in both
The complete restoration of the inflamed tissue back to a normal status. Inflammatory measures such as vasodilation, chemical production, and leukocyte infiltration cease, and damaged
Large amounts of tissue destruction, or damage in tissues unable to regenerate, cannot be regenerated completely by the body. Fibrous
A cavity is formed containing pus, an opaque liquid containing dead white blood cells and bacteria with general debris from destroyed cells. # Chronic inflammation
In acute inflammation, if the injurious agent persists then chronic inflammation will ensue. This process, marked by inflammation lasting many days, months or even years, may lead to the formation of a
File:Acute_Appendicitis.jpg, Acute
pathogen
In biology, a pathogen ( el, πάθος, "suffering", "passion" and , "producer of") in the oldest and broadest sense, is any organism or agent that can produce disease. A pathogen may also be referred to as an infectious agent, or simply a germ ...
s, damaged cells, or irritants, and is a protective response involving immune cells
White blood cells, also called leukocytes or leucocytes, are the cells of the immune system that are involved in protecting the body against both infectious disease and foreign invaders. All white blood cells are produced and derived from mult ...
, blood vessels
The blood vessels are the components of the circulatory system that transport blood throughout the human body. These vessels transport blood cells, nutrients, and oxygen to the tissues of the body. They also take waste and carbon dioxide away f ...
, and molecular mediators. The function of inflammation is to eliminate the initial cause of cell injury, clear out necrotic
Necrosis () is a form of cell injury which results in the premature death of cells in living tissue by autolysis. Necrosis is caused by factors external to the cell or tissue, such as infection, or trauma which result in the unregulated dige ...
cells and tissues damaged from the original insult and the inflammatory process, and initiate tissue repair.
The five cardinal signs
Signs and symptoms are the observed or detectable signs, and experienced symptoms of an illness, injury, or condition. A sign for example may be a higher or lower temperature than normal, raised or lowered blood pressure or an abnormality showin ...
are heat, pain, redness, swelling, and loss of function
In biology, a mutation is an alteration in the nucleic acid sequence of the genome of an organism, virus, or extrachromosomal DNA. Viral genomes contain either DNA or RNA. Mutations result from errors during DNA or viral replication, mitos ...
(Latin ''calor'', ''dolor'', ''rubor'', ''tumor'', and ''functio laesa''). Inflammation is a generic response, and therefore it is considered as a mechanism of innate immunity
The innate, or nonspecific, immune system is one of the two main immunity strategies (the other being the adaptive immune system) in vertebrates. The innate immune system is an older evolutionary defense strategy, relatively speaking, and is the ...
, as compared to adaptive immunity
The adaptive immune system, also known as the acquired immune system, is a subsystem of the immune system that is composed of specialized, systemic cells and processes that eliminate pathogens or prevent their growth. The acquired immune system ...
, which is specific for each pathogen. Too little inflammation could lead to progressive tissue destruction by the harmful stimulus (e.g. bacteria) and compromise the survival of the organism. In contrast, too much inflammation, in the form of chronic inflammation, is associated with various diseases, such as hay fever
Allergic rhinitis, of which the seasonal type is called hay fever, is a type of inflammation in the nose that occurs when the immune system overreacts to allergens in the air. Signs and symptoms include a runny or stuffy nose, sneezing, red, i ...
, periodontal disease
Periodontal disease, also known as gum disease, is a set of inflammatory conditions affecting the tissues surrounding the teeth. In its early stage, called gingivitis, the gums become swollen and red and may bleed. It is considered the main caus ...
, atherosclerosis
Atherosclerosis is a pattern of the disease arteriosclerosis in which the wall of the artery develops abnormalities, called lesions. These lesions may lead to narrowing due to the buildup of atheroma, atheromatous plaque. At onset there are usu ...
, and osteoarthritis
Osteoarthritis (OA) is a type of degenerative joint disease that results from breakdown of joint cartilage and underlying bone which affects 1 in 7 adults in the United States. It is believed to be the fourth leading cause of disability in the w ...
.
Inflammation can be classified as either ''acute'' or ''chronic''. Acute inflammation is the initial response of the body to harmful stimuli, and is achieved by the increased movement of plasma
Plasma or plasm may refer to:
Science
* Plasma (physics), one of the four fundamental states of matter
* Plasma (mineral), a green translucent silica mineral
* Quark–gluon plasma, a state of matter in quantum chromodynamics
Biology
* Blood pla ...
and leukocyte
White blood cells, also called leukocytes or leucocytes, are the cells of the immune system that are involved in protecting the body against both infectious disease and foreign invaders. All white blood cells are produced and derived from mult ...
s (in particular granulocyte
Granulocytes are
cells in the innate immune system characterized by the presence of specific granules in their cytoplasm. Such granules distinguish them from the various agranulocytes. All myeloblastic granulocytes are polymorphonuclear. They ha ...
s) from the blood into the injured tissues. A series of biochemical events propagates and matures the inflammatory response, involving the local vascular system
The blood circulatory system is a system of organs that includes the heart, blood vessels, and blood which is circulated throughout the entire body of a human or other vertebrate. It includes the cardiovascular system, or vascular system, tha ...
, the immune system
The immune system is a network of biological processes that protects an organism from diseases. It detects and responds to a wide variety of pathogens, from viruses to parasitic worms, as well as cancer cells and objects such as wood splinte ...
, and various cells within the injured tissue. Prolonged inflammation, known as ''chronic inflammation'', leads to a progressive shift in the type of cells present at the site of inflammation, such as mononuclear cells, and is characterized by simultaneous destruction and healing
With physical trauma or disease suffered by an organism, healing involves the repairing of damaged tissue(s), organs and the biological system as a whole and resumption of (normal) functioning. Medicine includes the process by which the cells i ...
of the tissue from the inflammatory process.
Inflammation has also been classified as Type 1 and Type 2 based on the type of cytokines
Cytokines are a broad and loose category of small proteins (~5–25 kDa) important in cell signaling. Cytokines are peptides and cannot cross the lipid bilayer of cells to enter the cytoplasm. Cytokines have been shown to be involved in autocrin ...
and helper T cells
The T helper cells (Th cells), also known as CD4+ cells or CD4-positive cells, are a type of T cell that play an important role in the adaptive immune system. They aid the activity of other immune cells by releasing cytokines. They are considere ...
(Th1 and Th2) involved.
Inflammation is not a synonym for infection
An infection is the invasion of tissues by pathogens, their multiplication, and the reaction of host tissues to the infectious agent and the toxins they produce. An infectious disease, also known as a transmissible disease or communicable dise ...
. Infection describes the interaction between the action of microbial invasion and the reaction of the body's inflammatory response—the two components are considered together when discussing an infection, and the word is used to imply a microbial invasive cause for the observed inflammatory reaction. Inflammation, on the other hand, describes purely the body's immunovascular response—whatever the cause may be. But because of how often the two are correlated
In statistics, correlation or dependence is any statistical relationship, whether causal or not, between two random variables or bivariate data. Although in the broadest sense, "correlation" may indicate any type of association, in statistics ...
, words ending in the suffix ''-itis
Inflammation (from la, inflammatio) is part of the complex biological response of body tissues to harmful stimuli, such as pathogens, damaged cells, or irritants, and is a protective response involving immune cells, blood vessels, and molec ...
'' (which refers to inflammation) are sometimes informally described as referring to infection. For example, the word ''urethritis
Urethritis is the inflammation of the urethra. The most common symptoms include painful or difficult urination and urethral discharge. It is a commonly treatable condition usually caused by infection with bacteria. This bacterial infection is oft ...
'' strictly means only "urethral inflammation", but clinical health care provider
A health care provider is an individual health professional or a health facility organization licensed to provide health care diagnosis and treatment services including medication, surgery and medical devices. Health care providers often receive ...
s usually discuss urethritis as a urethral infection because urethral microbial invasion is the most common cause of urethritis.
However, the inflammation–infection distinction becomes crucial for situations in pathology
Pathology is the study of the causes and effects of disease or injury. The word ''pathology'' also refers to the study of disease in general, incorporating a wide range of biology research fields and medical practices. However, when used in ...
and medical diagnosis
Medical diagnosis (abbreviated Dx, Dx, or Ds) is the process of determining which disease or condition explains a person's symptoms and signs. It is most often referred to as diagnosis with the medical context being implicit. The information re ...
where inflammation is not driven by microbial invasion, such as the cases of atherosclerosis
Atherosclerosis is a pattern of the disease arteriosclerosis in which the wall of the artery develops abnormalities, called lesions. These lesions may lead to narrowing due to the buildup of atheroma, atheromatous plaque. At onset there are usu ...
, trauma
Trauma most often refers to:
* Major trauma, in physical medicine, severe physical injury caused by an external source
* Psychological trauma, a type of damage to the psyche that occurs as a result of a severely distressing event
*Traumatic i ...
, ischemia
Ischemia or ischaemia is a restriction in blood supply to any tissue, muscle group, or organ of the body, causing a shortage of oxygen that is needed for cellular metabolism (to keep tissue alive). Ischemia is generally caused by problems wi ...
, and autoimmune diseases
An autoimmune disease is a condition arising from an abnormal immune response to a functioning body part. At least 80 types of autoimmune diseases have been identified, with some evidence suggesting that there may be more than 100 types. Nearly a ...
(including type III hypersensitivity
Type III hypersensitivity, in the Gell and Coombs classification of allergic reactions, occurs when there is accumulation of immune complexes (antigen-antibody complexes) that have not been adequately cleared by innate immune cells, giving rise ...
).
Causes
Types
*Appendicitis
Appendicitis is inflammation of the appendix. Symptoms commonly include right lower abdominal pain, nausea, vomiting, and decreased appetite. However, approximately 40% of people do not have these typical symptoms. Severe complications of a rup ...
* Bursitis
Bursitis is the inflammation of one or more bursae (fluid filled sacs) of synovial fluid in the body. They are lined with a synovial membrane that secretes a lubricating synovial fluid. There are more than 150 bursae in the human body. The bursa ...
* Colitis
Colitis is swelling or inflammation of the large intestine ( colon). Colitis may be acute and self-limited or long-term. It broadly fits into the category of digestive diseases.
In a medical context, the label ''colitis'' (without qualification) ...
* Cystitis
A urinary tract infection (UTI) is an infection that affects part of the urinary tract. When it affects the lower urinary tract it is known as a bladder infection (cystitis) and when it affects the upper urinary tract it is known as a kidney ...
* Dermatitis
Dermatitis is inflammation of the skin, typically characterized by itchiness, redness and a rash. In cases of short duration, there may be small blisters, while in long-term cases the skin may become thickened. The area of skin involved can v ...
* Epididymitis
Epididymitis is a medical condition characterized by inflammation of the epididymis, a curved structure at the back of the testicle. Onset of pain is typically over a day or two. The pain may improve with raising the testicle. Other symptoms may ...
* Encephalitis
Encephalitis is inflammation of the brain. The severity can be variable with symptoms including reduction or alteration in consciousness, headache, fever, confusion, a stiff neck, and vomiting. Complications may include seizures, hallucinations, ...
* Gingivitis
Gingivitis is a non-destructive disease that causes inflammation of the gums. The most common form of gingivitis, and the most common form of periodontal disease overall, is in response to bacterial biofilms (also called plaque) that is attached ...
* Meningitis
Meningitis is acute or chronic inflammation of the protective membranes covering the brain and spinal cord, collectively called the meninges. The most common symptoms are fever, headache, and neck stiffness. Other symptoms include confusion or ...
* Myelitis
Myelitis is inflammation of the spinal cord which can disrupt the normal responses from the brain to the rest of the body, and from the rest of the body to the brain. Inflammation in the spinal cord, can cause the myelin and axon to be damaged re ...
* Nephritis
Nephritis is inflammation of the kidneys and may involve the glomeruli, tubules, or interstitial tissue surrounding the glomeruli and tubules. It is one of several different types of nephropathy.
Types
* Glomerulonephritis is inflammation of th ...
* Neuritis
Neuritis () is inflammation of a nerve or the general inflammation of the peripheral nervous system. Inflammation, and frequently concomitant demyelination, cause impaired transmission of neural signals and leads to aberrant nerve function. Neurit ...
* Pancreatitis
Pancreatitis is a condition characterized by inflammation of the pancreas. The pancreas is a large organ behind the stomach that produces digestive enzymes and a number of hormones. There are two main types: acute pancreatitis, and chronic pancr ...
* Periodontitis
Periodontal disease, also known as gum disease, is a set of inflammatory conditions affecting the tissues surrounding the teeth. In its early stage, called gingivitis, the gums become swollen and red and may bleed. It is considered the main caus ...
* Pharyngitis
Pharyngitis is inflammation of the back of the throat, known as the pharynx. It typically results in a sore throat and fever. Other symptoms may include a runny nose, cough, headache, difficulty swallowing, swollen lymph nodes, and a hoarse voice ...
* Phlebitis
Phlebitis (or Venitis) is inflammation of a vein, usually in the legs. It most commonly occurs in superficial veins. Phlebitis often occurs in conjunction with thrombosis and is then called thrombophlebitis or superficial thrombophlebitis. Unlike ...
* Prostatitis
Prostatitis is an umbrella term for a variety of medical conditions that incorporate bacterial and non-bacterial origin illnesses in the pelvic region. In contrast with the plain meaning of the word (which means "inflammation of the prostate"), the ...
* RSD/CRPS
* Rhinitis
Rhinitis, also known as coryza, is irritation and inflammation of the mucous membrane inside the nose. Common symptoms are a stuffy nose, runny nose, sneezing, and post-nasal drip.
The inflammation is caused by viruses, bacteria, irritants o ...
* Sinusitis
Sinusitis, also known as rhinosinusitis, is inflammation of the nasal mucosa, mucous membranes that line the paranasal sinuses, sinuses resulting in symptoms that may include thick Mucus#Respiratory system, nasal mucus, a nasal congestion, plugg ...
* Tendonitis
Tendinopathy, a type of tendon disorder that results in pain, swelling, and impaired function. The pain is typically worse with movement. It most commonly occurs around the shoulder (rotator cuff tendinitis, biceps tendinitis), elbow (tennis elb ...
* Tonsillitis
Tonsillitis is inflammation of the tonsils in the upper part of the throat. It can be acute or chronic. Acute tonsillitis typically has a rapid onset. Symptoms may include sore throat, fever, enlargement of the tonsils, trouble swallowing, and en ...
* Urethritis
Urethritis is the inflammation of the urethra. The most common symptoms include painful or difficult urination and urethral discharge. It is a commonly treatable condition usually caused by infection with bacteria. This bacterial infection is oft ...
* Vasculitis
Vasculitis is a group of disorders that destroy blood vessels by inflammation. Both arteries and veins are affected. Lymphangitis (inflammation of lymphatic vessels) is sometimes considered a type of vasculitis. Vasculitis is primarily caused b ...
* Vaginitis
Vaginitis, also known as vulvovaginitis, is inflammation of the vagina and vulva. Symptoms may include itching, burning, pain, discharge, and a bad smell. Certain types of vaginitis may result in complications during pregnancy.
The three main ...
Acute
Acute inflammation occurs immediately upon injury, lasting only a few days.Cytokine
Cytokines are a broad and loose category of small proteins (~5–25 kDa) important in cell signaling. Cytokines are peptides and cannot cross the lipid bilayer of cells to enter the cytoplasm. Cytokines have been shown to be involved in autocrin ...
s and chemokine
Chemokines (), or chemotactic cytokines, are a family of small cytokines or signaling proteins secreted by cells that induce directional movement of leukocytes, as well as other cell types, including endothelial and epithelial cells. In additio ...
s promote the migration of neutrophil
Neutrophils (also known as neutrocytes or heterophils) are the most abundant type of granulocytes and make up 40% to 70% of all white blood cells in humans. They form an essential part of the innate immune system, with their functions varying in ...
s and macrophage
Macrophages (abbreviated as M φ, MΦ or MP) ( el, large eaters, from Greek ''μακρός'' (') = large, ''φαγεῖν'' (') = to eat) are a type of white blood cell of the immune system that engulfs and digests pathogens, such as cancer cel ...
s to the site of inflammation. Pathogens, allergens, toxins, burns, and frostbite are some of the typical causes of acute inflammation. Toll-like receptor
Toll-like receptors (TLRs) are a class of proteins that play a key role in the innate immune system. They are Bitopic protein, single-pass membrane-spanning Receptor (biochemistry), receptors usually expressed on sentinel cells such as macrophage ...
s (TLRs) recognize microbial pathogens. Acute inflammation can be a defensive mechanism to protect tissues against injury. Inflammation lasting 2–6 weeks is designated subacute inflammation.
Cardinal signs
Acute inflammation is a short-term process, usually appearing within a few minutes or hours and begins to cease upon the removal of the injurious stimulus. It involves a coordinated and systemic mobilization response locally of various immune, endocrine and neurological mediators of acute inflammation. In a normal healthy response, it becomes activated, clears the pathogen and begins a repair process and then ceases. It is characterized by fivecardinal signs
Signs and symptoms are the observed or detectable signs, and experienced symptoms of an illness, injury, or condition. A sign for example may be a higher or lower temperature than normal, raised or lowered blood pressure or an abnormality showin ...
:
The traditional names for signs of inflammation come from Latin:
* Dolor
Dolor may refer to:
* Pain
* Suffering
* The unit of measurement in utilitarianism, see Felicific calculus#Hedons and dolors
* ''Dolor'' (sculpture), a work by Clemente Islas Allende in Mexico City
See also
* Dolors
Dolors is a Catalan feminin ...
(pain
Pain is a distressing feeling often caused by intense or damaging stimuli. The International Association for the Study of Pain defines pain as "an unpleasant sensory and emotional experience associated with, or resembling that associated with, ...
)
* Calor (heat
In thermodynamics, heat is defined as the form of energy crossing the boundary of a thermodynamic system by virtue of a temperature difference across the boundary. A thermodynamic system does not ''contain'' heat. Nevertheless, the term is al ...
)
* Rubor
Flushing is to become markedly red in the face and often other areas of the skin, from various physiological conditions. Flushing is generally distinguished, despite a close physiological relation between them, from blushing, which is milder, gen ...
(redness)
* Tumor ( swelling)
* Functio laesa (loss of function)
The first four (classical signs) were described by Celsus
Celsus (; grc-x-hellen, Κέλσος, ''Kélsos''; ) was a 2nd-century Greek philosopher and opponent of early Christianity. His literary work, ''The True Word'' (also ''Account'', ''Doctrine'' or ''Discourse''; Greek: grc-x-hellen, Λόγ� ...
(–38 AD), while ''loss of function'' was probably added later by Galen
Aelius Galenus or Claudius Galenus ( el, Κλαύδιος Γαληνός; September 129 – c. AD 216), often Anglicized as Galen () or Galen of Pergamon, was a Greek physician, surgeon and philosopher in the Roman Empire. Considered to be one of ...
. However, the addition of this fifth sign has also been ascribed to Thomas Sydenham
Thomas Sydenham (10 September 1624 – 29 December 1689) was an English physician. He was the author of ''Observationes Medicae'' which became a standard textbook of medicine for two centuries so that he became known as 'The English Hippocrate ...
and Virchow.
Redness and heat are due to increased blood flow at body core temperature to the inflamed site; swelling is caused by accumulation of fluid; pain
Pain is a distressing feeling often caused by intense or damaging stimuli. The International Association for the Study of Pain defines pain as "an unpleasant sensory and emotional experience associated with, or resembling that associated with, ...
is due to the release of chemicals such as bradykinin and histamine that stimulate nerve endings. Loss of function has multiple causes.
Acute inflammation of the lung (usually as in response to pneumonia
Pneumonia is an inflammatory condition of the lung primarily affecting the small air sacs known as alveoli. Symptoms typically include some combination of productive or dry cough, chest pain, fever, and difficulty breathing. The severity ...
) does not cause pain unless the inflammation involves the parietal pleura
The pulmonary pleurae (''sing.'' pleura) are the two opposing layers of serous membrane overlying the lungs and the inside of the surrounding chest walls.
The inner pleura, called the visceral pleura, covers the surface of each lung and dips bet ...
, which does have pain-sensitive nerve endings.
Acute process
The process of acute inflammation is initiated by resident immune cells already present in the involved tissue, mainly residentmacrophages
Macrophages (abbreviated as M φ, MΦ or MP) ( el, large eaters, from Greek ''μακρός'' (') = large, ''φαγεῖν'' (') = to eat) are a type of white blood cell of the immune system that engulfs and digests pathogens, such as cancer ce ...
, dendritic cells
Dendritic cells (DCs) are antigen-presenting cells (also known as ''accessory cells'') of the mammalian immune system. Their main function is to process antigen material and present it on the cell surface to the T cells of the immune system. The ...
, histiocytes
A histiocyte is a vertebrate cell that is part of the mononuclear phagocyte system (also known as the reticuloendothelial system or lymphoreticular system). The mononuclear phagocytic system is part of the organism's immune system. The histiocyt ...
, Kupffer cells
Kupffer cells, also known as stellate macrophages and Kupffer–Browicz cells, are specialized cells localized in the liver within the lumen of the liver sinusoids and are adhesive to their endothelial cells which make up the blood vessel walls. Ku ...
and mast cell
A mast cell (also known as a mastocyte or a labrocyte) is a resident cell of connective tissue that contains many granules rich in histamine and heparin. Specifically, it is a type of granulocyte derived from the myeloid stem cell that is a par ...
s. These cells possess surface receptors known as ''pattern recognition receptor
Pattern recognition receptors (PRRs) play a crucial role in the proper function of the innate immune system. PRRs are germline-encoded host sensors, which detect molecules typical for the pathogens. They are proteins expressed, mainly, by cells of ...
s'' (PRRs), which recognize (i.e., bind) two subclasses of molecules: pathogen-associated molecular pattern
Pathogen-associated molecular patterns (PAMPs) are small molecular motifs conserved within a class of microbes. They are recognized by toll-like receptors (TLRs) and other pattern recognition receptors (PRRs) in both plants and animals. A vast arra ...
s (PAMPs) and damage-associated molecular pattern
Damage-associated molecular patterns (DAMPs) are molecules within cells that are a component of the innate immune response released from damaged or dying cells due to trauma or an infection by a pathogen. They are also known as danger-associated m ...
s (DAMPs). PAMPs are compounds that are associated with various pathogen
In biology, a pathogen ( el, πάθος, "suffering", "passion" and , "producer of") in the oldest and broadest sense, is any organism or agent that can produce disease. A pathogen may also be referred to as an infectious agent, or simply a germ ...
s, but which are distinguishable from host molecules. DAMPs are compounds that are associated with host-related injury and cell damage.
At the onset of an infection, burn, or other injuries, these cells undergo activation (one of the PRRs recognize a PAMP or DAMP) and release inflammatory mediators responsible for the clinical signs of inflammation. Vasodilation and its resulting increased blood flow causes the redness (''rubor'') and increased heat (''calor''). Increased permeability of the blood vessels results in an exudation (leakage) of plasma
Plasma or plasm may refer to:
Science
* Plasma (physics), one of the four fundamental states of matter
* Plasma (mineral), a green translucent silica mineral
* Quark–gluon plasma, a state of matter in quantum chromodynamics
Biology
* Blood pla ...
proteins and fluid into the tissue (edema
Edema, also spelled oedema, and also known as fluid retention, dropsy, hydropsy and swelling, is the build-up of fluid in the body's Tissue (biology), tissue. Most commonly, the legs or arms are affected. Symptoms may include skin which feels t ...
), which manifests itself as swelling (''tumor''). Some of the released mediators such as bradykinin
Bradykinin (BK) (Greek brady-, slow; -kinin, kīn(eîn) to move) is a peptide that promotes inflammation. It causes arterioles to dilate (enlarge) via the release of prostacyclin, nitric oxide, and endothelium-derived hyperpolarizing factor and ...
increase the sensitivity to pain (hyperalgesia
Hyperalgesia ( or ; 'hyper' from Greek ὑπέρ (huper, “over”), '-algesia' from Greek algos, ἄλγος (pain)) is an abnormally increased sensitivity to pain, which may be caused by damage to nociceptors or peripheral nerves and can ...
, ''dolor''). The mediator molecules also alter the blood vessels to permit the migration of leukocytes, mainly neutrophils
Neutrophils (also known as neutrocytes or heterophils) are the most abundant type of granulocytes and make up 40% to 70% of all white blood cells in humans. They form an essential part of the innate immune system, with their functions varying in ...
and macrophages
Macrophages (abbreviated as M φ, MΦ or MP) ( el, large eaters, from Greek ''μακρός'' (') = large, ''φαγεῖν'' (') = to eat) are a type of white blood cell of the immune system that engulfs and digests pathogens, such as cancer ce ...
, to flow out of the blood vessels (extravasation) and into the tissue. The neutrophils migrate along a chemotactic
Chemotaxis (from '' chemo-'' + ''taxis'') is the movement of an organism or entity in response to a chemical stimulus. Somatic cells, bacteria, and other single-cell or multicellular organisms direct their movements according to certain chemical ...
gradient created by the local cells to reach the site of injury. The loss of function (''functio laesa'') is probably the result of a neurological reflex in response to pain.
In addition to cell-derived mediators, several acellular biochemical cascade systems—consisting of preformed plasma proteins—act in parallel to initiate and propagate the inflammatory response. These include the complement system
The complement system, also known as complement cascade, is a part of the immune system that enhances (complements) the ability of antibodies and phagocytic cells to clear microbes and damaged cells from an organism, promote inflammation, and at ...
activated by bacteria and the coagulation
Coagulation, also known as clotting, is the process by which blood changes from a liquid to a gel, forming a blood clot. It potentially results in hemostasis, the cessation of blood loss from a damaged vessel, followed by repair. The mechanism o ...
and fibrinolysis system
Fibrinolysis is a process that prevents blood clots from growing and becoming problematic. Primary fibrinolysis is a normal body process, while secondary fibrinolysis is the breakdown of clots due to a medicine, a medical disorder, or some other c ...
s activated by necrosis
Necrosis () is a form of cell injury which results in the premature death of cells in living tissue by autolysis. Necrosis is caused by factors external to the cell or tissue, such as infection, or trauma which result in the unregulated dige ...
(e.g., burn, trauma).
Acute inflammation may be regarded as the first line of defense against injury. Acute inflammatory response requires constant stimulation to be sustained. Inflammatory mediators are short-lived and are quickly degraded in the tissue. Hence, acute inflammation begins to cease once the stimulus has been removed.
Chronic
Chronic inflammation is inflammation that lasts for months or years. Macrophages,lymphocytes
A lymphocyte is a type of white blood cell (leukocyte) in the immune system of most vertebrates. Lymphocytes include natural killer cells (which function in cell-mediated, cytotoxic innate immunity), T cells (for cell-mediated, cytotoxic adap ...
, and plasma cells
Plasma cells, also called plasma B cells or effector B cells, are white blood cells that originate in the lymphoid organs as B lymphocytes and secrete large quantities of proteins called antibodies in response to being presented specific substan ...
predominate in chronic inflammation, in contrast to the neutrophils that predominate in acute inflammation. Diabetes
Diabetes, also known as diabetes mellitus, is a group of metabolic disorders characterized by a high blood sugar level ( hyperglycemia) over a prolonged period of time. Symptoms often include frequent urination, increased thirst and increased ap ...
, cardiovascular disease
Cardiovascular disease (CVD) is a class of diseases that involve the heart or blood vessels. CVD includes coronary artery diseases (CAD) such as angina and myocardial infarction (commonly known as a heart attack). Other CVDs include stroke, h ...
, allergies
Allergies, also known as allergic diseases, refer a number of conditions caused by the hypersensitivity of the immune system to typically harmless substances in the environment. These diseases include hay fever, food allergies, atopic derma ...
, and chronic obstructive pulmonary disease
Chronic obstructive pulmonary disease (COPD) is a type of progressive lung disease characterized by long-term respiratory symptoms and airflow limitation. The main symptoms include shortness of breath and a cough, which may or may not produce ...
(COPD) are examples of diseases mediated by chronic inflammation. Obesity
Obesity is a medical condition, sometimes considered a disease, in which excess body fat has accumulated to such an extent that it may negatively affect health. People are classified as obese when their body mass index (BMI)—a person's we ...
, smoking, stress and insufficient diet are some of the factors that promote chronic inflammation. A 2014 study reported that 60% of Americans had at least one chronic inflammatory condition, and 42% had more than one.
Cardinal signs
Common signs and symptoms that develop during chronic inflammation are: * Body pain, arthralgia, myalgia * Chronic fatigue and insomnia * Depression, anxiety and mood disorders * Gastrointestinal complications such as constipation, diarrhea, and acid reflux * Weight gain or loss * Frequent infectionsVascular component
Vasodilation and increased permeability
As defined, acute inflammation is an immunovascular response to inflammatory stimuli. This means acute inflammation can be broadly divided into a vascular phase that occurs first, followed by a cellular phase involving immune cells (more specifically myeloidgranulocytes
Granulocytes are
cells in the innate immune system characterized by the presence of specific granules in their cytoplasm. Such granules distinguish them from the various agranulocytes. All myeloblastic granulocytes are polymorphonuclear. They hav ...
in the acute setting). The vascular component of acute inflammation involves the movement of plasma fluid, containing important protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, respo ...
s such as fibrin
Fibrin (also called Factor Ia) is a fibrous, non-globular protein involved in the clotting of blood. It is formed by the action of the protease thrombin on fibrinogen, which causes it to polymerize. The polymerized fibrin, together with platele ...
and immunoglobulin
An antibody (Ab), also known as an immunoglobulin (Ig), is a large, Y-shaped protein used by the immune system to identify and neutralize foreign objects such as pathogenic bacteria and viruses. The antibody recognizes a unique molecule of the ...
s (antibodies
An antibody (Ab), also known as an immunoglobulin (Ig), is a large, Y-shaped protein used by the immune system to identify and neutralize foreign objects such as pathogenic bacteria and viruses. The antibody recognizes a unique molecule of the ...
), into inflamed tissue.
Upon contact with PAMPs, tissue macrophages
Macrophages (abbreviated as M φ, MΦ or MP) ( el, large eaters, from Greek ''μακρός'' (') = large, ''φαγεῖν'' (') = to eat) are a type of white blood cell of the immune system that engulfs and digests pathogens, such as cancer ce ...
and mastocytes
A mast cell (also known as a mastocyte or a labrocyte) is a resident cell of connective tissue that contains many granules rich in histamine and heparin. Specifically, it is a type of granulocyte derived from the myeloid stem cell that is a p ...
release vasoactive amines such as histamine
Histamine is an organic nitrogenous compound involved in local immune responses, as well as regulating physiological functions in the gut and acting as a neurotransmitter for the brain, spinal cord, and uterus. Since histamine was discovered in ...
and serotonin
Serotonin () or 5-hydroxytryptamine (5-HT) is a monoamine neurotransmitter. Its biological function is complex and multifaceted, modulating mood, cognition, reward, learning, memory, and numerous physiological processes such as vomiting and vas ...
, as well as eicosanoids
Eicosanoids are signaling molecules made by the enzymatic or non-enzymatic oxidation of arachidonic acid or other polyunsaturated fatty acids (PUFAs) that are, similar to arachidonic acid, around 20 carbon units in length. Eicosanoids are a sub-c ...
such as prostaglandin E2
Prostaglandin E2 (PGE2), also known as dinoprostone, is a naturally occurring prostaglandin with oxytocic properties that is used as a medication. Dinoprostone is used in labor induction, bleeding after delivery, termination of pregnancy, and ...
and leukotriene B4
Leukotriene B4 (LTB4) is a leukotriene involved in inflammation. It has been shown to promote insulin resistance in obese mice.
Biochemistry
Leukotriene B4 (LTB4) is a leukotriene involved in inflammation. It is produced from leukocytes in r ...
to remodel the local vasculature. Macrophages and endothelial cells release nitric oxide
Nitric oxide (nitrogen oxide or nitrogen monoxide) is a colorless gas with the formula . It is one of the principal oxides of nitrogen. Nitric oxide is a free radical: it has an unpaired electron, which is sometimes denoted by a dot in its che ...
. These mediators vasodilate and permeabilize the blood vessel
The blood vessels are the components of the circulatory system that transport blood throughout the human body. These vessels transport blood cells, nutrients, and oxygen to the tissues of the body. They also take waste and carbon dioxide away ...
s, which results in the net distribution of blood plasma
Blood plasma is a light amber-colored liquid component of blood in which blood cells are absent, but contains proteins and other constituents of whole blood in suspension. It makes up about 55% of the body's total blood volume. It is the intra ...
from the vessel into the tissue space. The increased collection of fluid into the tissue causes it to swell (edema
Edema, also spelled oedema, and also known as fluid retention, dropsy, hydropsy and swelling, is the build-up of fluid in the body's Tissue (biology), tissue. Most commonly, the legs or arms are affected. Symptoms may include skin which feels t ...
). This exuded tissue fluid contains various antimicrobial mediators from the plasma such as complement
A complement is something that completes something else.
Complement may refer specifically to:
The arts
* Complement (music), an interval that, when added to another, spans an octave
** Aggregate complementation, the separation of pitch-class ...
, lysozyme
Lysozyme (EC 3.2.1.17, muramidase, ''N''-acetylmuramide glycanhydrolase; systematic name peptidoglycan ''N''-acetylmuramoylhydrolase) is an antimicrobial enzyme produced by animals that forms part of the innate immune system. It is a glycoside ...
, antibodies
An antibody (Ab), also known as an immunoglobulin (Ig), is a large, Y-shaped protein used by the immune system to identify and neutralize foreign objects such as pathogenic bacteria and viruses. The antibody recognizes a unique molecule of the ...
, which can immediately deal damage to microbes, and opsonise the microbes in preparation for the cellular phase. If the inflammatory stimulus is a lacerating wound, exuded platelet
Platelets, also called thrombocytes (from Greek θρόμβος, "clot" and κύτος, "cell"), are a component of blood whose function (along with the coagulation factors) is to react to bleeding from blood vessel injury by clumping, thereby ini ...
s, coagulants, plasmin
Plasmin is an important enzyme () present in blood that degrades many blood plasma proteins, including fibrin clots. The degradation of fibrin is termed fibrinolysis. In humans, the plasmin protein (in the zymogen form of plasminogen) is encoded ...
and kinin A kinin is any of various structurally related polypeptides, such as bradykinin and kallidin. They are members of the autacoid family. Kinins are peptides that are cleaved from kininogens by the process of kallikreins. Kallikreins activate kinins wh ...
s can clot
A thrombus (plural thrombi), colloquially called a blood clot, is the final product of the blood coagulation step in hemostasis. There are two components to a thrombus: aggregated platelets and red blood cells that form a plug, and a mesh of c ...
the wounded area and provide haemostasis
In biology, hemostasis American and British English spelling differences#ae and oe, or haemostasis is a process to prevent and stop bleeding, meaning to keep blood within a damaged blood vessel (the opposite of hemostasis is Bleeding, hemorrhage). ...
in the first instance. These clotting mediators also provide a structural staging framework at the inflammatory tissue site in the form of a fibrin
Fibrin (also called Factor Ia) is a fibrous, non-globular protein involved in the clotting of blood. It is formed by the action of the protease thrombin on fibrinogen, which causes it to polymerize. The polymerized fibrin, together with platele ...
lattice – as would construction scaffolding
Scaffolding, also called scaffold or staging, is a temporary structure used to support a work crew and materials to aid in the construction, maintenance and repair of buildings, bridges and all other man-made structures. Scaffolds are widely use ...
at a construction site – for the purpose of aiding phagocytic debridement and wound repair
Wound healing refers to a living organism's replacement of destroyed or damaged tissue by newly produced tissue.
In undamaged skin, the epidermis (surface, epithelial layer) and dermis (deeper, connective layer) form a protective barrier agains ...
later on. Some of the exuded tissue fluid is also funneled by lymphatics
The lymphatic vessels (or lymph vessels or lymphatics) are thin-walled vessels (tubes), structured like blood vessels, that carry lymph. As part of the lymphatic system, lymph vessels are complementary to the cardiovascular system. Lymph vessel ...
to the regional lymph nodes, flushing bacteria along to start the recognition and attack phase of the adaptive immune system
The adaptive immune system, also known as the acquired immune system, is a subsystem of the immune system that is composed of specialized, systemic cells and processes that eliminate pathogens or prevent their growth. The acquired immune system ...
.
vasodilation
Vasodilation is the widening of blood vessels. It results from relaxation of smooth muscle cells within the vessel walls, in particular in the large veins, large arteries, and smaller arterioles. The process is the opposite of vasoconstriction, ...
, increased permeability and increased blood flow, which are induced by the actions of various inflammatory mediators. Vasodilation occurs first at the arteriole
An arteriole is a small-diameter blood vessel in the microcirculation that extends and branches out from an artery and leads to capillaries.
Arterioles have muscular walls (usually only one to two layers of smooth muscle cells) and are the primar ...
level, progressing to the capillary
A capillary is a small blood vessel from 5 to 10 micrometres (μm) in diameter. Capillaries are composed of only the tunica intima, consisting of a thin wall of simple squamous endothelial cells. They are the smallest blood vessels in the body: ...
level, and brings about a net increase in the amount of blood present, causing the redness and heat of inflammation. Increased permeability of the vessels results in the movement of plasma
Plasma or plasm may refer to:
Science
* Plasma (physics), one of the four fundamental states of matter
* Plasma (mineral), a green translucent silica mineral
* Quark–gluon plasma, a state of matter in quantum chromodynamics
Biology
* Blood pla ...
into the tissues, with resultant stasis due to the increase in the concentration of the cells within blood – a condition characterized by enlarged vessels packed with cells. Stasis allows leukocytes
White blood cells, also called leukocytes or leucocytes, are the cells of the immune system that are involved in protecting the body against both infectious disease and foreign invaders. All white blood cells are produced and derived from mult ...
to marginate (move) along the endothelium
The endothelium is a single layer of squamous endothelial cells that line the interior surface of blood vessels and lymphatic vessels. The endothelium forms an interface between circulating blood or lymph in the lumen and the rest of the vessel ...
, a process critical to their recruitment into the tissues. Normal flowing blood prevents this, as the shearing force
In solid mechanics, shearing forces are unaligned forces acting on one part of a Rigid body, body in a specific direction, and another part of the body in the opposite direction. When the forces are collinear (aligned with each other), they ...
along the periphery of the vessels moves cells in the blood into the middle of the vessel.
Plasma cascade systems
* Thecomplement system
The complement system, also known as complement cascade, is a part of the immune system that enhances (complements) the ability of antibodies and phagocytic cells to clear microbes and damaged cells from an organism, promote inflammation, and at ...
, when activated, creates a cascade of chemical reactions that promotes opsonization
Opsonins are extracellular proteins that, when bound to substances or cells, induce phagocytes to phagocytose the substances or cells with the opsonins bound. Thus, opsonins act as tags to label things in the body that should be phagocytosed (i.e. ...
, chemotaxis
Chemotaxis (from '' chemo-'' + ''taxis'') is the movement of an organism or entity in response to a chemical stimulus. Somatic cells, bacteria, and other single-cell or multicellular organisms direct their movements according to certain chemica ...
, and agglutination
In linguistics, agglutination is a morphological process in which words are formed by stringing together morphemes, each of which corresponds to a single syntactic feature. Languages that use agglutination widely are called agglutinative lang ...
, and produces the MAC.
* The kinin system A kinin is any of various structurally related polypeptides, such as bradykinin and kallidin. They are members of the autacoid family. Kinins are peptides that are cleaved from kininogens by the process of kallikreins. Kallikreins activate kinins wh ...
generates proteins capable of sustaining vasodilation and other physical inflammatory effects.
* The coagulation system
Coagulation, also known as clotting, is the process by which blood changes from a liquid to a gel, forming a blood clot. It potentially results in hemostasis, the cessation of blood loss from a damaged vessel, followed by repair. The mechanism o ...
or ''clotting cascade'', which forms a protective protein mesh over sites of injury.
* The fibrinolysis system
Fibrinolysis is a process that prevents blood clots from growing and becoming problematic. Primary fibrinolysis is a normal body process, while secondary fibrinolysis is the breakdown of clots due to a medicine, a medical disorder, or some other c ...
, which acts in opposition to the ''coagulation system'', to counterbalance clotting and generate several other inflammatory mediators.
Plasma-derived mediators
Cellular component
The ''cellular component'' involvesleukocyte
White blood cells, also called leukocytes or leucocytes, are the cells of the immune system that are involved in protecting the body against both infectious disease and foreign invaders. All white blood cells are produced and derived from mult ...
s, which normally reside in blood and must move into the inflamed tissue via ''extravasation'' to aid in inflammation. Some act as phagocyte
Phagocytes are cells that protect the body by ingesting harmful foreign particles, bacteria, and dead or dying cells. Their name comes from the Greek ', "to eat" or "devour", and "-cyte", the suffix in biology denoting "cell", from the Greek ...
s, ingesting bacteria
Bacteria (; singular: bacterium) are ubiquitous, mostly free-living organisms often consisting of one biological cell. They constitute a large domain of prokaryotic microorganisms. Typically a few micrometres in length, bacteria were among ...
, viruses, and cellular debris. Others release enzymatic granules that damage pathogenic invaders. Leukocytes also release inflammatory mediators that develop and maintain the inflammatory response. In general, acute inflammation is mediated by granulocyte
Granulocytes are
cells in the innate immune system characterized by the presence of specific granules in their cytoplasm. Such granules distinguish them from the various agranulocytes. All myeloblastic granulocytes are polymorphonuclear. They ha ...
s, whereas chronic inflammation is mediated by mononuclear cells such as monocyte
Monocytes are a type of leukocyte or white blood cell. They are the largest type of leukocyte in blood and can differentiate into macrophages and conventional dendritic cells. As a part of the vertebrate innate immune system monocytes also inf ...
s and lymphocyte
A lymphocyte is a type of white blood cell (leukocyte) in the immune system of most vertebrates. Lymphocytes include natural killer cells (which function in cell-mediated, cytotoxic innate immunity), T cells (for cell-mediated, cytotoxic ad ...
s.
Leukocyte extravasation
leukocyte
White blood cells, also called leukocytes or leucocytes, are the cells of the immune system that are involved in protecting the body against both infectious disease and foreign invaders. All white blood cells are produced and derived from mult ...
s, particularly neutrophils, are critically involved in the initiation and maintenance of inflammation. These cells must be able to move to the site of injury from their usual location in the blood, therefore mechanisms exist to recruit and direct leukocytes to the appropriate place. The process of leukocyte movement from the blood to the tissues through the blood vessels is known as ''extravasation'' and can be broadly divided up into a number of steps:
# Leukocyte margination and endothelial adhesion: The white blood cells within the vessels which are generally centrally located move peripherally towards the walls of the vessels. Activated macrophages in the tissue release cytokines
Cytokines are a broad and loose category of small proteins (~5–25 kDa) important in cell signaling. Cytokines are peptides and cannot cross the lipid bilayer of cells to enter the cytoplasm. Cytokines have been shown to be involved in autocrin ...
such as IL-1 and TNFα
Tumor necrosis factor (TNF, cachexin, or cachectin; formerly known as tumor necrosis factor alpha or TNF-α) is an adipokine and a cytokine. TNF is a member of the TNF superfamily, which consists of various transmembrane proteins with a homolog ...
, which in turn leads to production of chemokine
Chemokines (), or chemotactic cytokines, are a family of small cytokines or signaling proteins secreted by cells that induce directional movement of leukocytes, as well as other cell types, including endothelial and epithelial cells. In additio ...
s that bind to proteoglycan
Proteoglycans are proteins that are heavily glycosylated. The basic proteoglycan unit consists of a "core protein" with one or more covalently attached glycosaminoglycan (GAG) chain(s). The point of attachment is a serine (Ser) residue to whic ...
s forming gradient in the inflamed tissue and along the endothelial
The endothelium is a single layer of squamous endothelial cells that line the interior surface of blood vessels and lymphatic vessels. The endothelium forms an interface between circulating blood or lymph in the lumen and the rest of the vessel ...
wall. Inflammatory cytokines induce the immediate expression of P-selectin
P-selectin is a type-1 transmembrane protein that in humans is encoded by the SELP gene.
P-selectin functions as a cell adhesion molecule (CAM) on the surfaces of activated endothelial cells, which line the inner surface of blood vessels, and act ...
on endothelial cell surfaces and P-selectin binds weakly to carbohydrate ligands on the surface of leukocytes and causes them to "roll" along the endothelial surface as bonds are made and broken. Cytokines released from injured cells induce the expression of E-selectin
E-selectin, also known as CD62 antigen-like family member E (CD62E), endothelial-leukocyte adhesion molecule 1 (ELAM-1), or leukocyte-endothelial cell adhesion molecule 2 (LECAM2), is a selectin cell adhesion molecule expressed only on endothelial ...
on endothelial cells, which functions similarly to P-selectin. Cytokines also induce the expression of integrin
Integrins are transmembrane receptors that facilitate cell-cell and cell-extracellular matrix (ECM) adhesion. Upon ligand binding, integrins activate signal transduction pathways that mediate cellular signals such as regulation of the cell cycle, ...
ligands such as ICAM-1
ICAM-1 (Intercellular Adhesion Molecule 1) also known as CD54 (Cluster of Differentiation 54) is a protein that in humans is encoded by the ''ICAM1'' gene. This gene encodes a cell surface glycoprotein which is typically expressed on endothelial ...
and VCAM-1
Vascular cell adhesion protein 1 also known as vascular cell adhesion molecule 1 (VCAM-1) or cluster of differentiation 106 (CD106) is a protein that in humans is encoded by the ''VCAM1'' gene. VCAM-1 functions as a cell adhesion molecule.
Stru ...
on endothelial cells, which mediate the adhesion and further slow leukocytes down. These weakly bound leukocytes are free to detach if not activated by chemokines produced in injured tissue after signal transduction
Signal transduction is the process by which a chemical or physical signal is transmitted through a cell as a series of molecular events, most commonly protein phosphorylation catalyzed by protein kinases, which ultimately results in a cellula ...
via respective G protein-coupled receptors
G protein-coupled receptors (GPCRs), also known as seven-(pass)-transmembrane domain receptors, 7TM receptors, heptahelical receptors, serpentine receptors, and G protein-linked receptors (GPLR), form a large group of evolutionarily-related p ...
that activates integrins on the leukocyte surface for firm adhesion. Such activation increases the affinity of bound integrin receptors for ICAM-1 and VCAM-1 on the endothelial cell surface, firmly binding the leukocytes to the endothelium.
# Migration across the endothelium, known as'' transmigration, ''via the process of diapedesis
Leukocyte extravasation (also commonly known as leukocyte adhesion cascade or diapedesis – the passage of cells through the intact vessel wall) is the movement of leukocytes out of the circulatory system and towards the site of tissue damage or ...
: Chemokine gradients stimulate the adhered leukocytes to move between adjacent endothelial cells. The endothelial cells retract and the leukocytes pass through the basement membrane into the surrounding tissue using adhesion molecules such as ICAM-1.
# Movement of leukocytes within the tissue via chemotaxis
Chemotaxis (from '' chemo-'' + ''taxis'') is the movement of an organism or entity in response to a chemical stimulus. Somatic cells, bacteria, and other single-cell or multicellular organisms direct their movements according to certain chemica ...
: Leukocytes reaching the tissue interstitium bind to extracellular matrix
In biology, the extracellular matrix (ECM), also called intercellular matrix, is a three-dimensional network consisting of extracellular macromolecules and minerals, such as collagen, enzymes, glycoproteins and hydroxyapatite that provide stru ...
proteins via expressed integrins and CD44
The CD44 antigen is a cell-surface glycoprotein involved in cell–cell interactions, cell adhesion and migration. In humans, the CD44 antigen is encoded by the ''CD44'' gene on chromosome 11. CD44 has been referred to as HCAM (homing cell adhes ...
to prevent them from leaving the site. A variety of molecules behave as chemoattractant
Chemotaxis (from '' chemo-'' + ''taxis'') is the movement of an organism or entity in response to a chemical stimulus. Somatic cells, bacteria, and other single-cell or multicellular organisms direct their movements according to certain chemical ...
s, for example, C3a or C5, and cause the leukocytes to move along a chemotactic gradient towards the source of inflammation.
Phagocytosis
Extravasated neutrophils in the cellular phase come into contact with microbes at the inflamed tissue.Phagocyte
Phagocytes are cells that protect the body by ingesting harmful foreign particles, bacteria, and dead or dying cells. Their name comes from the Greek ', "to eat" or "devour", and "-cyte", the suffix in biology denoting "cell", from the Greek ...
s express cell-surface endocytic pattern recognition receptors
Pattern recognition receptors (PRRs) play a crucial role in the proper function of the innate immune system. PRRs are germline-encoded host sensors, which detect molecules typical for the pathogens. They are proteins expressed, mainly, by cells of ...
(PRRs) that have affinity and efficacy against non-specific microbe-associated molecular patterns
Pathogen-associated molecular patterns (PAMPs) are small molecular motifs conserved within a class of microbes. They are recognized by toll-like receptors (TLRs) and other pattern recognition receptors (PRRs) in both plants and animals. A vast arra ...
(PAMPs). Most PAMPs that bind to endocytic PRRs and initiate phagocytosis
Phagocytosis () is the process by which a cell uses its plasma membrane to engulf a large particle (≥ 0.5 μm), giving rise to an internal compartment called the phagosome. It is one type of endocytosis. A cell that performs phagocytosis is ...
are cell wall components, including complex carbohydrates such as mannans and β-glucans
A glucan is a polysaccharide derived from D-glucose, linked by glycosidic bonds. Glucans are noted in two forms: alpha glucans and beta glucans. Many beta-glucans are medically important. They represent a drug target for antifungal medications of ...
, lipopolysaccharides
Lipopolysaccharides (LPS) are large molecules consisting of a lipid and a polysaccharide that are bacterial toxins. They are composed of an O-antigen, an outer core, and an inner core all joined by a covalent bond, and are found in the outer me ...
(LPS), peptidoglycans, and surface proteins. Endocytic PRRs on phagocytes reflect these molecular patterns, with C-type lectin
A C-type lectin (CLEC) is a type of carbohydrate-binding protein known as a lectin. The C-type designation is from their requirement for calcium for binding. Proteins that contain C-type lectin domains have a diverse range of functions including ...
receptors binding to mannans and β-glucans, and scavenger receptors binding to LPS.
Upon endocytic PRR binding, actin
Actin is a family of globular multi-functional proteins that form microfilaments in the cytoskeleton, and the thin filaments in muscle fibrils. It is found in essentially all eukaryotic cells, where it may be present at a concentration of over ...
-myosin
Myosins () are a superfamily of motor proteins best known for their roles in muscle contraction and in a wide range of other motility processes in eukaryotes. They are ATP-dependent and responsible for actin-based motility.
The first myosin ...
cytoskeletal
The cytoskeleton is a complex, dynamic network of interlinking protein filaments present in the cytoplasm of all cells, including those of bacteria and archaea. In eukaryotes, it extends from the cell nucleus to the cell membrane and is compo ...
rearrangement adjacent to the plasma membrane occurs in a way that endocytoses the plasma membrane containing the PRR-PAMP complex, and the microbe. Phosphatidylinositol
Phosphatidylinositol (or Inositol Phospholipid) consists of a family of lipids as illustrated on the right, where red is x, blue is y, and black is z, in the context of independent variation, a class of the phosphatidylglycerides. In such molecul ...
and Vps34 Class III PI 3-kinase is a subgroup of the enzyme family, phosphoinositide 3-kinase that share a common protein domain structure, substrate specificity and method of activation.
There is only one known class III PI 3-kinase, Vps34, which is also th ...
- Vps15- Beclin1 signalling pathways have been implicated to traffic the endocytosed phagosome to intracellular lysosomes
A lysosome () is a membrane-bound organelle found in many animal cells. They are spherical vesicles that contain hydrolytic enzymes that can break down many kinds of biomolecules. A lysosome has a specific composition, of both its membrane prote ...
, where fusion of the phagosome and the lysosome produces a phagolysosome. The reactive oxygen species
In chemistry, reactive oxygen species (ROS) are highly reactive chemicals formed from diatomic oxygen (). Examples of ROS include peroxides, superoxide, hydroxyl radical, singlet oxygen, and alpha-oxygen.
The reduction of molecular oxygen () p ...
, superoxides
In chemistry, a superoxide is a compound that contains the superoxide ion, which has the chemical formula . The systematic name of the anion is dioxide(1−). The reactive oxygen ion superoxide is particularly important as the product of the ...
and hypochlorite
In chemistry, hypochlorite is an anion with the chemical formula ClO−. It combines with a number of cations to form hypochlorite salts. Common examples include sodium hypochlorite (household bleach) and calcium hypochlorite (a component of ble ...
bleach within the phagolysosomes then kill microbes inside the phagocyte.
Phagocytic efficacy can be enhanced by opsonization
Opsonins are extracellular proteins that, when bound to substances or cells, induce phagocytes to phagocytose the substances or cells with the opsonins bound. Thus, opsonins act as tags to label things in the body that should be phagocytosed (i.e. ...
. Plasma derived complement C3b
C3b is the larger of two elements formed by the cleavage of complement component 3, and is considered an important part of the innate immune system. C3b is potent in opsonization: tagging pathogens, immune complexes (antigen-antibody), and apopto ...
and antibodies that exude into the inflamed tissue during the vascular phase bind to and coat the microbial antigens. As well as endocytic PRRs, phagocytes also express opsonin
Opsonins are extracellular proteins that, when bound to substances or cells, induce phagocytes to phagocytose the substances or cells with the opsonins bound. Thus, opsonins act as tags to label things in the body that should be phagocytosed (i.e. ...
receptors Fc receptor and complement receptor 1
Complement receptor type 1 (CR1) also known as C3b/C4b receptor or CD35 (cluster of differentiation 35) is a protein that in humans is encoded by the ''CR1'' gene.
This gene is a member of the regulators of complement activation (RCA) family a ...
(CR1), which bind to antibodies and C3b, respectively. The co-stimulation of endocytic PRR and opsonin receptor increases the efficacy of the phagocytic process, enhancing the lysosomal
A lysosome () is a membrane-bound organelle found in many animal Cell (biology), cells. They are spherical Vesicle (biology and chemistry), vesicles that contain Hydrolysis, hydrolytic enzymes that can break down many kinds of biomolecules. A ly ...
elimination of the infective agent.
Cell-derived mediators
Morphologic patterns
Specific patterns of acute and chronic inflammation are seen during particular situations that arise in the body, such as when inflammation occurs on anepithelial
Epithelium or epithelial tissue is one of the four basic types of animal tissue, along with connective tissue, muscle tissue and nervous tissue. It is a thin, continuous, protective layer of compactly packed cells with a little intercellula ...
surface, or pyogenic
Pus is an exudate, typically white-yellow, yellow, or yellow-brown, formed at the site of inflammation during bacterial or fungal infection. An accumulation of pus in an enclosed tissue space is known as an abscess, whereas a visible collection ...
bacteria are involved.
* Granulomatous inflammation: Characterised by the formation of granuloma
A granuloma is an aggregation of macrophages that forms in response to chronic inflammation. This occurs when the immune system attempts to isolate foreign substances that it is otherwise unable to eliminate. Such substances include infectious ...
s, they are the result of a limited but diverse number of diseases, which include among others tuberculosis
Tuberculosis (TB) is an infectious disease usually caused by '' Mycobacterium tuberculosis'' (MTB) bacteria. Tuberculosis generally affects the lungs, but it can also affect other parts of the body. Most infections show no symptoms, in ...
, leprosy
Leprosy, also known as Hansen's disease (HD), is a long-term infection by the bacteria ''Mycobacterium leprae'' or ''Mycobacterium lepromatosis''. Infection can lead to damage of the nerves, respiratory tract, skin, and eyes. This nerve damag ...
, sarcoidosis
Sarcoidosis (also known as ''Besnier-Boeck-Schaumann disease'') is a disease involving abnormal collections of inflammatory cells that form lumps known as granulomata. The disease usually begins in the lungs, skin, or lymph nodes. Less commonly af ...
, and syphilis
Syphilis () is a sexually transmitted infection caused by the bacterium ''Treponema pallidum'' subspecies ''pallidum''. The signs and symptoms of syphilis vary depending in which of the four stages it presents (primary, secondary, latent, an ...
.
* Fibrinous inflammation: Inflammation resulting in a large increase in vascular permeability allows fibrin
Fibrin (also called Factor Ia) is a fibrous, non-globular protein involved in the clotting of blood. It is formed by the action of the protease thrombin on fibrinogen, which causes it to polymerize. The polymerized fibrin, together with platele ...
to pass through the blood vessels. If an appropriate ''procoagulative'' stimulus is present, such as cancer cells, a fibrinous exudate is deposited. This is commonly seen in serous cavities, where the conversion of fibrinous exudate into a scar can occur between serous membranes, limiting their function. The deposit sometimes forms a pseudomembrane sheet. During inflammation of the intestine (pseudomembranous colitis
Colitis is swelling or inflammation of the large intestine ( colon). Colitis may be acute and self-limited or long-term. It broadly fits into the category of digestive diseases.
In a medical context, the label ''colitis'' (without qualification) ...
), pseudomembranous tubes can be formed.
* Purulent inflammation: Inflammation resulting in large amount of pus
Pus is an exudate, typically white-yellow, yellow, or yellow-brown, formed at the site of inflammation during bacterial or fungal infection. An accumulation of pus in an enclosed tissue space is known as an abscess, whereas a visible collection ...
, which consists of neutrophils, dead cells, and fluid. Infection by pyogenic bacteria such as staphylococci
''Staphylococcus'' is a genus of Gram-positive bacteria in the family Staphylococcaceae from the order Bacillales. Under the microscope, they appear spherical (cocci), and form in grape-like clusters. ''Staphylococcus'' species are facultative ...
is characteristic of this kind of inflammation. Large, localised collections of pus enclosed by surrounding tissues are called abscess
An abscess is a collection of pus that has built up within the tissue of the body. Signs and symptoms of abscesses include redness, pain, warmth, and swelling. The swelling may feel fluid-filled when pressed. The area of redness often extends b ...
es.
* Serous inflammation: Characterised by the copious effusion of non-viscous serous fluid, commonly produced by mesothelial
The mesothelium is a membrane composed of simple squamous epithelial cells of mesodermal origin, which forms the lining of several body cavities: the pleura ( pleural cavity around the lungs), peritoneum ( abdominopelvic cavity including the ...
cells of serous membrane
The serous membrane (or serosa) is a smooth tissue membrane of mesothelium lining the contents and inner walls of body cavities, which secrete serous fluid to allow lubricated sliding movements between opposing surfaces. The serous membrane ...
s, but may be derived from blood plasma. Skin blister
A blister is a small pocket of body fluid (lymph, serum, plasma, blood, or pus) within the upper layers of the skin, usually caused by forceful rubbing (friction), burning, freezing, chemical exposure or infection. Most blisters are filled wi ...
s exemplify this pattern of inflammation.
* Ulcerative inflammation: Inflammation occurring near an epithelium can result in the necrotic
Necrosis () is a form of cell injury which results in the premature death of cells in living tissue by autolysis. Necrosis is caused by factors external to the cell or tissue, such as infection, or trauma which result in the unregulated dige ...
loss of tissue from the surface, exposing lower layers. The subsequent excavation in the epithelium is known as an ulcer
An ulcer is a discontinuity or break in a bodily membrane that impedes normal function of the affected organ. According to Robbins's pathology, "ulcer is the breach of the continuity of skin, epithelium or mucous membrane caused by sloughing o ...
.
Disorders

 Inflammatory abnormalities are a large group of disorders that underlie a vast variety of human diseases. The immune system is often involved with inflammatory disorders, as demonstrated in both
Inflammatory abnormalities are a large group of disorders that underlie a vast variety of human diseases. The immune system is often involved with inflammatory disorders, as demonstrated in both allergic reaction
Allergies, also known as allergic diseases, refer a number of conditions caused by the hypersensitivity of the immune system to typically harmless substances in the environment. These diseases include hay fever, food allergies, atopic derma ...
s and some myopathies
In medicine, myopathy is a disease of the muscle in which the muscle fibers do not function properly. This results in muscular weakness. ''Myopathy'' means muscle disease (Greek : myo- ''muscle'' + patheia '' -pathy'' : ''suffering''). This mean ...
, with many immune system disorder
An autoimmune disease is a condition arising from an abnormal immune response to a functioning body part. At least 80 types of autoimmune diseases have been identified, with some evidence suggesting that there may be more than 100 types. Nearly a ...
s resulting in abnormal inflammation. Non-immune diseases with causal origins in inflammatory processes include cancer, atherosclerosis
Atherosclerosis is a pattern of the disease arteriosclerosis in which the wall of the artery develops abnormalities, called lesions. These lesions may lead to narrowing due to the buildup of atheroma, atheromatous plaque. At onset there are usu ...
, and ischemic heart disease
Coronary artery disease (CAD), also called coronary heart disease (CHD), ischemic heart disease (IHD), myocardial ischemia, or simply heart disease, involves the reduction of blood flow to the heart muscle due to build-up of atherosclerotic pla ...
.
Examples of disorders associated with inflammation include:
* Acne vulgaris
Acne, also known as ''acne vulgaris'', is a long-term skin condition that occurs when dead skin cells and oil from the skin clog hair follicles. Typical features of the condition include blackheads or whiteheads, pimples, oily skin, and po ...
* Asthma
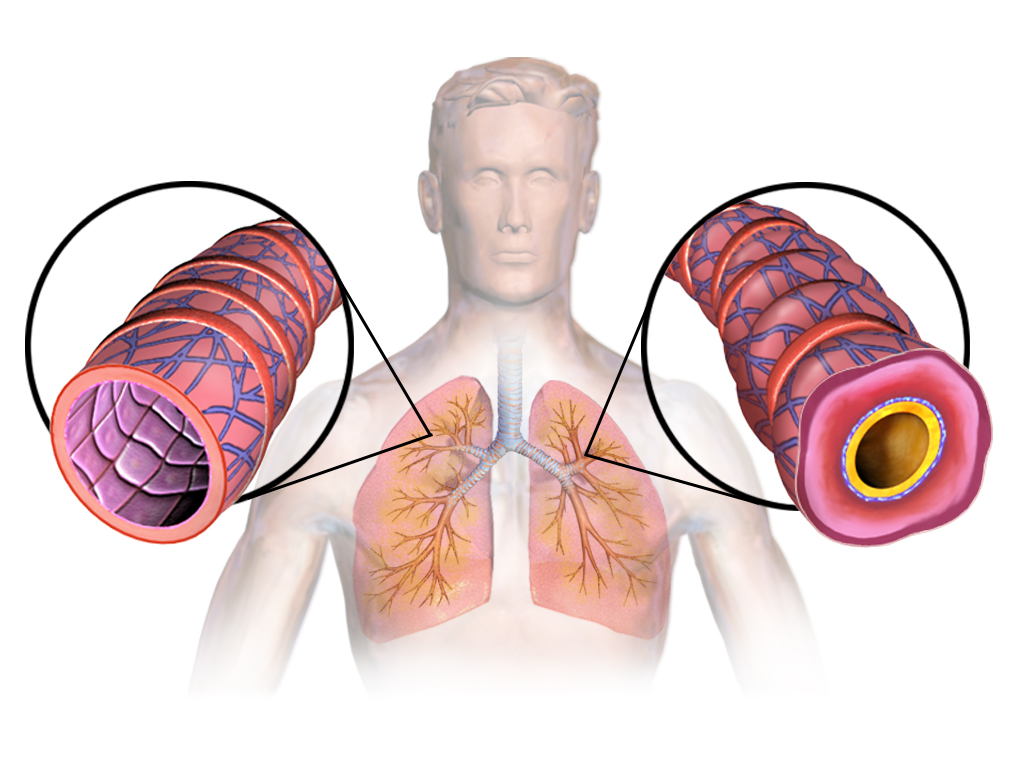
Asthma is a long-term inflammatory disease of the airways of the lungs. It is characterized by variable and recurring symptoms, reversible airflow obstruction, and easily triggered bronchospasms. Symptoms include episodes of wheezing, cou ...
* Autoimmune disease
An autoimmune disease is a condition arising from an abnormal immune response to a functioning body part. At least 80 types of autoimmune diseases have been identified, with some evidence suggesting that there may be more than 100 types. Nearly a ...
s
* Autoinflammatory disease
Periodic fever syndromes are a set of disorders characterized by recurrent episodes of systemic and organ-specific inflammation. Unlike autoimmune disorders such as systemic lupus erythematosus, in which the disease is caused by abnormalities of ...
s
* Celiac disease
Coeliac disease (British English) or celiac disease (American English) is a long-term autoimmune disorder, primarily affecting the small intestine, where individuals develop intolerance to gluten, present in foods such as wheat, rye and barl ...
* Chronic prostatitis
* Colitis
Colitis is swelling or inflammation of the large intestine ( colon). Colitis may be acute and self-limited or long-term. It broadly fits into the category of digestive diseases.
In a medical context, the label ''colitis'' (without qualification) ...
* Diverticulitis
Diverticulitis, specifically colonic diverticulitis, is a gastrointestinal disease characterized by inflammation of abnormal pouches—diverticula—which can develop in the wall of the large intestine. Symptoms typically include lower abdominal ...
* Familial Mediterranean Fever
Familial Mediterranean fever (FMF) is a hereditary inflammatory disorder. FMF is an autoinflammatory disease caused by mutations in Mediterranean fever gene, which encodes a 781–amino acid protein called pyrin. While all ethnic groups are su ...
* Glomerulonephritis
Glomerulonephritis (GN) is a term used to refer to several kidney diseases (usually affecting both kidneys). Many of the diseases are characterised by inflammation either of the glomeruli or of the small blood vessels in the kidneys, hence the ...
* Hidradenitis suppurativa
Hidradenitis suppurativa (HS), sometimes known as acne inversa or Verneuil's disease, is a long-term dermatological condition characterized by the occurrence of inflamed and swollen lumps. These are typically painful and break open, releasing ...
* Hypersensitivities
* Inflammatory bowel disease
Inflammatory bowel disease (IBD) is a group of inflammation, inflammatory conditions of the colon (anatomy), colon and small intestine, Crohn's disease and ulcerative colitis being the principal types. Crohn's disease affects the small intestine a ...
s
* Interstitial cystitis
Interstitial cystitis (IC), a type of bladder pain syndrome (BPS), is chronic pain in the bladder and pelvic floor of unknown cause. It is the urologic chronic pelvic pain syndrome of women. Symptoms include feeling the need to urinate right awa ...
* Lichen planus
Lichen planus (LP) is a chronic inflammatory and immune-mediated disease that affects the skin, nails, hair, and mucous membranes. It is not an actual lichen, and is only named that because it looks like one. It is characterized by polygonal, fla ...
* Mast Cell Activation Syndrome
Mast cell activation syndrome (MCAS) is a type of mast cell activation disorder (MCAD), and is an immunological condition in which mast cells inappropriately and excessively release chemical mediators, resulting in a range of chronic symptoms, ...
* Mastocytosis
Mastocytosis, a type of mast cell disease, is a rare disorder affecting both children and adults caused by the accumulation of functionally defective mast cells (also called ''mastocytes'') and CD34+ mast cell precursors.
People affected by masto ...
* Otitis
Otitis is a general term for inflammation or infection, inner ear infection, middle ear infection of the ear, in both humans and other animals. When infection is present, it may be viral or bacterial. When inflammation is present due to fluid buil ...
* Pelvic inflammatory disease
Pelvic inflammatory disease, also known as pelvic inflammatory disorder (PID), is an infection of the upper part of the female reproductive system, namely the uterus, fallopian tubes, and ovaries, and inside of the pelvis. Often, there may be no ...
* Peripheral ulcerative keratitis
Peripheral Ulcerative Keratitis (PUK) is a group of destructive inflammatory diseases involving the peripheral cornea in human eyes. The symptoms of PUK include pain, redness of the eyeball, photophobia, and decreased vision accompanied by distinc ...
* Pneumonia
Pneumonia is an inflammatory condition of the lung primarily affecting the small air sacs known as alveoli. Symptoms typically include some combination of productive or dry cough, chest pain, fever, and difficulty breathing. The severity ...
* Reperfusion injury
Reperfusion injury, sometimes called ischemia-reperfusion injury (IRI) or reoxygenation injury, is the tissue damage caused when blood supply returns to tissue ('' re-'' + '' perfusion'') after a period of ischemia or lack of oxygen (anoxia or h ...
* Rheumatic fever
Rheumatic fever (RF) is an inflammatory disease that can involve the heart, joints, skin, and brain. The disease typically develops two to four weeks after a streptococcal throat infection. Signs and symptoms include fever, multiple painful jo ...
* Rheumatoid arthritis
Rheumatoid arthritis (RA) is a long-term autoimmune disorder that primarily affects joints. It typically results in warm, swollen, and painful joints. Pain and stiffness often worsen following rest. Most commonly, the wrist and hands are involv ...
* Rhinitis
Rhinitis, also known as coryza, is irritation and inflammation of the mucous membrane inside the nose. Common symptoms are a stuffy nose, runny nose, sneezing, and post-nasal drip.
The inflammation is caused by viruses, bacteria, irritants o ...
* Sarcoidosis
Sarcoidosis (also known as ''Besnier-Boeck-Schaumann disease'') is a disease involving abnormal collections of inflammatory cells that form lumps known as granulomata. The disease usually begins in the lungs, skin, or lymph nodes. Less commonly af ...
* Transplant rejection
Transplant rejection occurs when Organ transplant, transplanted tissue is rejected by the recipient's immune system, which destroys the transplanted tissue. Transplant rejection can be lessened by determining the molecular similitude between don ...
* Vasculitis
Vasculitis is a group of disorders that destroy blood vessels by inflammation. Both arteries and veins are affected. Lymphangitis (inflammation of lymphatic vessels) is sometimes considered a type of vasculitis. Vasculitis is primarily caused b ...
Atherosclerosis
Atherosclerosis, formerly considered a bland lipid storage disease, actually involves an ongoing inflammatory response. Recent advances in basic science have established a fundamental role for inflammation in mediating all stages of atherosclerosis from initiation through progression and, ultimately, the thrombotic complications from it. These new findings provide important links between risk factors and the mechanisms ofatherogenesis
Atherosclerosis is a pattern of the disease arteriosclerosis in which the wall of the artery develops abnormalities, called lesions. These lesions may lead to narrowing due to the buildup of atheromatous plaque. At onset there are usually no sy ...
. Clinical studies have shown that this emerging biology of inflammation in atherosclerosis applies directly to human patients. Elevation in markers of inflammation predicts outcomes of patients with acute coronary syndromes, independently of myocardial damage. In addition, low-grade chronic inflammation, as indicated by levels of the inflammatory marker C-reactive protein
C-reactive protein (CRP) is an annular (ring-shaped) pentameric protein found in blood plasma, whose circulating concentrations rise in response to inflammation. It is an acute-phase protein of hepatic origin that increases following interleukin-6 ...
, prospectively defines risk of atherosclerotic complications, thus adding to prognostic information provided by traditional risk factors. Moreover, certain treatments that reduce coronary risk also limit inflammation. In the case of lipid lowering with statins, the anti-inflammatory effect does not appear to correlate with reduction in low-density lipoprotein levels. These new insights on inflammation contribute to the etiology of atherosclerosis, and the practical clinical applications in risk stratification and the targeting of therapy for atherosclerosis.
Allergy
An allergic reaction, formally known astype 1 hypersensitivity
Type I hypersensitivity (or immediate hypersensitivity), in the Gell and Coombs classification of allergic reactions, is an allergic reaction provoked by re-exposure to a specific type of antigen referred to as an allergen. Type I is distinct fro ...
, is the result of an inappropriate immune response triggering inflammation, vasodilation, and nerve irritation. A common example is hay fever
Allergic rhinitis, of which the seasonal type is called hay fever, is a type of inflammation in the nose that occurs when the immune system overreacts to allergens in the air. Signs and symptoms include a runny or stuffy nose, sneezing, red, i ...
, which is caused by a hypersensitive response by mast cell
A mast cell (also known as a mastocyte or a labrocyte) is a resident cell of connective tissue that contains many granules rich in histamine and heparin. Specifically, it is a type of granulocyte derived from the myeloid stem cell that is a par ...
s to allergen
An allergen is a type of antigen that produces an abnormally vigorous immune response in which the immune system fights off a perceived threat that would otherwise be harmless to the body. Such reactions are called allergies.
In technical terms ...
s. Pre-sensitised mast cells respond by degranulating, releasing vasoactive
A vasoactive substance is an endogenous agent or pharmaceutical drug that has the effect of either increasing or decreasing blood pressure and/or heart rate through its vasoactivity, that is, vascular activity (effect on blood vessels). By adju ...
chemicals such as histamine. These chemicals propagate an excessive inflammatory response characterised by blood vessel dilation, production of pro-inflammatory molecules, cytokine release, and recruitment of leukocytes. Severe inflammatory response may mature into a systemic response known as anaphylaxis
Anaphylaxis is a serious, potentially fatal allergic reaction and medical emergency that is rapid in onset and requires immediate medical attention regardless of use of emergency medication on site. It typically causes more than one of the follow ...
.
Myopathies
Inflammatory myopathies are caused by the immune system inappropriately attacking components of muscle, leading to signs of muscle inflammation. They may occur in conjunction with other immune disorders, such assystemic sclerosis
Systemic scleroderma, or systemic sclerosis, is an autoimmune rheumatic disease characterised by excessive production and accumulation of collagen, called fibrosis, in the skin and internal organs and by injuries to small arteries. There are two m ...
, and include dermatomyositis
Dermatomyositis (DM) is a long-term inflammatory disorder which affects skin and the muscles. Its symptoms are generally a skin rash and worsening muscle weakness over time. These may occur suddenly or develop over months. Other symptoms may inc ...
, polymyositis
Polymyositis (PM) is a type of chronic inflammation of the muscles (inflammatory myopathy) related to dermatomyositis and inclusion body myositis. Its name means "inflammation of many muscles" ('' poly-'' + '' myos-'' + ''-itis''). The inflammati ...
, and inclusion body myositis
Inclusion body myositis (IBM) () (sometimes called sporadic inclusion body myositis, sIBM) is the most common inflammatory muscle disease in older adults. The disease is characterized by slowly progressive weakness and wasting of both proximal ...
.
Leukocyte defects
Due to the central role of leukocytes in the development and propagation of inflammation, defects in leukocyte functionality often result in a decreased capacity for inflammatory defense with subsequent vulnerability to infection. Dysfunctional leukocytes may be unable to correctly bind to blood vessels due to surface receptor mutations, digest bacteria (Chédiak–Higashi syndrome
Chédiak–Higashi syndrome (CHS) is a rare autosomal recessive disorder that arises from a mutation of a lysosomal trafficking regulator protein, which leads to a decrease in phagocytosis. The decrease in phagocytosis results in recurrent pyogeni ...
), or produce microbicide
An antimicrobial is an agent that kills microorganisms or stops their growth. Antimicrobial medicines can be grouped according to the microorganisms they act primarily against. For example, antibiotics are used against bacteria, and antifungals ar ...
s (chronic granulomatous disease
Chronic granulomatous disease (CGD), also known as Bridges–Good syndrome, chronic granulomatous disorder, and Quie syndrome, is a diverse group of hereditary diseases in which certain cells of the immune system have difficulty forming the reacti ...
). In addition, diseases affecting the bone marrow
Bone marrow is a semi-solid tissue found within the spongy (also known as cancellous) portions of bones. In birds and mammals, bone marrow is the primary site of new blood cell production (or haematopoiesis). It is composed of hematopoietic ce ...
may result in abnormal or few leukocytes.
Pharmacological
Certain drugs or exogenous chemical compounds are known to affect inflammation.Vitamin A
Vitamin A is a fat-soluble vitamin and an essential nutrient for humans. It is a group of organic compounds that includes retinol, retinal (also known as retinaldehyde), retinoic acid, and several provitamin A carotenoids (most notably bet ...
deficiency, for example, causes an increase in inflammatory responses, and anti-inflammatory
Anti-inflammatory is the property of a substance or treatment that reduces inflammation or swelling. Anti-inflammatory drugs, also called anti-inflammatories, make up about half of analgesics. These drugs remedy pain by reducing inflammation as o ...
drugs work specifically by inhibiting the enzymes that produce inflammatory eicosanoids
Eicosanoids are signaling molecules made by the enzymatic or non-enzymatic oxidation of arachidonic acid or other polyunsaturated fatty acids (PUFAs) that are, similar to arachidonic acid, around 20 carbon units in length. Eicosanoids are a sub-c ...
. Additionally, certain illicit drugs such as cocaine
Cocaine (from , from , ultimately from Quechuan languages, Quechua: ''kúka'') is a central nervous system (CNS) stimulant mainly recreational drug use, used recreationally for its euphoria, euphoric effects. It is primarily obtained from t ...
and ecstasy may exert some of their detrimental effects by activating transcription factors intimately involved with inflammation (e.g. NF-κB
Nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) is a protein complex that controls transcription of DNA, cytokine production and cell survival. NF-κB is found in almost all animal cell types and is involved in cellular ...
).
Cancer
Inflammation orchestrates the microenvironment around tumours, contributing to proliferation, survival and migration. Cancer cells useselectins
The selectins (cluster of differentiation 62 or CD62) are a family of cell adhesion molecules (or CAMs). All selectins are single-chain transmembrane glycoproteins that share similar properties to C-type lectins due to a related amino terminus a ...
, chemokines
Chemokines (), or chemotactic cytokines, are a family of small cytokines or signaling proteins secreted by cells that induce directional movement of leukocytes, as well as other cell types, including endothelial and epithelial cells. In addition ...
and their receptors for invasion, migration and metastasis. On the other hand, many cells of the immune system contribute to cancer immunology
Cancer immunology is an interdisciplinary branch of biology that is concerned with understanding the role of the immune system in the progression and development of cancer; the most well known application is cancer immunotherapy, which utilises ...
, suppressing cancer.
Molecular intersection between receptors of steroid hormones, which have important effects on cellular development, and transcription factors that play key roles in inflammation, such as NF-κB
Nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) is a protein complex that controls transcription of DNA, cytokine production and cell survival. NF-κB is found in almost all animal cell types and is involved in cellular ...
, may mediate some of the most critical effects of inflammatory stimuli on cancer cells. This capacity of a mediator of inflammation to influence the effects of steroid hormones in cells is very likely to affect carcinogenesis. On the other hand, due to the modular nature of many steroid hormone receptors, this interaction may offer ways to interfere with cancer progression, through targeting of a specific protein domain in a specific cell type. Such an approach may limit side effects that are unrelated to the tumor of interest, and may help preserve vital homeostatic functions and developmental processes in the organism.
According to a review of 2009, recent data suggests that cancer-related inflammation (CRI) may lead to accumulation of random genetic alterations in cancer cells.
Role in cancer
In 1863,Rudolf Virchow
Rudolf Ludwig Carl Virchow (; or ; 13 October 18215 September 1902) was a German physician, anthropologist, pathologist, prehistorian, biologist, writer, editor, and politician. He is known as "the father of modern pathology" and as the founder ...
hypothesized that the origin of cancer was at sites of chronic inflammation. At present, chronic inflammation is estimated to contribute to approximately 15% to 25% of human cancers.
Mediators and DNA damage in cancer
An inflammatory mediator is a messenger that acts on blood vessels and/or cells to promote an inflammatory response. Inflammatory mediators that contribute to neoplasia includeprostaglandin
The prostaglandins (PG) are a group of physiologically active lipid compounds called eicosanoids having diverse hormone-like effects in animals. Prostaglandins have been found in almost every tissue in humans and other animals. They are derive ...
s, inflammatory cytokine
Cytokines are a broad and loose category of small proteins (~5–25 kDa) important in cell signaling. Cytokines are peptides and cannot cross the lipid bilayer of cells to enter the cytoplasm. Cytokines have been shown to be involved in autocrin ...
s such as IL-1β
Interleukin-1 beta (IL-1β) also known as leukocytic pyrogen, leukocytic endogenous mediator, mononuclear cell factor, lymphocyte activating factor and other names, is a cytokine protein that in humans is encoded by the ''IL1B'' gene."Catabolin" ...
, TNF-α
Tumor necrosis factor (TNF, cachexin, or cachectin; formerly known as tumor necrosis factor alpha or TNF-α) is an adipokine and a cytokine. TNF is a member of the TNF superfamily, which consists of various transmembrane proteins with a homolog ...
, IL-6 and IL-15 and chemokine
Chemokines (), or chemotactic cytokines, are a family of small cytokines or signaling proteins secreted by cells that induce directional movement of leukocytes, as well as other cell types, including endothelial and epithelial cells. In additio ...
s such as IL-8 and GRO-alpha. These inflammatory mediators, and others, orchestrate an environment that fosters proliferation and survival.
Inflammation also causes DNA damages due to the induction of reactive oxygen species
In chemistry, reactive oxygen species (ROS) are highly reactive chemicals formed from diatomic oxygen (). Examples of ROS include peroxides, superoxide, hydroxyl radical, singlet oxygen, and alpha-oxygen.
The reduction of molecular oxygen () p ...
(ROS) by various intracellular inflammatory mediators. In addition, leukocytes
White blood cells, also called leukocytes or leucocytes, are the cells of the immune system that are involved in protecting the body against both infectious disease and foreign invaders. All white blood cells are produced and derived from mult ...
and other phagocytic cells
Phagocytes are cells that protect the body by ingesting harmful foreign particles, bacteria, and dead or dying cells. Their name comes from the Greek ', "to eat" or "devour", and "-cyte", the suffix in biology denoting "cell", from the Greek ' ...
attracted to the site of inflammation induce DNA damages in proliferating cells through their generation of ROS and reactive nitrogen species
Reactive nitrogen species (RNS) are a family of antimicrobial molecules derived from nitric oxide (•NO) and superoxide (O2•−) produced via the enzymatic activity of inducible nitric oxide synthase 2 ( NOS2) and NADPH oxidase respectivel ...
(RNS). ROS and RNS are normally produced by these cells to fight infection. ROS, alone, cause more than 20 types of DNA damage. Oxidative DNA damages cause both mutation
In biology, a mutation is an alteration in the nucleic acid sequence of the genome of an organism, virus, or extrachromosomal DNA. Viral genomes contain either DNA or RNA. Mutations result from errors during DNA or viral replication, mi ...
s and epigenetic alterations. RNS also cause mutagenic DNA damages.
A normal cell may undergo carcinogenesis
Carcinogenesis, also called oncogenesis or tumorigenesis, is the formation of a cancer, whereby normal cells are transformed into cancer cells. The process is characterized by changes at the cellular, genetic, and epigenetic levels and abnor ...
to become a cancer cell if it is frequently subjected to DNA damage during long periods of chronic inflammation. DNA damages may cause genetic mutation
In biology, a mutation is an alteration in the nucleic acid sequence of the genome of an organism, virus, or extrachromosomal DNA. Viral genomes contain either DNA or RNA. Mutations result from errors during DNA or viral replication, mi ...
s due to inaccurate repair. In addition, mistakes in the DNA repair process may cause epigenetic
In biology, epigenetics is the study of stable phenotypic changes (known as ''marks'') that do not involve alterations in the DNA sequence. The Greek prefix '' epi-'' ( "over, outside of, around") in ''epigenetics'' implies features that are "o ...
alterations. Mutations and epigenetic alterations that are replicated and provide a selective advantage during somatic cell proliferation may be carcinogenic.
Genome-wide analyses of human cancer tissues reveal that a single typical cancer cell may possess roughly 100 mutations in coding region
The coding region of a gene, also known as the coding sequence (CDS), is the portion of a gene's DNA or RNA that codes for protein. Studying the length, composition, regulation, splicing, structures, and functions of coding regions compared to no ...
s, 10-20 of which are "driver mutations" that contribute to cancer development. However, chronic inflammation also causes epigenetic changes such as DNA methylation
DNA methylation is a biological process by which methyl groups are added to the DNA molecule. Methylation can change the activity of a DNA segment without changing the sequence. When located in a gene promoter, DNA methylation typically acts t ...
s, that are often more common than mutations. Typically, several hundreds to thousands of genes are methylated in a cancer cell (see DNA methylation in cancer DNA methylation in cancer plays a variety of roles, helping to change the healthy cells by regulation of gene expression to a cancer cells or a diseased cells disease pattern. One of the most widely studied DNA methylation dysregulation is the pro ...
). Sites of oxidative damage in chromatin
Chromatin is a complex of DNA and protein found in eukaryotic cells. The primary function is to package long DNA molecules into more compact, denser structures. This prevents the strands from becoming tangled and also plays important roles in r ...
can recruit complexes that contain DNA methyltransferase
In biochemistry, the DNA methyltransferase (DNA MTase, DNMT) family of enzymes catalyze the transfer of a methyl group to DNA. DNA methylation serves a wide variety of biological functions. All the known DNA methyltransferases use S-adenosyl m ...
s (DNMTs), a histone deacetylase (SIRT1
Sirtuin 1, also known as NAD-dependent deacetylase sirtuin-1, is a protein that in humans is encoded by the SIRT1 gene.
SIRT1 stands for sirtuin (silent mating type information regulation 2 homolog) 1 (''S. cerevisiae''), referring to the fact t ...
), and a histone methyltransferase (EZH2), and thus induce DNA methylation. DNA methylation of a CpG island
The CpG sites or CG sites are regions of DNA where a cytosine nucleotide is followed by a guanine nucleotide in the linear sequence of bases along its 5' → 3' direction. CpG sites occur with high frequency in genomic regions called CpG isl ...
in a promoter region
In genetics, a promoter is a sequence of DNA to which proteins bind to initiate transcription of a single RNA transcript from the DNA downstream of the promoter. The RNA transcript may encode a protein (mRNA), or can have a function in and of i ...
may cause silencing of its downstream gene (see CpG site
The CpG sites or CG sites are regions of DNA where a cytosine nucleotide is followed by a guanine nucleotide in the linear sequence of bases along its 5' → 3' direction. CpG sites occur with high frequency in genomic regions called CpG isl ...
and regulation of transcription in cancer Generally, in progression to cancer, hundreds of genes are silenced or activated. Although silencing of some genes in cancers occurs by mutation, a large proportion of carcinogenic gene silencing is a result of altered DNA methylation (see DNA meth ...
). DNA repair genes, in particular, are frequently inactivated by methylation in various cancers (see hypermethylation of DNA repair genes in cancer). A 2018 report evaluated the relative importance of mutations and epigenetic alterations in progression to two different types of cancer. This report showed that epigenetic alterations were much more important than mutations in generating gastric cancers (associated with inflammation). However, mutations and epigenetic alterations were of roughly equal importance in generating esophageal squamous cell cancers (associated with tobacco chemicals and acetaldehyde
Acetaldehyde (IUPAC systematic name ethanal) is an organic chemical compound with the formula CH3 CHO, sometimes abbreviated by chemists as MeCHO (Me = methyl). It is a colorless liquid or gas, boiling near room temperature. It is one of the mos ...
, a product of alcohol metabolism).
HIV and AIDS
It has long been recognized that infection with HIV is characterized not only by development of profoundimmunodeficiency
Immunodeficiency, also known as immunocompromisation, is a state in which the immune system's ability to fight infectious diseases and cancer is compromised or entirely absent. Most cases are acquired ("secondary") due to extrinsic factors that a ...
but also by sustained inflammation and immune activation. A substantial body of evidence implicates chronic inflammation as a critical driver of immune dysfunction, premature appearance of aging-related diseases, and immune deficiency. Many now regard HIV infection not only as an evolving virus-induced immunodeficiency, but also as chronic inflammatory disease. Even after the introduction of effective antiretroviral therapy (ART) and effective suppression of viremia in HIV-infected individuals, chronic inflammation persists. Animal studies also support the relationship between immune activation and progressive cellular immune deficiency: SIVsooty mangabey
The sooty mangabey (''Cercocebus atys'') is an Old World monkey found in forests from Senegal in a margin along the coast down to the Ivory Coast.
Habitat and ecology
The sooty mangabey is native to tropical West Africa, being found in Guinea, G ...
, causes high-level viral replication but limited evidence of disease. This lack of pathogenicity is accompanied by a lack of inflammation, immune activation and cellular proliferation. In sharp contrast, experimental SIVrhesus macaque
The rhesus macaque (''Macaca mulatta''), colloquially rhesus monkey, is a species of Old World monkey. There are between six and nine recognised subspecies that are split between two groups, the Chinese-derived and the Indian-derived. Generally b ...
produces immune activation and AIDS-like disease with many parallels to human HIV infection.
Delineating how CD4 T cells are depleted and how chronic inflammation and immune activation are induced lies at the heart of understanding HIV pathogenesis––one of the top priorities for HIV research by the Office of AIDS Research, National Institutes of Health
The National Institutes of Health, commonly referred to as NIH (with each letter pronounced individually), is the primary agency of the United States government responsible for biomedical and public health research. It was founded in the late ...
. Recent studies demonstrated that caspase-1
Caspase-1/Interleukin-1 converting enzyme (ICE) is an evolutionarily conserved enzyme that proteolytically cleaves other proteins, such as the precursors of the inflammatory cytokines interleukin 1β and interleukin 18 as well as the pyroptosis ...
-mediated pyroptosis Pyroptosis is a highly inflammatory form of lytic programmed cell death that occurs most frequently upon infection with intracellular pathogens and is likely to form part of the antimicrobial response. This process promotes the rapid clearance of va ...
, a highly inflammatory form of programmed cell death, drives CD4 T-cell depletion and inflammation by HIV. These are the two signature events that propel HIV disease progression to AIDS
Human immunodeficiency virus infection and acquired immunodeficiency syndrome (HIV/AIDS) is a spectrum of conditions caused by infection with the human immunodeficiency virus (HIV), a retrovirus. Following initial infection an individual m ...
. Pyroptosis appears to create a pathogenic vicious cycle in which dying CD4 T cells and other immune cells (including macrophages and neutrophils) release inflammatory signals that recruit more cells into the infected lymphoid tissues to die. The feed-forward nature of this inflammatory response produces chronic inflammation and tissue injury. Identifying pyroptosis as the predominant mechanism that causes CD4 T-cell depletion and chronic inflammation, provides novel therapeutic opportunities, namely caspase-1 which controls the pyroptotic pathway. In this regard, pyroptosis of CD4 T cells and secretion of pro-inflammatory cytokines such as IL-1β
Interleukin-1 beta (IL-1β) also known as leukocytic pyrogen, leukocytic endogenous mediator, mononuclear cell factor, lymphocyte activating factor and other names, is a cytokine protein that in humans is encoded by the ''IL1B'' gene."Catabolin" ...
and IL-18 can be blocked in HIV-infected human lymphoid tissues by addition of the caspase-1 inhibitor VX-765, which has already proven to be safe and well tolerated in phase II human clinical trials. These findings could propel development of an entirely new class of "anti-AIDS" therapies that act by targeting the host rather than the virus. Such agents would almost certainly be used in combination with ART. By promoting "tolerance" of the virus instead of suppressing its replication, VX-765 or related drugs may mimic the evolutionary solutions occurring in multiple monkey hosts (e.g. the sooty mangabey) infected with species-specific lentiviruses that have led to a lack of disease, no decline in CD4 T-cell counts, and no chronic inflammation.
Resolution
The inflammatory response must be actively terminated when no longer needed to prevent unnecessary "bystander" damage to tissues. Failure to do so results in chronic inflammation, and cellular destruction. Resolution of inflammation occurs by different mechanisms in different tissues. Mechanisms that serve to terminate inflammation include:Connection to depression
There is evidence for a link between inflammation and depression. Inflammatory processes can be triggered by negative cognitions or their consequences, such as stress, violence, or deprivation. Thus, negative cognitions can cause inflammation that can, in turn, lead to depression. In addition, there is increasing evidence that inflammation can cause depression because of the increase of cytokines, setting the brain into a "sickness mode". Classical symptoms of being physically sick, such as lethargy, show a large overlap in behaviors that characterize depression. Levels of cytokines tend to increase sharply during the depressive episodes of people with bipolar disorder and drop off during remission. Furthermore, it has been shown in clinical trials that anti-inflammatory medicines taken in addition to antidepressants not only significantly improves symptoms but also increases the proportion of subjects positively responding to treatment. Inflammations that lead to serious depression could be caused by common infections such as those caused by a virus, bacteria or even parasites.Connection to delirium
There is evidence for a link between inflammation anddelirium
Delirium (also known as acute confusional state) is an organically caused decline from a previous baseline of mental function that develops over a short period of time, typically hours to days. Delirium is a syndrome encompassing disturbances in ...
based on the results of a recent longitudinal study investigating CRP in COVID-19 patients.
Systemic effects
An infectious organism can escape the confines of the immediate tissue via thecirculatory system
The blood circulatory system is a system of organs that includes the heart, blood vessels, and blood which is circulated throughout the entire body of a human or other vertebrate. It includes the cardiovascular system, or vascular system, tha ...
or lymphatic system
The lymphatic system, or lymphoid system, is an organ system in vertebrates that is part of the immune system, and complementary to the circulatory system. It consists of a large network of lymphatic vessels, lymph nodes, lymphatic or lymphoid o ...
, where it may spread to other parts of the body. If an organism is not contained by the actions of acute inflammation, it may gain access to the lymphatic system via nearby lymph vessel
The lymphatic vessels (or lymph vessels or lymphatics) are thin-walled vessels (tubes), structured like blood vessels, that carry lymph. As part of the lymphatic system, lymph vessels are complementary to the cardiovascular system. Lymph vessel ...
s. An infection of the lymph vessels is known as lymphangitis
Lymphangitis is an inflammation or an infection of the lymphatic channels that occurs as a result of infection at a site distal to the channel. The most common cause of lymphangitis in humans is ''Streptococcus pyogenes'' (Group A strep), hemolyti ...
, and infection of a lymph node is known as lymphadenitis
Lymphadenopathy or adenopathy is a disease of the lymph nodes, in which they are abnormal in size or consistency. Lymphadenopathy of an inflammatory type (the most common type) is lymphadenitis, producing swollen or enlarged lymph nodes. In cli ...
. When lymph nodes cannot destroy all pathogens, the infection spreads further. A pathogen can gain access to the bloodstream through lymphatic drainage into the circulatory system.
When inflammation overwhelms the host, systemic inflammatory response syndrome
Systemic inflammatory response syndrome (SIRS) is an inflammatory state affecting the whole body. It is the body's response to an infectious or noninfectious insult. Although the definition of SIRS refers to it as an "inflammatory" response, i ...
is diagnosed. When it is due to infection
An infection is the invasion of tissues by pathogens, their multiplication, and the reaction of host tissues to the infectious agent and the toxins they produce. An infectious disease, also known as a transmissible disease or communicable dise ...
, the term sepsis
Sepsis, formerly known as septicemia (septicaemia in British English) or blood poisoning, is a life-threatening condition that arises when the body's response to infection causes injury to its own tissues and organs. This initial stage is follo ...
is applied, with the terms bacteremia
Bloodstream infections (BSIs), which include bacteremias when the infections are bacterial and fungemias when the infections are fungal, are infections present in the blood. Blood is normally a sterile environment, so the detection of microb ...
being applied specifically for bacterial sepsis and viremia
Viremia is a medical condition where viruses enter the bloodstream and hence have access to the rest of the body. It is similar to ''bacteremia'', a condition where bacteria enter the bloodstream. The name comes from combining the word "virus" wit ...
specifically to viral sepsis. Vasodilation
Vasodilation is the widening of blood vessels. It results from relaxation of smooth muscle cells within the vessel walls, in particular in the large veins, large arteries, and smaller arterioles. The process is the opposite of vasoconstriction, ...
and organ dysfunction are serious problems associated with widespread infection that may lead to septic shock
Septic shock is a potentially fatal medical condition that occurs when sepsis, which is organ injury or damage in response to infection, leads to dangerously low blood pressure and abnormalities in cellular metabolism. The Third International Con ...
and death.
Acute-phase proteins
Inflammation also is characterized by high systemic levels ofacute-phase protein
Acute-phase proteins (APPs) are a class of proteins whose concentrations in blood plasma either increase (positive acute-phase proteins) or decrease (negative acute-phase proteins) in response to inflammation. This response is called the ''acute-p ...
s. In acute inflammation, these proteins prove beneficial; however, in chronic inflammation, they can contribute to amyloidosis
Amyloidosis is a group of diseases in which abnormal proteins, known as amyloid fibrils, build up in tissue. There are several non-specific and vague signs and symptoms associated with amyloidosis. These include fatigue, peripheral edema, weight ...
. These proteins include C-reactive protein
C-reactive protein (CRP) is an annular (ring-shaped) pentameric protein found in blood plasma, whose circulating concentrations rise in response to inflammation. It is an acute-phase protein of hepatic origin that increases following interleukin-6 ...
, serum amyloid A
Serum amyloid A (SAA) proteins are a family of apolipoproteins associated with high-density lipoprotein (HDL) in plasma. Different isoforms of SAA are expressed constitutively (constitutive SAAs) at different levels or in response to inflammato ...
, and serum amyloid P, which cause a range of systemic effects including:
* Fever
Fever, also referred to as pyrexia, is defined as having a body temperature, temperature above the human body temperature, normal range due to an increase in the body's temperature Human body temperature#Fever, set point. There is not a single ...
* Increased blood pressure
Blood pressure (BP) is the pressure of circulating blood against the walls of blood vessels. Most of this pressure results from the heart pumping blood through the circulatory system. When used without qualification, the term "blood pressure" r ...
* Decreased sweating
Perspiration, also known as sweating, is the production of fluids secreted by the sweat glands in the skin of mammals.
Two types of sweat glands can be found in humans: eccrine glands and apocrine glands. The eccrine sweat glands are distr ...
* Malaise
As a medical term, malaise is a feeling of general discomfort, uneasiness or lack of wellbeing and often the first sign of an infection or other disease. The word has existed in French since at least the 12th century.
The term is often used ...
* Loss of appetite
Anorexia is a medical term for a loss of appetite. While the term in non-scientific publications is often used interchangeably with anorexia nervosa, many possible causes exist for a loss of appetite, some of which may be harmless, while others i ...
* Somnolence
Somnolence (alternatively sleepiness or drowsiness) is a state of strong desire for sleep, or sleeping for unusually long periods (compare hypersomnia). It has distinct meanings and causes. It can refer to the usual state preceding falling asleep ...
Leukocyte numbers
Inflammation often affects the numbers of leukocytes present in the body: *Leukocytosis
Leukocytosis is a condition in which the white cell (leukocyte count) is above the normal range in the blood. It is frequently a sign of an inflammatory response, most commonly the result of infection, but may also occur following certain parasi ...
is often seen during inflammation induced by infection, where it results in a large increase in the amount of leukocytes in the blood, especially immature cells. Leukocyte numbers usually increase to between 15 000 and 20 000 cells per microliter, but extreme cases can see it approach 100 000 cells per microliter. Bacterial infection usually results in an increase of neutrophil
Neutrophils (also known as neutrocytes or heterophils) are the most abundant type of granulocytes and make up 40% to 70% of all white blood cells in humans. They form an essential part of the innate immune system, with their functions varying in ...
s, creating neutrophilia
Neutrophilia (also called neutrophil leukocytosis or occasionally neutrocytosis) is leukocytosis of neutrophils, that is, a high number of neutrophils in the blood. Because neutrophils are the main type of granulocytes, mentions of granulocytosis ...
, whereas diseases such as asthma
Asthma is a long-term inflammatory disease of the airways of the lungs. It is characterized by variable and recurring symptoms, reversible airflow obstruction, and easily triggered bronchospasms. Symptoms include episodes of wheezing, cou ...
, hay fever
Allergic rhinitis, of which the seasonal type is called hay fever, is a type of inflammation in the nose that occurs when the immune system overreacts to allergens in the air. Signs and symptoms include a runny or stuffy nose, sneezing, red, i ...
, and parasite infestation result in an increase in eosinophil
Eosinophils, sometimes called eosinophiles or, less commonly, acidophils, are a variety of white blood cells (WBCs) and one of the immune system components responsible for combating multicellular parasites and certain infections in vertebrates. A ...
s, creating eosinophilia
Eosinophilia is a condition in which the eosinophil count in the peripheral blood exceeds . Hypereosinophilia is an elevation in an individual's circulating blood eosinophil count above 1.5 x 109/ L (i.e. 1,500/μL). The hypereosinophilic syndro ...
.
* Leukopenia
Leukopenia () is a decrease in the number of leukocytes (WBC). Found in the blood, they are the white blood cells, and are the body's primary defense against an infection. Thus the condition of leukopenia places individuals at increased risk of inf ...
can be induced by certain infections and diseases, including viral infection, ''Rickettsia
''Rickettsia'' is a genus of nonmotile, gram-negative, nonspore-forming, highly pleomorphic bacteria that may occur in the forms of cocci (0.1 μm in diameter), bacilli (1–4 μm long), or threads (up to about 10 μm long). The term "rickett ...
'' infection, some protozoa
Protozoa (singular: protozoan or protozoon; alternative plural: protozoans) are a group of single-celled eukaryotes, either free-living or parasitic, that feed on organic matter such as other microorganisms or organic tissues and debris. Histo ...
, tuberculosis
Tuberculosis (TB) is an infectious disease usually caused by '' Mycobacterium tuberculosis'' (MTB) bacteria. Tuberculosis generally affects the lungs, but it can also affect other parts of the body. Most infections show no symptoms, in ...
, and some cancer
Cancer is a group of diseases involving abnormal cell growth with the potential to invade or spread to other parts of the body. These contrast with benign tumors, which do not spread. Possible signs and symptoms include a lump, abnormal b ...
s.
Interleukins and obesity
With the discovery ofinterleukin
Interleukins (ILs) are a group of cytokines (secreted proteins and signal molecules) that are expressed and secreted by white blood cells (leukocytes) as well as some other body cells. The human genome encodes more than 50 interleukins and related ...
s (IL), the concept of systemic inflammation developed. Although the processes involved are identical to tissue inflammation, systemic inflammation is not confined to a particular tissue but involves the endothelium
The endothelium is a single layer of squamous endothelial cells that line the interior surface of blood vessels and lymphatic vessels. The endothelium forms an interface between circulating blood or lymph in the lumen and the rest of the vessel ...
and other organ systems.
Chronic inflammation is widely observed in obesity
Obesity is a medical condition, sometimes considered a disease, in which excess body fat has accumulated to such an extent that it may negatively affect health. People are classified as obese when their body mass index (BMI)—a person's we ...
. Obese people commonly have many elevated markers of inflammation, including:
* IL-6 (Interleukin-6)
Low-grade chronic inflammation
Grade most commonly refers to:
* Grade (education), a measurement of a student's performance
* Grade, the number of the year a student has reached in a given educational stage
* Grade (slope), the steepness of a slope
Grade or grading may also r ...
is characterized by a two- to threefold increase in the systemic concentrations of cytokines such as TNF-α, IL-6, and CRP. Waist circumference correlates significantly with systemic inflammatory response.
Loss of white adipose tissue
White adipose tissue or white fat is one of the two types of adipose tissue found in mammals. The other kind is brown adipose tissue. White adipose tissue is composed of monolocular adipocytes.
In humans, the healthy amount of white adipose ti ...
reduces levels of inflammation markers. As of 2017 the association of systemic inflammation with insulin resistance
Insulin resistance (IR) is a pathological condition in which cell (biology), cells fail to respond normally to the hormone insulin.
Insulin is a hormone that facilitates the transport of glucose from blood into cells, thereby reducing blood gluco ...
and type 2 diabetes
Type 2 diabetes, formerly known as adult-onset diabetes, is a form of diabetes mellitus that is characterized by high blood sugar, insulin resistance, and relative lack of insulin. Common symptoms include increased thirst, frequent urination, ...
, and with atherosclerosis
Atherosclerosis is a pattern of the disease arteriosclerosis in which the wall of the artery develops abnormalities, called lesions. These lesions may lead to narrowing due to the buildup of atheroma, atheromatous plaque. At onset there are usu ...
was under preliminary research, although rigorous clinical trial
Clinical trials are prospective biomedical or behavioral research studies on human participants designed to answer specific questions about biomedical or behavioral interventions, including new treatments (such as novel vaccines, drugs, dietar ...
s had not been conducted to confirm such relationships.
C-reactive protein
C-reactive protein (CRP) is an annular (ring-shaped) pentameric protein found in blood plasma, whose circulating concentrations rise in response to inflammation. It is an acute-phase protein of hepatic origin that increases following interleukin-6 ...
(CRP) is generated at a higher level in obese people, and may increase the risk for cardiovascular diseases
Cardiovascular disease (CVD) is a class of diseases that involve the heart or blood vessels. CVD includes coronary artery diseases (CAD) such as angina and myocardial infarction (commonly known as a heart attack). Other CVDs include stroke, ...
.
Outcomes
The outcome in a particular circumstance will be determined by the tissue in which the injury has occurred—and the injurious agent that is causing it. Here are the possible outcomes to inflammation: # ResolutionThe complete restoration of the inflamed tissue back to a normal status. Inflammatory measures such as vasodilation, chemical production, and leukocyte infiltration cease, and damaged
parenchyma
Parenchyma () is the bulk of functional substance in an animal organ or structure such as a tumour. In zoology it is the name for the tissue that fills the interior of flatworms.
Etymology
The term ''parenchyma'' is New Latin from the word π ...
l cells regenerate. Such is usually the outcome when limited or short-lived inflammation has occurred.
# Fibrosis
Fibrosis, also known as fibrotic scarring, is a pathological wound healing in which connective tissue replaces normal parenchymal tissue to the extent that it goes unchecked, leading to considerable tissue remodelling and the formation of perma ...
Large amounts of tissue destruction, or damage in tissues unable to regenerate, cannot be regenerated completely by the body. Fibrous
scarring
A scar (or scar tissue) is an area of fibrous tissue that replaces normal skin after an injury. Scars result from the biological process of wound repair in the skin, as well as in other organs, and tissues of the body. Thus, scarring is a na ...
occurs in these areas of damage, forming a scar composed primarily of collagen
Collagen () is the main structural protein in the extracellular matrix found in the body's various connective tissues. As the main component of connective tissue, it is the most abundant protein in mammals, making up from 25% to 35% of the whole ...
. The scar will not contain any specialized structures, such as parenchyma
Parenchyma () is the bulk of functional substance in an animal organ or structure such as a tumour. In zoology it is the name for the tissue that fills the interior of flatworms.
Etymology
The term ''parenchyma'' is New Latin from the word π ...
l cells, hence functional impairment may occur.
# Abscess formationA cavity is formed containing pus, an opaque liquid containing dead white blood cells and bacteria with general debris from destroyed cells. # Chronic inflammation
In acute inflammation, if the injurious agent persists then chronic inflammation will ensue. This process, marked by inflammation lasting many days, months or even years, may lead to the formation of a
chronic wound A chronic wound is a wound that does not heal in an orderly set of stages and in a predictable amount of time the way most wounds do; wounds that do not heal within three months are often considered chronic.
Chronic wounds seem to be detained in on ...
. Chronic inflammation is characterised by the dominating presence of macrophages in the injured tissue. These cells are powerful defensive agents of the body, but the toxin
A toxin is a naturally occurring organic poison produced by metabolic activities of living cells or organisms. Toxins occur especially as a protein or conjugated protein. The term toxin was first used by organic chemist Ludwig Brieger (1849– ...
s they release—including reactive oxygen species
In chemistry, reactive oxygen species (ROS) are highly reactive chemicals formed from diatomic oxygen (). Examples of ROS include peroxides, superoxide, hydroxyl radical, singlet oxygen, and alpha-oxygen.
The reduction of molecular oxygen () p ...
—are injurious to the organism's own tissues as well as invading agents. As a consequence, chronic inflammation is almost always accompanied by tissue destruction.
Examples
Inflammation is usually indicated by adding the suffix "itis
The Integrated Taxonomic Information System (ITIS) is an American partnership of federal agencies designed to provide consistent and reliable information on the taxonomy of biological species. ITIS was originally formed in 1996 as an interagenc ...
", as shown below. However, some conditions, such as asthma
Asthma is a long-term inflammatory disease of the airways of the lungs. It is characterized by variable and recurring symptoms, reversible airflow obstruction, and easily triggered bronchospasms. Symptoms include episodes of wheezing, cou ...
and pneumonia
Pneumonia is an inflammatory condition of the lung primarily affecting the small air sacs known as alveoli. Symptoms typically include some combination of productive or dry cough, chest pain, fever, and difficulty breathing. The severity ...
, do not follow this convention. More examples are available at List of types of inflammation.
appendicitis
Appendicitis is inflammation of the appendix. Symptoms commonly include right lower abdominal pain, nausea, vomiting, and decreased appetite. However, approximately 40% of people do not have these typical symptoms. Severe complications of a rup ...
File:Dermatitis.jpg, Acute dermatitis
Dermatitis is inflammation of the skin, typically characterized by itchiness, redness and a rash. In cases of short duration, there may be small blisters, while in long-term cases the skin may become thickened. The area of skin involved can v ...
File:Streptococcus pneumoniae meningitis, gross pathology 33 lores.jpg, Acute infective meningitis
Meningitis is acute or chronic inflammation of the protective membranes covering the brain and spinal cord, collectively called the meninges. The most common symptoms are fever, headache, and neck stiffness. Other symptoms include confusion or ...
File:Tonsillitis.jpg, Acute tonsillitis
Tonsillitis is inflammation of the tonsils in the upper part of the throat. It can be acute or chronic. Acute tonsillitis typically has a rapid onset. Symptoms may include sore throat, fever, enlargement of the tonsils, trouble swallowing, and en ...
See also
Notes
References
External links
* {{Authority control Immunology Animal physiology Inflammations Human physiology