Guillain–Barré syndrome on:
[Wikipedia]
[Google]
[Amazon]
Guillain–Barré syndrome (GBS) is a rapid-onset
 In children, particularly those younger than six years old, the diagnosis can be difficult and the condition is often initially mistaken (sometimes for up to two weeks) for other causes of pains and difficulty walking, such as viral infections, or bone and joint problems.
On
In children, particularly those younger than six years old, the diagnosis can be difficult and the condition is often initially mistaken (sometimes for up to two weeks) for other causes of pains and difficulty walking, such as viral infections, or bone and joint problems.
On
 Two-thirds of people with Guillain–Barré syndrome have experienced an infection before the onset of the condition. Most commonly, these are episodes of
Two-thirds of people with Guillain–Barré syndrome have experienced an infection before the onset of the condition. Most commonly, these are episodes of
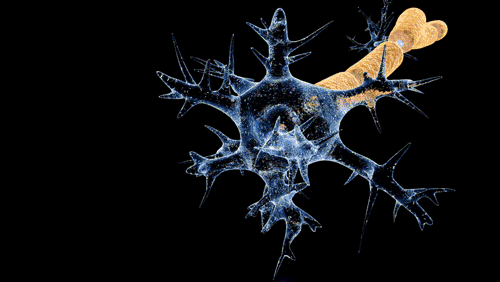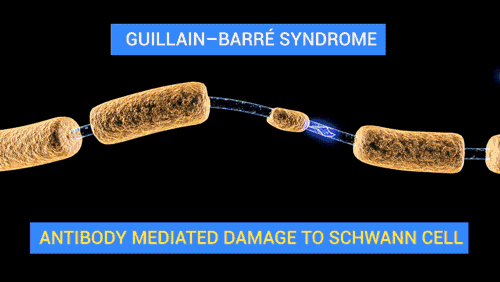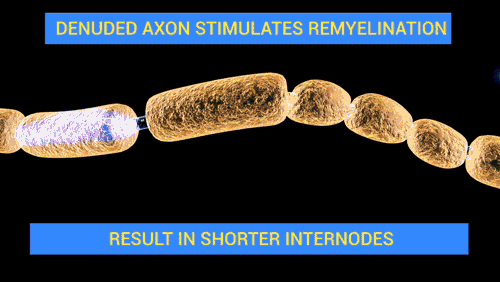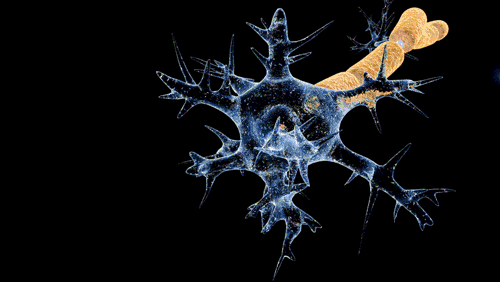 The nerve dysfunction in Guillain–Barré syndrome is caused by an immune attack on the nerve cells of the peripheral nervous system and their support structures. The nerve cells have their body (the soma) in the
The nerve dysfunction in Guillain–Barré syndrome is caused by an immune attack on the nerve cells of the peripheral nervous system and their support structures. The nerve cells have their body (the soma) in the
 Jean-Baptiste Octave Landry first described the disorder in 1859. In 1916,
Jean-Baptiste Octave Landry first described the disorder in 1859. In 1916,
muscle weakness
Muscle weakness is a lack of muscle strength. Its causes are many and can be divided into conditions that have either true or perceived muscle weakness. True muscle weakness is a primary symptom of a variety of skeletal muscle diseases, includi ...
caused by the immune system
The immune system is a network of biological processes that protects an organism from diseases. It detects and responds to a wide variety of pathogens, from viruses to parasitic worms, as well as cancer cells and objects such as wood splinte ...
damaging the peripheral nervous system
The peripheral nervous system (PNS) is one of two components that make up the nervous system of bilateral animals, with the other part being the central nervous system (CNS). The PNS consists of nerves and ganglia, which lie outside the brain ...
. Typically, both sides of the body are involved, and the initial symptoms are changes in sensation or pain often in the back along with muscle weakness, beginning in the feet and hands, often spreading to the arms and upper body. The symptoms may develop over hours to a few weeks. During the acute phase, the disorder can be life-threatening, with about 15% of people developing weakness of the breathing muscle
Breathing (or ventilation) is the process of moving air into and from the lungs to facilitate gas exchange with the internal environment, mostly to flush out carbon dioxide and bring in oxygen.
All aerobic creatures need oxygen for cellular ...
s and, therefore, requiring mechanical ventilation
Mechanical ventilation, assisted ventilation or intermittent mandatory ventilation (IMV), is the medical term for using a machine called a ventilator to fully or partially provide artificial ventilation. Mechanical ventilation helps move air ...
. Some are affected by changes in the function of the autonomic nervous system
The autonomic nervous system (ANS), formerly referred to as the vegetative nervous system, is a division of the peripheral nervous system that supplies viscera, internal organs, smooth muscle and glands. The autonomic nervous system is a control ...
, which can lead to dangerous abnormalities in heart rate
Heart rate (or pulse rate) is the frequency of the heartbeat measured by the number of contractions (beats) of the heart per minute (bpm). The heart rate can vary according to the body's physical needs, including the need to absorb oxygen and excr ...
and blood pressure
Blood pressure (BP) is the pressure of circulating blood against the walls of blood vessels. Most of this pressure results from the heart pumping blood through the circulatory system. When used without qualification, the term "blood pressure" r ...
.
Although the cause is unknown, the underlying mechanism involves an autoimmune disorder
An autoimmune disease is a condition arising from an abnormal immune response to a functioning body part. At least 80 types of autoimmune diseases have been identified, with some evidence suggesting that there may be more than 100 types. Nearly ...
in which the body's immune system mistakenly attacks the peripheral nerves and damages their myelin
Myelin is a lipid-rich material that surrounds nerve cell axons (the nervous system's "wires") to insulate them and increase the rate at which electrical impulses (called action potentials) are passed along the axon. The myelinated axon can be ...
insulation. Sometimes this immune dysfunction is triggered by an infection or, less commonly, by surgery, and rarely, by vaccination
Vaccination is the administration of a vaccine to help the immune system develop immunity from a disease. Vaccines contain a microorganism or virus in a weakened, live or killed state, or proteins or toxins from the organism. In stimulating ...
. The diagnosis is usually based on the signs and symptoms through the exclusion of alternative causes and supported by tests such as nerve conduction studies
A nerve conduction study (NCS) is a medical diagnostic test commonly used to evaluate the function, especially the ability of electrical conduction, of the motor and sensory nerves of the human body. These tests may be performed by medical spec ...
and examination of the cerebrospinal fluid
Cerebrospinal fluid (CSF) is a clear, colorless body fluid found within the tissue that surrounds the brain and spinal cord of all vertebrates.
CSF is produced by specialised ependymal cells in the choroid plexus of the ventricles of the bra ...
. There are a number of subtypes based on the areas of weakness, results of nerve conduction studies, and the presence of certain antibodies. It is classified as an acute polyneuropathy.
In those with severe weakness, prompt treatment with intravenous immunoglobulins or plasmapheresis
Plasmapheresis (from the Greek πλάσμα, ''plasma'', something molded, and ἀφαίρεσις ''aphairesis'', taking away) is the removal, treatment, and return or exchange of blood plasma or components thereof from and to the blood circulati ...
, together with supportive care, will lead to good recovery in the majority of cases. Recovery may take weeks to years, with about a third having some permanent weakness. Globally, death occurs in approximately 7.5% of those affected. Guillain–Barré syndrome is rare, at 1 or 2 cases per 100,000 people every year. Both sexes and all parts of the world have similar rates of disease.
The syndrome is named after the French neurologist
Neurology (from el, νεῦρον (neûron), "string, nerve" and the suffix -logia, "study of") is the branch of medicine dealing with the diagnosis and treatment of all categories of conditions and disease involving the brain, the spinal c ...
s Georges Guillain
Georges Charles Guillain () (3 March 1876 – 29 June 1961) was a French neurologist born in Rouen.
He studied medicine in Rouen and Paris, where he learned clinical education at several hospitals. He developed an interest in neurology, and his ...
and Jean Alexandre Barré
Jean Alexandre Barré (25 May 1880, Nantes – 26 April 1967, Strasbourg) was a French neurologist who in 1916 worked on the identification of Guillain-Barré-Strohl syndrome, as well as Barré–Liéou syndrome.
Biography First studies
He st ...
, who, together with French physician André Strohl
André Strohl (20 March 1887 – 10 March 1977) was a French physiologist who was a native of Poitiers. He is remembered for his role in the diagnosis of Guillain–Barré syndrome (sometimes called Guillain–Barré–Strohl syndrome), a form o ...
, described the condition in 1916.
Signs and symptoms
The first symptoms of Guillain–Barré syndrome are numbness, tingling, and pain, alone or in combination. This is followed by weakness of the legs and arms that affects both sides equally and worsens over time. The weakness can take half a day to over two weeks to reach maximum severity, and then becomes steady. In one in five people, the weakness continues to progress for as long as four weeks. The muscles of the neck may also be affected, and about half experience involvement of thecranial nerves
Cranial nerves are the nerves that emerge directly from the brain (including the brainstem), of which there are conventionally considered twelve pairs. Cranial nerves relay information between the brain and parts of the body, primarily to and f ...
that supply the head and face; this may lead to weakness of the muscles of the face, swallowing difficulties and sometimes weakness of the eye muscles. In 8%, the weakness affects only the legs (paraplegia or paraparesis). Involvement of the muscles that control the bladder and anus is unusual. In total, about a third of people with Guillain–Barré syndrome continue to be able to walk. Once the weakness has stopped progressing, it persists at a stable level ("plateau phase") before improvement occurs. The plateau phase can take between two days and six months, but the most common duration is a week. Pain-related symptoms affect more than half, and include back pain
Back pain is pain felt in the back. It may be classified as neck pain (cervical), middle back pain (thoracic), lower back pain (lumbar) or coccydynia (tailbone or sacral pain) based on the segment affected. The lumbar area is the most common area ...
, painful tingling, muscle pain
Myalgia (also called muscle pain and muscle ache in layman's terms) is the medical term for muscle pain. Myalgia is a symptom of many diseases. The most common cause of acute myalgia is the overuse of a muscle or group of muscles; another like ...
, and pain in the head and neck relating to irritation of the lining of the brain.
Many people with Guillain–Barré syndrome have experienced the signs and symptoms of an infection in the 3–6 weeks before the onset of the neurological symptoms. This may consist of upper respiratory tract infection
An upper respiratory tract infection (URTI) is an illness caused by an acute infection, which involves the upper respiratory tract, including the nose, sinuses, pharynx, larynx or trachea. This commonly includes nasal obstruction, sore thro ...
(rhinitis, sore throat), or diarrhea
Diarrhea, also spelled diarrhoea, is the condition of having at least three loose, liquid, or watery bowel movements each day. It often lasts for a few days and can result in dehydration due to fluid loss. Signs of dehydration often begin wi ...
.
 In children, particularly those younger than six years old, the diagnosis can be difficult and the condition is often initially mistaken (sometimes for up to two weeks) for other causes of pains and difficulty walking, such as viral infections, or bone and joint problems.
On
In children, particularly those younger than six years old, the diagnosis can be difficult and the condition is often initially mistaken (sometimes for up to two weeks) for other causes of pains and difficulty walking, such as viral infections, or bone and joint problems.
On neurological examination
A neurological examination is the assessment of sensory neuron and motor responses, especially reflexes, to determine whether the nervous system is impaired. This typically includes a physical examination and a review of the patient's medical hist ...
, characteristic features are the reduced strength of muscles and reduced or absent tendon reflexes ( hypo- or areflexia, respectively). However, a small proportion have normal reflexes in affected limbs before developing areflexia, and some may have exaggerated reflexes. In the Miller Fisher variant of Guillain–Barré syndrome (see below), a triad of weakness of the eye muscles, abnormalities in coordination, as well as absent reflexes can be found. The level of consciousness
An altered level of consciousness is any measure of arousal other than normal. Level of consciousness (LOC) is a measurement of a person's arousability and responsiveness to stimuli from the environment.
A mildly depressed level of consciousn ...
is normally unaffected in Guillain–Barré syndrome, but the Bickerstaff brainstem encephalitis
Bickerstaff brainstem encephalitis is a rare inflammatory disorder of the central nervous system, first described by Edwin Bickerstaff in 1951. It may also affect the peripheral nervous system, and has features in common with both Miller Fish ...
subtype may feature drowsiness, sleepiness, or coma.
Respiratory failure
A quarter of all people with Guillain–Barré syndrome develop weakness of the breathing muscles leading torespiratory failure
Respiratory failure results from inadequate gas exchange by the respiratory system, meaning that the arterial oxygen, carbon dioxide, or both cannot be kept at normal levels. A drop in the oxygen carried in the blood is known as hypoxemia; a rise ...
, the inability to breathe adequately to maintain healthy levels of oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as wel ...
, and/or carbon dioxide
Carbon dioxide (chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is transpar ...
in the blood. This life-threatening scenario is complicated by other medical problems such as pneumonia
Pneumonia is an inflammatory condition of the lung primarily affecting the small air sacs known as alveoli. Symptoms typically include some combination of productive or dry cough, chest pain, fever, and difficulty breathing. The severity ...
, severe infections, blood clots in the lungs, and bleeding in the digestive tract in 60% of those who require artificial ventilation.
Autonomic dysfunction
The autonomic or involuntary nervous system, which is involved in the control of body functions such asheart rate
Heart rate (or pulse rate) is the frequency of the heartbeat measured by the number of contractions (beats) of the heart per minute (bpm). The heart rate can vary according to the body's physical needs, including the need to absorb oxygen and excr ...
and blood pressure
Blood pressure (BP) is the pressure of circulating blood against the walls of blood vessels. Most of this pressure results from the heart pumping blood through the circulatory system. When used without qualification, the term "blood pressure" r ...
, is affected in two-thirds of people with Guillain–Barré syndrome, but the impact is variable. Twenty percent may experience severe blood-pressure fluctuations and irregularities in the heart beat, sometimes to the point that the heart beat stops and requires pacemaker-based treatment. Other associated problems are abnormalities in perspiration
Perspiration, also known as sweating, is the production of fluids secreted by the sweat glands in the skin of mammals.
Two types of sweat glands can be found in humans: eccrine glands and apocrine glands. The eccrine sweat glands are distrib ...
and changes in the reactivity of the pupils. Autonomic nervous system involvement can affect even those who do not have severe muscle weakness.
Causes
Infection onset
 Two-thirds of people with Guillain–Barré syndrome have experienced an infection before the onset of the condition. Most commonly, these are episodes of
Two-thirds of people with Guillain–Barré syndrome have experienced an infection before the onset of the condition. Most commonly, these are episodes of gastroenteritis
Gastroenteritis, also known as infectious diarrhea and gastro, is an inflammation of the gastrointestinal tract including the stomach and intestine. Symptoms may include diarrhea, vomiting, and abdominal pain. Fever, lack of energy, and dehydra ...
or a respiratory tract infection
Respiratory tract infections (RTIs) are infectious diseases involving the respiratory tract. An infection of this type usually is further classified as an upper respiratory tract infection (URI or URTI) or a lower respiratory tract infection (LRI ...
. In many cases, the exact nature of the infection can be confirmed. Approximately 30% of cases are provoked by ''Campylobacter jejuni'' bacteria, which cause diarrhea. A further 10% are attributable to cytomegalovirus
''Cytomegalovirus'' (''CMV'') (from ''cyto-'' 'cell' via Greek - 'container' + 'big, megalo-' + -''virus'' via Latin 'poison') is a genus of viruses in the order ''Herpesvirales'', in the family ''Herpesviridae'', in the subfamily ''Betaherpe ...
(CMV, HHV-5). Despite this, only very few people with ''Campylobacter'' or CMV infections develop Guillain–Barré syndrome (0.25–0.65 per 1000 and 0.6–2.2 per 1000 episodes, respectively). The strain of ''Campylobacter'' involved may determine the risk of GBS; different forms of the bacteria have different lipopolysaccharide
Lipopolysaccharides (LPS) are large molecules consisting of a lipid and a polysaccharide that are bacterial toxins. They are composed of an O-antigen, an outer core, and an inner core all joined by a covalent bond, and are found in the outer m ...
s on their surface, and some may induce illness (see below) while others will not.
Links between other infections and GBS are less certain. Two other herpes viruses (Epstein–Barr virus
The Epstein–Barr virus (EBV), formally called ''Human gammaherpesvirus 4'', is one of the nine known human herpesvirus types in the herpes family, and is one of the most common viruses in humans. EBV is a double-stranded DNA virus.
It is b ...
/HHV-4 and varicella zoster virus
Varicella-zoster virus (VZV), also known as human herpesvirus 3 (HHV-3, HHV3) or ''Human alphaherpesvirus 3'' (taxonomically), is one of nine known herpes viruses that can infect humans. It causes chickenpox (varicella) commonly affecting chil ...
/HHV-3) and the bacterium ''Mycoplasma pneumoniae
''Mycoplasma pneumoniae'' is a very small bacterium in the class Mollicutes. It is a human pathogen that causes the disease mycoplasma pneumonia, a form of atypical bacterial pneumonia related to cold agglutinin disease. ''M. pneumoniae'' is c ...
'' have been associated with GBS. GBS is known to occur after influenza, and influenza vaccination has been demonstrated to be associated with a reduced risk. The tropical flaviviral infections dengue fever
Dengue fever is a mosquito-borne tropical disease caused by the dengue virus. Symptoms typically begin three to fourteen days after infection. These may include a high fever, headache, vomiting, muscle and joint pains, and a characterist ...
and Zika virus
''Zika virus'' (ZIKV; pronounced or ) is a member of the virus family ''Flaviviridae''. It is spread by daytime-active '' Aedes'' mosquitoes, such as '' A. aegypti'' and '' A. albopictus''. Its name comes from the Ziika Forest of Uganda, w ...
have also been associated with episodes of GBS. Previous hepatitis E virus
The hepatitis E virus (HEV) is the causative agent of hepatitis E. It is of the species ''Orthohepevirus A.''
Globally, approximately 939 million corresponding to 1 in 8 individuals have ever experienced HEV infection. About 15–110 million ind ...
infection has been found to be more common in people with GBS.
Vaccine onset
An increased incidence of Guillain–Barré syndrome followed influenza immunization that followed the1976 swine flu outbreak
In 1976, an outbreak of the swine flu, influenza A virus subtype H1N1 at Fort Dix, New Jersey caused one death, hospitalized 13, and led to a mass immunization program. After the program began, the vaccine was associated with an increase in report ...
(H1N1 A/NJ/76); 8.8 cases per million (0.0088 per 1000) recipients developed it as a complication. GBS cases occurred in 362 patients during the 6 weeks after influenza vaccination of 45 million persons, an 8.8-fold increase over normal rates. The 1976 swine flu vaccination-induced GBS was an outlier; small increases in incidence have been observed in subsequent vaccination campaigns, but not to the same extent. The 2009 flu pandemic vaccine against pandemic swine flu virus H1N1/PDM09 did not cause a significant increase in cases. In fact, "studies found a small increase of approximately 1 case per million vaccines above the baseline rate, which is similar to that observed after administration of seasonal influenza vaccines over the past several years." Natural influenza infection is a stronger risk factor for the development of GBS than is influenza vaccination and the vaccination reduced the risk of GBS overall by lowering the risk of catching influenza.
In the United States, GBS after seasonal influenza vaccination is listed on the federal government's vaccine injury table. On March 24, 2021, after reviewing several post-marketing observational studies, where an increased risk of Guillain–Barré syndrome was observed after 42 days following vaccination with the Zoster vaccine
A zoster vaccine is a vaccine that reduces the incidence of herpes zoster (shingles), a disease caused by reactivation of the varicella zoster virus, which is also responsible for chickenpox. Shingles provokes a painful rash with blisters, and ...
Shingrix, the FDA
The United States Food and Drug Administration (FDA or US FDA) is a federal agency of the Department of Health and Human Services. The FDA is responsible for protecting and promoting public health through the control and supervision of food ...
required safety label changes from the manufacturer GlaxoSmithKline
GSK plc, formerly GlaxoSmithKline plc, is a British multinational pharmaceutical and biotechnology company with global headquarters in London, England. Established in 2000 by a merger of Glaxo Wellcome and SmithKline Beecham. GSK is the ten ...
to include warnings for risk of Guillain–Barré syndrome.
COVID-19 infection or vaccine related
GBS has been reported in association withCOVID-19
Coronavirus disease 2019 (COVID-19) is a contagious disease caused by a virus, the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The first known case was COVID-19 pandemic in Hubei, identified in Wuhan, China, in December ...
, and may be a potential neurological complication of the disease.
GBS has been reported as a very rare side effect of the Janssen and the Oxford–AstraZeneca COVID-19 vaccine for COVID-19 and European Medicines Agency (EMA) had issued warning to the patients and healthcare providers. The incidence of GBS following the vaccination with the Oxford-AstraZeneca vaccine was originally reported as being lower than the incidence of GBS following a COVID-19 infection. More recent studies, however, found no measurable link between COVID-19 infection and GBS, while correlations with a first dose of AstraZeneca or Janssen vaccines were still positive.
COVID-19 has been reported as causing peripheral neuropathy and more recently some evidence of aggravation of autoimmune disorders including GBS. Some studies are now finding Parkinson's Disease is more common in infection survivors.
Drug induced
Zimelidine
Zimelidine (INN, BAN) (brand names Zimeldine, Normud, Zelmid) was one of the first selective serotonin reuptake inhibitor (SSRI) antidepressants to be marketed. It is a pyridylallylamine, and is structurally different from other antidepressants ...
, an antidepressant, had a very favorable safety profile but as a result of rare case reports of Guillain–Barré syndrome was withdrawn from the market.
Mechanism
 The nerve dysfunction in Guillain–Barré syndrome is caused by an immune attack on the nerve cells of the peripheral nervous system and their support structures. The nerve cells have their body (the soma) in the
The nerve dysfunction in Guillain–Barré syndrome is caused by an immune attack on the nerve cells of the peripheral nervous system and their support structures. The nerve cells have their body (the soma) in the spinal cord
The spinal cord is a long, thin, tubular structure made up of nervous tissue, which extends from the medulla oblongata in the brainstem to the lumbar region of the vertebral column (backbone). The backbone encloses the central canal of the spi ...
and a long projection (the axon
An axon (from Greek ἄξων ''áxōn'', axis), or nerve fiber (or nerve fibre: see spelling differences), is a long, slender projection of a nerve cell, or neuron, in vertebrates, that typically conducts electrical impulses known as action po ...
) that carries electrical nerve impulses to the neuromuscular junction
A neuromuscular junction (or myoneural junction) is a chemical synapse between a motor neuron and a muscle fiber.
It allows the motor neuron to transmit a signal to the muscle fiber, causing muscle contraction.
Muscles require innervation to ...
, where the impulse is transferred to the muscle. Axons are wrapped in a sheath of Schwann cell
Schwann cells or neurolemmocytes (named after German physiologist Theodor Schwann) are the principal glia of the peripheral nervous system (PNS). Glial cells function to support neurons and in the PNS, also include satellite cells, olfactory ensh ...
s that contain myelin
Myelin is a lipid-rich material that surrounds nerve cell axons (the nervous system's "wires") to insulate them and increase the rate at which electrical impulses (called action potentials) are passed along the axon. The myelinated axon can be ...
. Between Schwann cells are gaps (nodes of Ranvier
In neuroscience and anatomy, nodes of Ranvier ( ), also known as myelin-sheath gaps, occur along a myelinated axon where the axolemma is exposed to the extracellular space. Nodes of Ranvier are uninsulated and highly enriched in ion channels, al ...
) where the axon is exposed. Different types of Guillain–Barré syndrome feature different types of immune attack. The demyelinating variant (AIDP, see below) features damage to the myelin sheath by white blood cell
White blood cells, also called leukocytes or leucocytes, are the cell (biology), cells of the immune system that are involved in protecting the body against both infectious disease and foreign invaders. All white blood cells are produced and de ...
s (T lymphocytes
A T cell is a type of lymphocyte. T cells are one of the important white blood cells of the immune system and play a central role in the adaptive immune response. T cells can be distinguished from other lymphocytes by the presence of a T-cell rec ...
and macrophage
Macrophages (abbreviated as M φ, MΦ or MP) ( el, large eaters, from Greek ''μακρός'' (') = large, ''φαγεῖν'' (') = to eat) are a type of white blood cell of the immune system that engulfs and digests pathogens, such as cancer cel ...
s); this process is preceded by activation of a group of blood proteins known as complement
A complement is something that completes something else.
Complement may refer specifically to:
The arts
* Complement (music), an interval that, when added to another, spans an octave
** Aggregate complementation, the separation of pitch-clas ...
. In contrast, the axonal variant is mediated by IgG antibodies
Immunoglobulin G (Ig G) is a type of antibody. Representing approximately 75% of serum antibodies in humans, IgG is the most common type of antibody found in blood circulation. IgG molecules are created and released by plasma B cells. Each IgG ...
and complement against the cell membrane covering the axon without direct lymphocyte involvement.
Various antibodies directed at nerve cells have been reported in Guillain–Barré syndrome. In the axonal subtype, these antibodies have been shown to bind to ganglioside
A ganglioside is a molecule composed of a glycosphingolipid (ceramide and oligosaccharide) with one or more sialic acids (e.g. ''N''-acetylneuraminic acid, NANA) linked on the sugar chain. NeuNAc, an acetylated derivative of the carbohydrate si ...
s, a group of substances found in peripheral nerves. A ganglioside is a molecule consisting of ceramide
Ceramides are a family of waxy lipid molecules. A ceramide is composed of N-acetylsphingosine and a fatty acid. Ceramides are found in high concentrations within the cell membrane of eukaryotic cells, since they are component lipids that make up ...
bound to a small group of hexose
In chemistry, a hexose is a monosaccharide (simple sugar) with six carbon atoms. The chemical formula for all hexoses is C6H12O6, and their molecular weight is 180.156 g/mol.
Hexoses exist in two forms, open-chain or cyclic, that easily convert ...
-type sugars and containing various numbers of ''N''-acetylneuraminic acid groups. The key four gangliosides against which antibodies have been described are GM1
GM1 (monosialotetrahexosylganglioside) the "prototype" ganglioside, is a member of the ganglio series of gangliosides which contain one sialic acid residue. GM1 has important physiological properties and impacts neuronal plasticity and repair m ...
, GD1a, GT1a, and GQ1b, with different antiganglioside antibodies being associated with particular features; for instance, GQ1b antibodies have been linked with Miller Fisher variant GBS and related forms including Bickerstaff encephalitis. The production of these antibodies after an infection probably is the result of molecular mimicry Molecular mimicry is defined as the theoretical possibility that sequence similarities between foreign and self-peptides are sufficient to result in the cross-activation of autoreactive T or B cells by pathogen-derived peptides. Despite the preva ...
, where the immune system is reacting to microbial substances, but the resultant antibodies also react with substances occurring naturally in the body. After a ''Campylobacter'' infection, the body produces antibodies of the IgA class; only a small proportion of people also produce IgG antibodies against bacterial substance cell wall substances (e.g. lipooligosaccharides) that cross react with human nerve cell gangliosides. It is not currently known how this process escapes central tolerance
In immunology, central tolerance (also known as negative selection) is the process of eliminating any ''developing'' T or B lymphocytes that are autoreactive, i.e. reactive to the body itself. Through elimination of autoreactive lymphocytes, to ...
to gangliosides, which is meant to suppress the production of antibodies against the body's own substances. Not all antiganglioside antibodies cause disease, and it has recently been suggested that some antibodies bind to more than one type of epitope
An epitope, also known as antigenic determinant, is the part of an antigen that is recognized by the immune system, specifically by antibodies, B cells, or T cells. The epitope is the specific piece of the antigen to which an antibody binds. The p ...
simultaneously (heterodimeric binding) and that this determines the response. Furthermore, the development of pathogenic antibodies may depend on the presence of other strains of bacteria in the bowel.
It has been suggested that a poor injection technique may also cause a direct injury to the axillary nerves adjacent to the injection site in deltoid muscle that may lead to peripheral neuropathy. The consequent vaccine transfection and translation in the nerves may spur an immune response against nerve cells potentially causing an autoimmune nerve damage, leading to conditions like Guillain–Barré syndrome.
Diagnosis
The diagnosis of Guillain–Barré syndrome depends on findings such as rapid development of muscle paralysis, absent reflexes, absence of fever, and absence of a likely cause. Cerebrospinal fluid analysis (through a lumbar spinal puncture) andnerve conduction studies
A nerve conduction study (NCS) is a medical diagnostic test commonly used to evaluate the function, especially the ability of electrical conduction, of the motor and sensory nerves of the human body. These tests may be performed by medical spec ...
are supportive investigations commonly performed in the diagnosis of GBS. Testing for antiganglioside antibodies is often performed, but their contribution to diagnosis is usually limited. Blood tests are generally performed to exclude the possibility of another cause for weakness, such as a low level of potassium in the blood. An abnormally low level of sodium in the blood is often encountered in Guillain–Barré syndrome. This has been attributed to the inappropriate secretion of antidiuretic hormone, leading to relative retention of water.
In many cases, magnetic resonance imaging
Magnetic resonance imaging (MRI) is a medical imaging technique used in radiology to form pictures of the anatomy and the physiological processes of the body. MRI scanners use strong magnetic fields, magnetic field gradients, and radio wave ...
of the spinal cord is performed to distinguish between Guillain–Barré syndrome and other conditions causing limb weakness, such as spinal cord compression
Spinal cord compression is a form of myelopathy in which the spinal cord is compressed. Causes can be bone fragments from a vertebral fracture, a tumor, abscess, ruptured intervertebral disc or other lesion.
When acute it can cause a medical ...
. If an MRI scan shows enhancement of the nerve root A nerve root (Latin: ''radix nervi'') is the initial segment of a nerve leaving the central nervous system. Nerve roots can be classified as:
*cranial nerves, Cranial nerve roots: the initial or proximal segment of one of the twelve pairs of crania ...
s, this may be indicative of GBS. In children, this feature is present in 95% of scans, but it is not specific to Guillain–Barré syndrome, so other confirmation is also needed.
Spinal fluid
Cerebrospinal fluid
Cerebrospinal fluid (CSF) is a clear, colorless body fluid found within the tissue that surrounds the brain and spinal cord of all vertebrates.
CSF is produced by specialised ependymal cells in the choroid plexus of the ventricles of the bra ...
envelops the brain and the spine, and lumbar puncture or spinal tap is the removal of a small amount of fluid using a needle inserted between the lumbar vertebrae
The lumbar vertebrae are, in human anatomy, the five vertebrae between the rib cage and the pelvis. They are the largest segments of the vertebral column and are characterized by the absence of the foramen transversarium within the transverse p ...
. Characteristic findings in Guillain–Barré syndrome are an elevated protein level, usually greater than 0.55 g/L, and fewer than 10 white blood cells per cubic millimeter of fluid ("albuminocytological dissociation"). This pattern distinguishes Guillain–Barré syndrome from other conditions (such as lymphoma
Lymphoma is a group of blood and lymph tumors that develop from lymphocytes (a type of white blood cell). In current usage the name usually refers to just the cancerous versions rather than all such tumours. Signs and symptoms may include enlar ...
and poliomyelitis
Poliomyelitis, commonly shortened to polio, is an infectious disease caused by the poliovirus. Approximately 70% of cases are asymptomatic; mild symptoms which can occur include sore throat and fever; in a proportion of cases more severe sym ...
) in which both the protein and the cell count are elevated. Elevated CSF protein levels are found in approximately 50% of patients in the first 3 days after onset of weakness, which increases to 80% after the first week.
Repeating the lumbar puncture during the disease course is not recommended. The protein levels may rise after treatment has been administered.
Neurophysiology
Directly assessing nerve conduction of electrical impulses can exclude other causes of acute muscle weakness, as well as distinguish the different types of Guillain–Barré syndrome. Needleelectromyography
Electromyography (EMG) is a technique for evaluating and recording the electrical activity produced by skeletal muscles. EMG is performed using an instrument called an electromyograph to produce a record called an electromyogram. An electromyog ...
(EMG) and nerve conduction studies may be performed. In the first two weeks, these investigations may not show any abnormality. Neurophysiology studies are not required for the diagnosis.
Formal criteria exist for each of the main subtypes of Guillain–Barré syndrome (AIDP and AMAN/AMSAN, see below), but these may misclassify some cases (particularly where there is reversible conduction failure) and therefore changes to these criteria have been proposed. Sometimes, repeated testing may be helpful.
Clinical subtypes
A number of subtypes of Guillain–Barré syndrome are recognized. Despite this, many people have overlapping symptoms that can make the classification difficult in individual cases. All types have partial forms. For instance, some people experience only isolated eye-movement or coordination problems; these are thought to be a subtype of Miller Fisher syndrome and have similar antiganglioside antibody patterns. Other diagnostic entities are often included in the spectrum of Guillain–Barré syndrome. Bickerstaff's brainstem encephalitis (BBE), for instance, is part of the group of conditions now regarded as forms of Miller Fisher syndrome (anti-GQ1b antibody syndrome), as well as a related condition labelled "acute ataxic hypersomnolence" where coordination problems and drowsiness are present but no muscle weakness can be detected. BBE is characterized by the rapid onset of ophthalmoplegia, ataxia, and disturbance of consciousness, and may be associated with absent or decreased tendon reflexes and as well asBabinski's sign
The plantar reflex is a reflex elicited when the sole of the foot is stimulated with a blunt instrument. The reflex can take one of two forms. In healthy adults, the plantar reflex causes a downward response of the hallux (flexion). An upward r ...
. The course of the disease is usually monophasic, but recurrent episodes have been reported. MRI abnormalities in the brainstem have been reported in 11%.
Whether isolated acute sensory loss can be regarded as a form of Guillain–Barré syndrome is a matter of dispute; this is a rare occurrence compared to GBS with muscle weakness but no sensory symptoms.
Treatment
Immunotherapy
Plasmapheresis
Plasmapheresis (from the Greek πλάσμα, ''plasma'', something molded, and ἀφαίρεσις ''aphairesis'', taking away) is the removal, treatment, and return or exchange of blood plasma or components thereof from and to the blood circulati ...
and intravenous immunoglobulins
Immunoglobulin therapy is the use of a mixture of antibodies (normal human immunoglobulin or NHIG) to treat several health conditions. These conditions include primary immunodeficiency, immune thrombocytopenic purpura, chronic inflammatory de ...
(IVIG) are the two main immunotherapy treatments for GBS. Plasmapheresis attempts to reduce the body's attack on the nervous system by filtering antibodies out of the bloodstream. Similarly, administration of IVIG neutralizes harmful antibodies and inflammation. These two treatments are equally effective, but a combination of the two is not significantly better than either alone. Plasmapheresis speeds recovery when used within four weeks of the onset of symptoms. IVIG works as well as plasmapheresis when started within two weeks of the onset of symptoms, and has fewer complications. IVIG is usually used first because of its ease of administration and safety; the risks include occasionally causing liver inflammation, or in rare cases, kidney failure. Glucocorticoids
Glucocorticoids (or, less commonly, glucocorticosteroids) are a class of corticosteroids, which are a class of steroid hormones. Glucocorticoids are corticosteroids that bind to the glucocorticoid receptor that is present in almost every vertebr ...
alone have not been found to be effective in speeding recovery and could potentially delay recovery.
Respiratory failure
Respiratory failure may require intubation of the trachea and breathing support throughmechanical ventilation
Mechanical ventilation, assisted ventilation or intermittent mandatory ventilation (IMV), is the medical term for using a machine called a ventilator to fully or partially provide artificial ventilation. Mechanical ventilation helps move air ...
, generally on an intensive care unit
220px, Intensive care unit
An intensive care unit (ICU), also known as an intensive therapy unit or intensive treatment unit (ITU) or critical care unit (CCU), is a special department of a hospital or health care facility that provides intensiv ...
. The need for ventilatory support can be anticipated by measurement of two spirometry-based breathing tests: the forced vital capacity
Spirometry (meaning ''the measuring of breath'') is the most common of the pulmonary function tests (PFTs). It measures lung function, specifically the amount (volume) and/or speed (flow) of air that can be inhaled and exhaled. Spirometry is he ...
(FVC) and the negative inspiratory force (NIF). An FVC of less than 15 mL per kilogram body weight or an NIF of less than 60 cmH2O are considered markers of severe respiratory failure.
Pain
While pain is common in people with Guillain–Barré syndrome, studies comparing different types ofpain medication
An analgesic drug, also called simply an analgesic (American English), analgaesic (British English), pain reliever, or painkiller, is any member of the group of drugs used to achieve relief from pain (that is, analgesia or pain management). It ...
are insufficient to make a recommendation as to which should be used.
Rehabilitation
Following the acute phase, around 40% of people require intensive rehabilitation with the help of a multidisciplinary team to focus on improvingactivities of daily living
Activity may refer to:
* Action (philosophy), in general
* Human activity: human behavior, in sociology behavior may refer to all basic human actions, economics may study human economic activities and along with cybernetics and psychology may st ...
(ADLs). Studies into the subject have been limited, but it is likely that intensive rehabilitation improves long-term symptoms. Teams may include physical therapists
Physical therapy (PT), also known as physiotherapy, is one of the allied health professions. It is provided by physical therapists who promote, maintain, or restore health through physical examination, diagnosis, management, prognosis, patien ...
, occupational therapists
Occupational therapists (OTs) are health care professionals specializing in occupational therapy and occupational science. OTs and occupational therapy assistants (OTAs) use scientific bases and a holistic perspective to promote a person's abili ...
, speech language pathologists, social workers, psychologist
A psychologist is a professional who practices psychology and studies mental states, perceptual, cognitive, emotional, and social processes and behavior. Their work often involves the experimentation, observation, and interpretation of how indi ...
s, other allied health professionals and nurses
Nursing is a profession within the health care sector focused on the care of individuals, families, and communities so they may attain, maintain, or recover optimal health and quality of life. Nurses may be differentiated from other health c ...
. The team usually works under the supervision of a neurologist or rehabilitation physician directing treatment goals.
Physiotherapy interventions include strength, endurance, and gait training with graduated increases in mobility, maintenance of posture and alignment as well as joint function. Occupational therapy aims to improve everyday function with domestic and community tasks as well as driving and work. Home modifications, gait aids, orthotics
Orthotics ( el, Ορθός, translit=ortho, lit=to straighten, to align) is a medical specialty that focuses on the design and application of orthoses, or braces. An is "an externally applied device used to influence the structural and functio ...
, and splints may be provided. Speech-language pathology input may be required in those with speech and swallowing problems, as well as to support communication in those who require ongoing breathing support (often through a tracheostomy
Tracheotomy (, ), or tracheostomy, is a surgical airway management procedure which consists of making an incision (cut) on the anterior aspect (front) of the neck and opening a direct airway through an incision in the trachea (windpipe). The r ...
). Nutritional support may be provided by the team and by dietitian
A dietitian, medical dietitian, or dietician is an expert in identifying and treating disease-related malnutrition and in conducting medical nutrition therapy, for example designing an enteral tube feeding regimen or mitigating the effects of ca ...
s. Psychologists may provide counseling and support. Psychological interventions may also be required for anxiety, fear, and depression.
Prognosis
Guillain–Barré syndrome can lead to death as a result of many complications: severe infections, blood clots, and cardiac arrest likely due to autonomic neuropathy. Despite optimum care, this occurs in about 5% of cases. There is a variation in the rate and extent of recovery. The prognosis of Guillain–Barré syndrome is determined mainly by age (those over 40 may have a poorer outcome), and by the severity of symptoms after two weeks. Furthermore, those who experienced diarrhea before the onset of the disease have a worse prognosis. On the nerve conduction study, the presence of conduction block predicts poorer outcome at 6 months. In those who have received intravenous immunoglobulins, a smaller increase in IgG in the blood two weeks after administration is associated with poorer mobility outcomes at six months than those whose IgG level increased substantially. If the disease continues to progress beyond four weeks, or there are multiple fluctuations in the severity (more than two in eight weeks), the diagnosis may bechronic inflammatory demyelinating polyneuropathy
Chronic inflammatory demyelinating polyneuropathy (CIDP) is an acquired autoimmune disease of the peripheral nervous system characterized by progressive weakness and impaired sensory function in the legs and arms. The disorder is sometimes called c ...
, which is treated differently.
In research studies, the outcome from an episode of Guillain–Barré syndrome is recorded on a scale from 0 to 6, where 0 denotes completely healthy; 1 very minor symptoms but able to run; 2 able to walk but not to run; 3 requiring a stick or other support; 4 confined to bed or chair; 5 requiring long-term respiratory support; 6 death.
The health-related quality of life (HRQL) after an attack of Guillain–Barré syndrome can be significantly impaired. About a fifth are unable to walk unaided after six months, and many experience chronic pain
Chronic pain is classified as pain that lasts longer than three to six months. In medicine, the distinction between Acute (medicine), acute and Chronic condition, chronic pain is sometimes determined by the amount of time since onset. Two commonly ...
, fatigue
Fatigue describes a state of tiredness that does not resolve with rest or sleep. In general usage, fatigue is synonymous with extreme tiredness or exhaustion that normally follows prolonged physical or mental activity. When it does not resolve ...
and difficulty with work, education, hobbies and social activities. HRQL improves significantly in the first year.
Epidemiology
In Western countries, the number of new episodes per year has been estimated to be between 0.89 and 1.89 cases per 100,000 people. Children and young adults are less likely to be affected than the elderly: the relative risk increases by 20% for every decade of life. Men are more likely to develop Guillain–Barré syndrome than women; the relative risk for men is 1.78 compared to women. The distribution of subtypes varies between countries. In Europe and the United States, 60–80% of people with Guillain–Barré syndrome have the demyelinating subtype (AIDP), and AMAN affects only a small number (6–7%). In Asia and Central and South America, that proportion is significantly higher (30–65%). This may be related to the exposure to different kinds of infection, but also the genetic characteristics of that population. Miller Fisher variant is thought to be more common in Southeast Asia.History
 Jean-Baptiste Octave Landry first described the disorder in 1859. In 1916,
Jean-Baptiste Octave Landry first described the disorder in 1859. In 1916, Georges Guillain
Georges Charles Guillain () (3 March 1876 – 29 June 1961) was a French neurologist born in Rouen.
He studied medicine in Rouen and Paris, where he learned clinical education at several hospitals. He developed an interest in neurology, and his ...
, Jean Alexandre Barré
Jean Alexandre Barré (25 May 1880, Nantes – 26 April 1967, Strasbourg) was a French neurologist who in 1916 worked on the identification of Guillain-Barré-Strohl syndrome, as well as Barré–Liéou syndrome.
Biography First studies
He st ...
, and André Strohl
André Strohl (20 March 1887 – 10 March 1977) was a French physiologist who was a native of Poitiers. He is remembered for his role in the diagnosis of Guillain–Barré syndrome (sometimes called Guillain–Barré–Strohl syndrome), a form o ...
diagnosed two soldiers with the illness and described the key diagnostic abnormality—albuminocytological dissociation—of increased spinal fluid protein concentration but a normal cell count.
C. Miller Fisher described the variant that bears his name in 1956. British neurologist Edwin Bickerstaff described the encephalitis
Encephalitis is inflammation of the brain. The severity can be variable with symptoms including reduction or alteration in consciousness, headache, fever, confusion, a stiff neck, and vomiting. Complications may include seizures, hallucinations, ...
type in 1951 and made further contributions with another paper in 1957. Guillain had reported on some of these features before their full description in 1938. Further subtypes have been described since then, such as the form featuring pure ataxia and the type causing pharyngeal-cervical-brachial weakness. The axonal subtype was first described in 1986.
Diagnostic criteria were developed in the late 1970s after the series of cases associated with swine flu
Swine influenza is an infection caused by any of several types of swine influenza viruses. Swine influenza virus (SIV) or swine-origin influenza virus (S-OIV) refers to any strain of the influenza family of viruses that is endemic in pigs. As ...
vaccination. These were refined in 1990. The case definition was revised by the Brighton Collaboration for vaccine safety in 2009, but is mainly intended for research. Plasma exchange was first used in 1978, and its benefit was confirmed in larger studies in 1985. Intravenous immunoglobulins were introduced in 1988, and studies in the early 1990s demonstrated that they were no less effective than plasma exchange.
Research directions
The understanding of the disease mechanism of Guillain–Barré syndrome has evolved in recent years. Development of new treatments has been limited since immunotherapy was introduced in the 1980s and 1990s. Current research is aimed at demonstrating whether some people who have received IVIg might benefit from a second course if the antibody levels measured in blood after treatment have shown only a small increase. Studies of the immunosuppressive drugsmycophenolate mofetil
Mycophenolic acid (MPA) is an immunosuppressant medication used to prevent rejection following organ transplantation and to treat autoimmune conditions such as Crohn's disease and lupus. Specifically it is used following kidney, heart, and liv ...
, brain-derived neurotrophic factor
Brain-derived neurotrophic factor (BDNF), or abrineurin, is a protein found in the and the periphery. that, in humans, is encoded by the ''BDNF'' gene. BDNF is a member of the neurotrophin family of growth factors, which are related to the cano ...
and interferon beta
The type-I interferons (IFN) are cytokines which play essential roles in inflammation, immunoregulation, tumor cells recognition, and T-cell responses. In the human genome, a cluster of thirteen functional IFN genes is located at the 9p21.3 cyto ...
(IFN-β) have not demonstrated benefit to support their widespread use.
An animal model (experimental autoimmune neuritis in rats) is often used for studies, and some agents have shown promise: glatiramer acetate
Glatiramer acetate (also known as Copolymer 1, Cop-1), sold under the brand name Copaxone among others, is an immunomodulator medication used to treat multiple sclerosis. Glatiramer acetate is approved in the United States to reduce the frequenc ...
, quinpramine, fasudil
Fasudil (INN) is a potent Rho-kinase inhibitor and vasodilator. Since it was discovered, it has been used for the treatment of cerebral vasospasm, which is often due to subarachnoid hemorrhage, as well as to improve the cognitive decline seen ...
(an inhibitor of the Rho-kinase enzyme), and the heart drug flecainide
Flecainide is a medication used to prevent and treat abnormally fast heart rates. This includes ventricular and supraventricular tachycardias. Its use is only recommended in those with dangerous arrhythmias or when significant symptoms cannot ...
. An antibody targeted against the anti-GD3 antiganglioside antibody has shown benefit in laboratory research. Given the role of the complement system in GBS, it has been suggested that complement inhibitors (such as the drug eculizumab
Eculizumab, sold under the brand name Soliris among others, is a medication used to treat paroxysmal nocturnal hemoglobinuria (PNH), atypical hemolytic uremic syndrome (aHUS), generalized myasthenia gravis, and neuromyelitis optica. In people ...
) may be effective.
In animals it is called acute polyradiculoneuritis or "coonhound paralysis", and may onset in the coonhound
A coonhound, colloquially a coon dog, is a type of scenthound, a member of the hound group. They are an American type of hunting dog developed for the hunting of raccoons, hence their name, and also for feral pigs, bobcats, cougars, and bears. ...
7 to 10 days after transmission from raccoons
The raccoon ( or , ''Procyon lotor''), sometimes called the common raccoon to distinguish it from other species, is a mammal native to North America. It is the largest of the procyonid family, having a body length of , and a body weight of . ...
. If the coonhound has not been around raccoons, the disease is called acute idiopathic polyradiculoneuritis.
References
Further reading
*External links
{{DEFAULTSORT:Guillain-Barre Syndrome 1976 health disasters Autoimmune diseases Cytomegalovirus-associated diseases Medical triads Myelin disorders Peripheral nervous system disorders Syndromes affecting the nervous system Wikipedia medicine articles ready to translate Wikipedia neurology articles ready to translate