Cyanobacteria on:
[Wikipedia]
[Google]
[Amazon]
Cyanobacteria (), also known as Cyanophyta, are a phylum of
 Cyanobacteria are a very large and diverse phylum of
Cyanobacteria are a very large and diverse phylum of  Material was copied from this source, which is available under
Material was copied from this source, which is available under
Creative Commons Attribution 4.0 International License
Cyanobacteria are a group of photosynthetic bacteria evolutionarily optimized for environmental conditions of low oxygen. Some species are Cyanobacteria are globally widespread photosynthetic prokaryotes and are major contributors to global biogeochemical cycles. They are the only oxygenic photosynthetic prokaryotes, and prosper in diverse and extreme habitats. They are among the oldest organisms on Earth with fossil records dating back 3.5 billion years. Since then, cyanobacteria have been essential players in the Earth's ecosystems. Planktonic cyanobacteria are a fundamental component of
Cyanobacteria are globally widespread photosynthetic prokaryotes and are major contributors to global biogeochemical cycles. They are the only oxygenic photosynthetic prokaryotes, and prosper in diverse and extreme habitats. They are among the oldest organisms on Earth with fossil records dating back 3.5 billion years. Since then, cyanobacteria have been essential players in the Earth's ecosystems. Planktonic cyanobacteria are a fundamental component of
 Some filamentous species can differentiate into several different
Some filamentous species can differentiate into several different
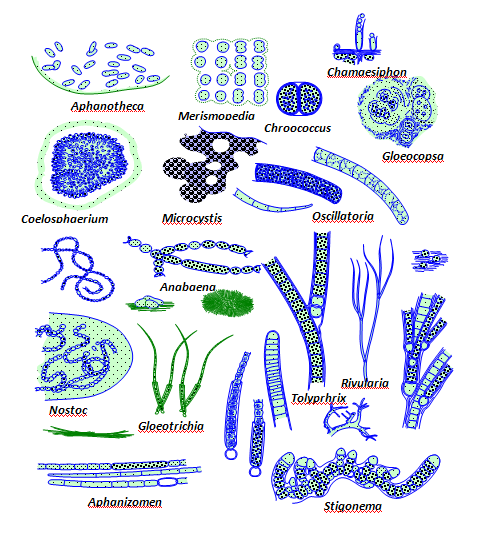 Some cyanobacteria can fix atmospheric
Some cyanobacteria can fix atmospheric

 Cyanobacteria can be found in almost every terrestrial and aquatic habitat – oceans, fresh water, damp soil, temporarily moistened rocks in deserts, bare rock and soil, and even Antarctic rocks. They can occur as planktonic cells or form phototrophic biofilms. They are found inside stones and shells (in endolithic ecosystems). A few are endosymbionts in lichens, plants, various protists, or Sea sponge, sponges and provide energy for the Host (biology), host. Some live in the fur of sloths, providing a form of camouflage.
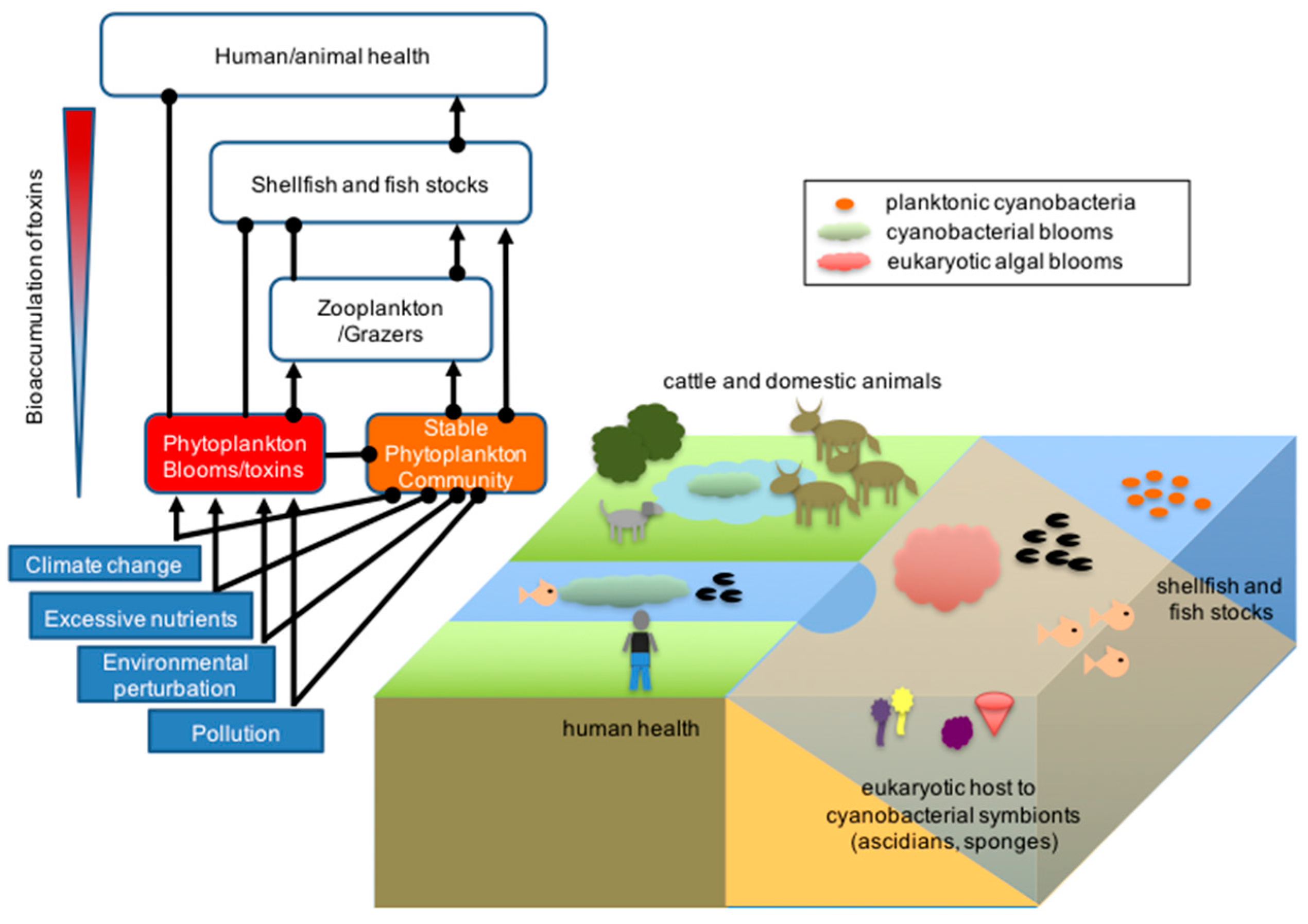
Aquatic cyanobacteria are known for their extensive and highly visible Algal bloom, blooms that can form in both freshwater and marine environments. The blooms can have the appearance of blue-green paint or scum. These blooms can be toxic, and frequently lead to the closure of recreational waters when spotted. Marine bacteriophages are significant parasites of unicellular marine cyanobacteria.
Cyanobacterial growth is favoured in ponds and lakes where waters are calm and have little turbulent mixing. Their lifecycles are disrupted when the water naturally or artificially mixes from churning currents caused by the flowing water of streams or the churning water of fountains. For this reason blooms of cyanobacteria seldom occur in rivers unless the water is flowing slowly. Growth is also favoured at higher temperatures which enable ''Microcystis'' species to outcompete diatoms and green algae, and potentially allow development of toxins.
Based on environmental trends, models and observations suggest cyanobacteria will likely increase their dominance in aquatic environments. This can lead to serious consequences, particularly the contamination of sources of drinking water. Researchers including Linda Lawton at Robert Gordon University, have developed techniques to study these. Cyanobacteria can interfere with water treatment in various ways, primarily by plugging filters (often large beds of sand and similar media) and by producing
Cyanobacteria can be found in almost every terrestrial and aquatic habitat – oceans, fresh water, damp soil, temporarily moistened rocks in deserts, bare rock and soil, and even Antarctic rocks. They can occur as planktonic cells or form phototrophic biofilms. They are found inside stones and shells (in endolithic ecosystems). A few are endosymbionts in lichens, plants, various protists, or Sea sponge, sponges and provide energy for the Host (biology), host. Some live in the fur of sloths, providing a form of camouflage.
Aquatic cyanobacteria are known for their extensive and highly visible Algal bloom, blooms that can form in both freshwater and marine environments. The blooms can have the appearance of blue-green paint or scum. These blooms can be toxic, and frequently lead to the closure of recreational waters when spotted. Marine bacteriophages are significant parasites of unicellular marine cyanobacteria.
Cyanobacterial growth is favoured in ponds and lakes where waters are calm and have little turbulent mixing. Their lifecycles are disrupted when the water naturally or artificially mixes from churning currents caused by the flowing water of streams or the churning water of fountains. For this reason blooms of cyanobacteria seldom occur in rivers unless the water is flowing slowly. Growth is also favoured at higher temperatures which enable ''Microcystis'' species to outcompete diatoms and green algae, and potentially allow development of toxins.
Based on environmental trends, models and observations suggest cyanobacteria will likely increase their dominance in aquatic environments. This can lead to serious consequences, particularly the contamination of sources of drinking water. Researchers including Linda Lawton at Robert Gordon University, have developed techniques to study these. Cyanobacteria can interfere with water treatment in various ways, primarily by plugging filters (often large beds of sand and similar media) and by producing  Cyanobacteria have been found to play an important role in terrestrial habitats. It has been widely reported that cyanobacteria soil crusts help to stabilize soil to prevent erosion and retain water. An example of a cyanobacterial species that does so is ''Microcoleus vaginatus''. ''M. vaginatus'' stabilizes soil using a polysaccharide sheath that binds to sand particles and absorbs water.
Some of these organisms contribute significantly to global ecology and the oxygen cycle. The tiny marine cyanobacterium ''
Cyanobacteria have been found to play an important role in terrestrial habitats. It has been widely reported that cyanobacteria soil crusts help to stabilize soil to prevent erosion and retain water. An example of a cyanobacterial species that does so is ''Microcoleus vaginatus''. ''M. vaginatus'' stabilizes soil using a polysaccharide sheath that binds to sand particles and absorbs water.
Some of these organisms contribute significantly to global ecology and the oxygen cycle. The tiny marine cyanobacterium ''
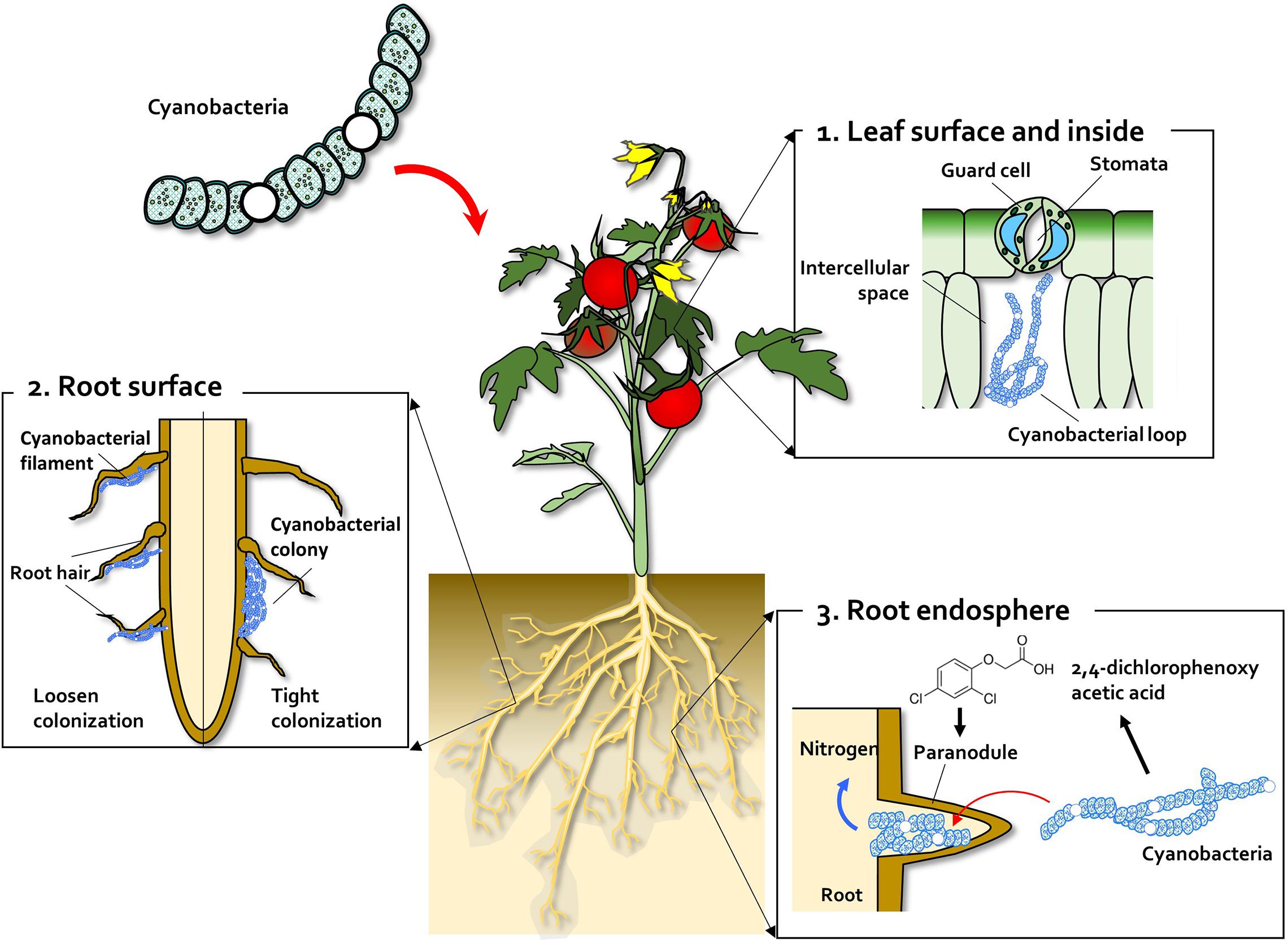 Some cyanobacteria, the so-called cyanobionts (cyanobacterial symbionts), have a symbiotic relationship with other organisms, both unicellular and multicellular. As illustrated on the right, there are many examples of cyanobacteria interacting symbiotically with land plants. Cyanobacteria can enter the plant through the stomata and colonize the intercellular space, forming loops and intracellular coils. ''
Some cyanobacteria, the so-called cyanobionts (cyanobacterial symbionts), have a symbiotic relationship with other organisms, both unicellular and multicellular. As illustrated on the right, there are many examples of cyanobacteria interacting symbiotically with land plants. Cyanobacteria can enter the plant through the stomata and colonize the intercellular space, forming loops and intracellular coils. '' The relationships between cyanobionts (cyanobacterial symbionts) and protistan hosts are particularly noteworthy, as some nitrogen-fixing cyanobacteria (diazotrophs) play an important role in Marine primary production, primary production, especially in nitrogen-limited oligotrophic oceans. Cyanobacteria, mostly pico-sized ''
The relationships between cyanobionts (cyanobacterial symbionts) and protistan hosts are particularly noteworthy, as some nitrogen-fixing cyanobacteria (diazotrophs) play an important role in Marine primary production, primary production, especially in nitrogen-limited oligotrophic oceans. Cyanobacteria, mostly pico-sized ''
 Some cyanobacteria – even single-celled ones – show striking collective behaviours and form colonies (or algal bloom, blooms) that can float on water and have important ecological roles. For instance, billions of years ago, communities of marine Paleoproterozoic cyanobacteria could have helped create the biosphere as we know it by burying carbon compounds and allowing the initial build-up of oxygen in the atmosphere. On the other hand, Harmful algal bloom, toxic cyanobacterial blooms are an increasing issue for society, as their toxins can be harmful to animals. Extreme blooms can also deplete water of oxygen and reduce the penetration of sunlight and visibility, thereby compromising the feeding and mating behaviour of light-reliant species.
As shown in the diagram on the right, bacteria can stay in suspension as individual cells, adhere collectively to surfaces to form biofilms, passively sediment, or flocculate to form suspended aggregates. Cyanobacteria are able to produce sulphated polysaccharides (yellow haze surrounding clumps of cells) that enable them to form floating aggregates. In 2021, Maeda et al. discovered that oxygen produced by cyanobacteria becomes trapped in the network of polysaccharides and cells, enabling the microorganisms to form buoyant blooms. It is thought that specific protein fibres known as Pilus, pili (represented as lines radiating from the cells) may act as an additional way to link cells to each other or onto surfaces. Some cyanobacteria also use sophisticated intracellular
Some cyanobacteria – even single-celled ones – show striking collective behaviours and form colonies (or algal bloom, blooms) that can float on water and have important ecological roles. For instance, billions of years ago, communities of marine Paleoproterozoic cyanobacteria could have helped create the biosphere as we know it by burying carbon compounds and allowing the initial build-up of oxygen in the atmosphere. On the other hand, Harmful algal bloom, toxic cyanobacterial blooms are an increasing issue for society, as their toxins can be harmful to animals. Extreme blooms can also deplete water of oxygen and reduce the penetration of sunlight and visibility, thereby compromising the feeding and mating behaviour of light-reliant species.
As shown in the diagram on the right, bacteria can stay in suspension as individual cells, adhere collectively to surfaces to form biofilms, passively sediment, or flocculate to form suspended aggregates. Cyanobacteria are able to produce sulphated polysaccharides (yellow haze surrounding clumps of cells) that enable them to form floating aggregates. In 2021, Maeda et al. discovered that oxygen produced by cyanobacteria becomes trapped in the network of polysaccharides and cells, enabling the microorganisms to form buoyant blooms. It is thought that specific protein fibres known as Pilus, pili (represented as lines radiating from the cells) may act as an additional way to link cells to each other or onto surfaces. Some cyanobacteria also use sophisticated intracellular 
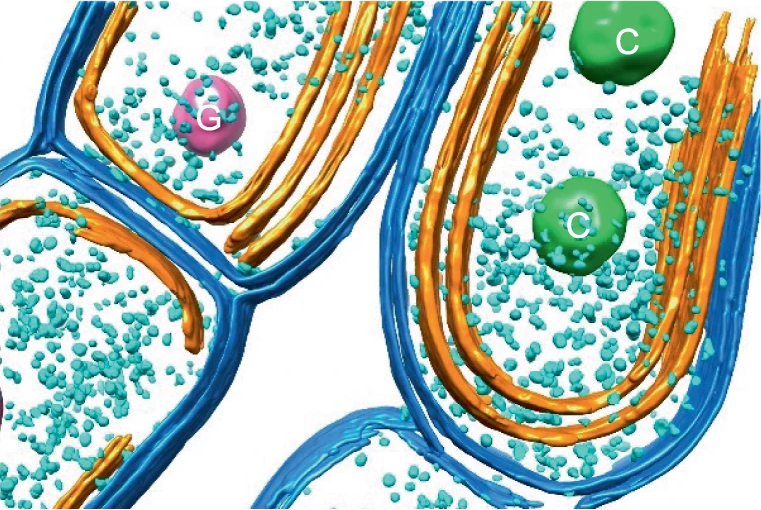
 The diagram on the left above shows a proposed model of microbial distribution, spatial organization, carbon and O2 cycling in clumps and adjacent areas. (a) Clumps contain denser cyanobacterial filaments and heterotrophic microbes. The initial differences in density depend on cyanobacterial motility and can be established over short timescales. Darker blue color outside of the clump indicates higher oxygen concentrations in areas adjacent to clumps. Oxic media increase the reversal frequencies of any filaments that begin to leave the clumps, thereby reducing the net migration away from the clump. This enables the persistence of the initial clumps over short timescales; (b) Spatial coupling between photosynthesis and respiration in clumps. Oxygen produced by cyanobacteria diffuses into the overlying medium or is used for aerobic respiration. Dissolved inorganic carbon (DIC) diffuses into the clump from the overlying medium and is also produced within the clump by respiration. In oxic solutions, high O2 concentrations reduce the efficiency of CO2 fixation and result in the excretion of glycolate. Under these conditions, clumping can be beneficial to cyanobacteria if it stimulates the retention of carbon and the assimilation of inorganic carbon by cyanobacteria within clumps. This effect appears to promote the accumulation of particulate organic carbon (cells, sheaths and heterotrophic organisms) in clumps.
It has been unclear why and how cyanobacteria form communities. Aggregation must divert resources away from the core business of making more cyanobacteria, as it generally involves the production of copious quantities of extracellular material. In addition, cells in the centre of dense aggregates can also suffer from both shading and shortage of nutrients. So, what advantage does this communal life bring for cyanobacteria?
The diagram on the left above shows a proposed model of microbial distribution, spatial organization, carbon and O2 cycling in clumps and adjacent areas. (a) Clumps contain denser cyanobacterial filaments and heterotrophic microbes. The initial differences in density depend on cyanobacterial motility and can be established over short timescales. Darker blue color outside of the clump indicates higher oxygen concentrations in areas adjacent to clumps. Oxic media increase the reversal frequencies of any filaments that begin to leave the clumps, thereby reducing the net migration away from the clump. This enables the persistence of the initial clumps over short timescales; (b) Spatial coupling between photosynthesis and respiration in clumps. Oxygen produced by cyanobacteria diffuses into the overlying medium or is used for aerobic respiration. Dissolved inorganic carbon (DIC) diffuses into the clump from the overlying medium and is also produced within the clump by respiration. In oxic solutions, high O2 concentrations reduce the efficiency of CO2 fixation and result in the excretion of glycolate. Under these conditions, clumping can be beneficial to cyanobacteria if it stimulates the retention of carbon and the assimilation of inorganic carbon by cyanobacteria within clumps. This effect appears to promote the accumulation of particulate organic carbon (cells, sheaths and heterotrophic organisms) in clumps.
It has been unclear why and how cyanobacteria form communities. Aggregation must divert resources away from the core business of making more cyanobacteria, as it generally involves the production of copious quantities of extracellular material. In addition, cells in the centre of dense aggregates can also suffer from both shading and shortage of nutrients. So, what advantage does this communal life bring for cyanobacteria?
 New insights into how cyanobacteria form blooms have come from a 2021 study on the cyanobacterium ''
New insights into how cyanobacteria form blooms have come from a 2021 study on the cyanobacterium ''
"Viability and niche segregation of ''Prochlorococcus'' and ''Synechococcus'' cells across the Central Atlantic Ocean."
Accessed: 30 July 2021). Different forms of cell demise have been observed in cyanobacteria under several stressful conditions, and cell death has been suggested to play a key role in developmental processes, such as akinete and heterocyst differentiation.
 It has long been known that filamentous cyanobacteria perform surface motions, and that these movements result from type IV pili. Additionally, ''
It has long been known that filamentous cyanobacteria perform surface motions, and that these movements result from type IV pili. Additionally, '' Material was copied from this source, which is available under
Material was copied from this source, which is available under
Creative Commons Attribution 4.0 International License
Cells are known to be motile by a gliding method and a novel uncharacterized, nonphototactic swimming method that does not involve flagellar motion. Many species of cyanobacteria are capable of gliding. Gliding motility, Gliding is a form of cell movement that differs from crawling or swimming in that it does not rely on any obvious external organ or change in cell shape and it occurs only in the presence of a substrate (biology), substrate. Gliding in filamentous cyanobacteria appears to be powered by a "slime jet" mechanism, in which the cells extrude a gel that expands quickly as it hydrates providing a propulsion force, although some
File:Stromatolites.jpg, Stromatolites left behind by cyanobacteria are the oldest known fossils of life on Earth. This fossil is one billion years old.
File:Oncolitic limestone (central Utah, USA) 3.jpg, Oncolitic limestone formed from successive layers of calcium carbonate precipitated by cyanobacteria
File:OncolitesAlamoBreccia.jpg, Oncolites from the Late Devonian Alamo bolide impact in Nevada
File:Oscillatoriopsis longa fossil.jpg,
 Historically, bacteria were first classified as plants constituting the class Schizomycetes, which along with the Schizophyceae (blue-green algae/Cyanobacteria) formed the phylum Schizophyta, then in the phylum Monera in the kingdom Protista by Haeckel in 1866, comprising ''Protogens, Protamaeba, Vampyrella, Protomonae'', and ''Vibrio'', but not ''Nostoc'' and other cyanobacteria, which were classified with algae, later reclassified as the ''Prokaryotes'' by Édouard Chatton, Chatton.
The cyanobacteria were traditionally classified by morphology into five sections, referred to by the numerals I–V. The first three – Chroococcales, Pleurocapsales, and Oscillatoriales – are not supported by phylogenetic studies. The latter two – Nostocales and Stigonematales – are monophyletic, and make up the heterocystous cyanobacteria.
The members of Chroococales are unicellular and usually aggregate in colonies. The classic taxonomic criterion has been the cell morphology and the plane of cell division. In Pleurocapsales, the cells have the ability to form internal spores (baeocytes). The rest of the sections include filamentous species. In Oscillatoriales, the cells are uniseriately arranged and do not form specialized cells (akinetes and heterocysts). In Nostocales and Stigonematales, the cells have the ability to develop heterocysts in certain conditions. Stigonematales, unlike Nostocales, include species with truly branched trichomes.
Most taxa included in the phylum or division Cyanobacteria have not yet been validly published under ''The International Code of Nomenclature of Prokaryotes'' (ICNP) except:
*The classes Chroobacteria, Hormogoneae, and Gloeobacteria
*The orders Chroococcales, Gloeobacterales, Nostocales, Oscillatoriales, Pleurocapsales, and Stigonematales
*The families Prochloraceae and Prochlorotrichaceae
*The genera ''Halospirulina, Planktothricoides,
Historically, bacteria were first classified as plants constituting the class Schizomycetes, which along with the Schizophyceae (blue-green algae/Cyanobacteria) formed the phylum Schizophyta, then in the phylum Monera in the kingdom Protista by Haeckel in 1866, comprising ''Protogens, Protamaeba, Vampyrella, Protomonae'', and ''Vibrio'', but not ''Nostoc'' and other cyanobacteria, which were classified with algae, later reclassified as the ''Prokaryotes'' by Édouard Chatton, Chatton.
The cyanobacteria were traditionally classified by morphology into five sections, referred to by the numerals I–V. The first three – Chroococcales, Pleurocapsales, and Oscillatoriales – are not supported by phylogenetic studies. The latter two – Nostocales and Stigonematales – are monophyletic, and make up the heterocystous cyanobacteria.
The members of Chroococales are unicellular and usually aggregate in colonies. The classic taxonomic criterion has been the cell morphology and the plane of cell division. In Pleurocapsales, the cells have the ability to form internal spores (baeocytes). The rest of the sections include filamentous species. In Oscillatoriales, the cells are uniseriately arranged and do not form specialized cells (akinetes and heterocysts). In Nostocales and Stigonematales, the cells have the ability to develop heterocysts in certain conditions. Stigonematales, unlike Nostocales, include species with truly branched trichomes.
Most taxa included in the phylum or division Cyanobacteria have not yet been validly published under ''The International Code of Nomenclature of Prokaryotes'' (ICNP) except:
*The classes Chroobacteria, Hormogoneae, and Gloeobacteria
*The orders Chroococcales, Gloeobacterales, Nostocales, Oscillatoriales, Pleurocapsales, and Stigonematales
*The families Prochloraceae and Prochlorotrichaceae
*The genera ''Halospirulina, Planktothricoides,
 The unicellular cyanobacterium ''
The unicellular cyanobacterium ''
 Some cyanobacteria are sold as food, notably ''Arthrospira platensis (Spirulina (dietary supplement), Spirulina) and others'' (''Aphanizomenon flos-aquae'').
Some microalgae contain substances of high biological value, such as polyunsaturated fatty acids, amino acids, proteins, pigments, antioxidants, vitamins, and minerals. Edible blue-green algae reduce the production of pro-inflammatory cytokines by inhibiting NF-κB pathway in macrophages and splenocytes. Sulfate polysaccharides exhibit immunomodulatory, antitumor, antithrombotic, anticoagulant, anti-mutagenic, anti-inflammatory, antimicrobial, and even antiviral activity against HIV, herpes, and hepatitis.
Some cyanobacteria are sold as food, notably ''Arthrospira platensis (Spirulina (dietary supplement), Spirulina) and others'' (''Aphanizomenon flos-aquae'').
Some microalgae contain substances of high biological value, such as polyunsaturated fatty acids, amino acids, proteins, pigments, antioxidants, vitamins, and minerals. Edible blue-green algae reduce the production of pro-inflammatory cytokines by inhibiting NF-κB pathway in macrophages and splenocytes. Sulfate polysaccharides exhibit immunomodulatory, antitumor, antithrombotic, anticoagulant, anti-mutagenic, anti-inflammatory, antimicrobial, and even antiviral activity against HIV, herpes, and hepatitis.
 Material was copied from this source, which is available under
Material was copied from this source, which is available under
Creative Commons Attribution 4.0 International License
Climate change favours cyanobacterial blooms both directly and indirectly. Many bloom-forming cyanobacteria can grow at relatively high temperatures. Increased thermal stratification of lakes and reservoirs enables buoyant cyanobacteria to float upwards and form dense surface blooms, which gives them better access to light and hence a selective advantage over nonbuoyant phytoplankton organisms. Protracted droughts during summer increase water residence times in reservoirs, rivers and estuaries, and these stagnant warm waters can provide ideal conditions for cyanobacterial bloom development. The capacity of the harmful cyanobacterial genus ''Microcystis'' to adapt to elevated CO2 levels was demonstrated in both laboratory and field experiments. ''Microcystis'' spp. take up CO2 and HCO3− and accumulate inorganic carbon in
File:Lago de coatepeque de color.jpg, Cyanobacteria activity turns Coatepeque Caldera lake a turquoise color
File:2010 Filamentous Cyanobacteria Bloom near Fiji.jpg, Cyanobacterial bloom near Fiji
File:Sinilevää Köyliönjärvessä 3.jpg, Cyanobacteria in Lake Köyliö.
File:Video- The Cyanobacteria- Oscillatoria and Gleocapsa.webm, Video – ''
What are Cyanobacteria and What are its Types?
Webserver for Cyanobacteria Research
Diving an Antarctic Time Capsule Filled With Primordial Life
{{Authority control Cyanobacteria, Phototrophic bacteria Photosynthesis Gram-negative bacteria Environmental chemistry Bacteria phyla
gram-negative bacteria
Gram-negative bacteria are bacteria that do not retain the crystal violet stain used in the Gram staining method of bacterial differentiation. They are characterized by their cell envelopes, which are composed of a thin peptidoglycan cell wall ...
that obtain energy via photosynthesis
Photosynthesis is a process used by plants and other organisms to convert light energy into chemical energy that, through cellular respiration, can later be released to fuel the organism's activities. Some of this chemical energy is stored i ...
. The name ''cyanobacteria'' refers to their color (), which similarly forms the basis of cyanobacteria's common name, blue-green algae, although they are not usually scientifically classified as algae. They appear to have originated in a freshwater or terrestrial environment. Sericytochromatia, the proposed name of the paraphyletic and most basal group, is the ancestor of both the non-photosynthetic group Melainabacteria
Melainabacteria is a phylum related to Cyanobacteria. Organisms belonging to this phylum have been found in the human gut and various aquatic habitats such as groundwater. By analyzing genomes of Melainabacteria, predictions are possible about th ...
and the photosynthetic cyanobacteria, also called Oxyphotobacteria.
Cyanobacteria use photosynthetic pigment
A photosynthetic pigment (accessory pigment; chloroplast pigment; antenna pigment) is a pigment that is present in chloroplasts or photosynthetic bacteria and captures the light energy necessary for photosynthesis.
List of photosynthetic pigme ...
s, such as carotenoids, phycobilins, and various forms of chlorophyll, which absorb energy from light. Unlike heterotroph
A heterotroph (; ) is an organism that cannot produce its own food, instead taking nutrition from other sources of organic carbon, mainly plant or animal matter. In the food chain, heterotrophs are primary, secondary and tertiary consumers, but ...
ic prokaryotes, cyanobacteria have internal membranes. These are flattened sacs called thylakoids
Thylakoids are membrane-bound compartments inside chloroplasts and cyanobacteria. They are the site of the light-dependent reactions of photosynthesis. Thylakoids consist of a thylakoid membrane surrounding a thylakoid lumen. Chloroplast th ...
where photosynthesis is performed. Phototroph
Phototrophs () are organisms that carry out photon capture to produce complex organic compounds (e.g. carbohydrates) and acquire energy. They use the energy from light to carry out various cellular metabolic processes. It is a common misconcep ...
ic eukaryotes such as green plants perform photosynthesis in plastids that are thought to have their ancestry in cyanobacteria, acquired long ago via a process called endosymbiosis
An ''endosymbiont'' or ''endobiont'' is any organism that lives within the body or cells of another organism most often, though not always, in a mutualistic relationship.
(The term endosymbiosis is from the Greek: ἔνδον ''endon'' "within ...
. These endosymbiotic cyanobacteria in eukaryotes then evolved and differentiated into specialized organelles such as chloroplasts, chromoplasts, etioplast
Etioplasts are an intermediate type of plastid that develop from proplastids that have not been exposed to light, and convert into chloroplasts upon exposure to light. They are usually found in stem and leaf tissue of flowering plants (Angiosperms) ...
s, and leucoplast
Leucoplasts (λευκός leukós "white", πλαστός plastós "formed, molded") are a category of plastid and as such are organelles found in plant cells. They are non-pigmented, in contrast to other plastids such as the chloroplast.
Lacki ...
s, collectively known as plastids
The plastid (Greek: πλαστός; plastós: formed, molded – plural plastids) is a membrane-bound organelle found in the cells of plants, algae, and some other eukaryotic organisms. They are considered to be intracellular endosymbiotic cyanobac ...
.
Cyanobacteria are the first organisms known to have produced oxygen. By producing and releasing oxygen as a byproduct of photosynthesis, cyanobacteria are thought to have converted the early oxygen-poor, reducing atmosphere
A reducing atmosphere is an Atmosphere, atmospheric condition in which oxidation is prevented by removal of oxygen and other oxidizing gases or vapours, and which may contain actively Reducing agent, reducing gases such as hydrogen, carbon monoxi ...
into an oxidizing
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a d ...
one, causing the Great Oxidation Event
The Great Oxidation Event (GOE), also called the Great Oxygenation Event, the Oxygen Catastrophe, the Oxygen Revolution, the Oxygen Crisis, or the Oxygen Holocaust, was a time interval during the Paleoproterozoic era when the Earth's atmosphere ...
and the "rusting of the Earth", which dramatically changed the composition of the Earth's life forms.
The cyanobacteria ''Synechocystis
''Synechocystis'' is a genus of unicellular, freshwater cyanobacteria in the family Merismopediaceae. It includes a strain, ''Synechocystis'' sp. PCC 6803, which is a well studied model organism.
Like all cyanobacteria, ''Synechocystis'' ...
'' and ''Cyanothece
''Cyanothece'' is a genus of unicellular, diazotrophic, oxygenic photosynthesizing cyanobacteria.
Modern organisms and cellular organization
In 1976, Jiří Komárek defined the prokaryotic cyanobacteria genus ''Cyanothece'' as distinct from ' ...
'' are important model organisms with potential applications in biotechnology for bioethanol
Ethanol (abbr. EtOH; also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an organic compound. It is an alcohol with the chemical formula . Its formula can be also written as or (an ethyl group linked to a hyd ...
production, food colorings, as a source of human and animal food, dietary supplements and raw materials. Cyanobacteria produce a range of toxins known as cyanotoxins
Cyanotoxins are toxins produced by cyanobacteria (also known as blue-green algae). Cyanobacteria are found almost everywhere, but particularly in lakes and in the ocean where, under high concentration of phosphorus conditions, they reproduce exp ...
that can pose a danger to humans and animals.
Overview
 Cyanobacteria are a very large and diverse phylum of
Cyanobacteria are a very large and diverse phylum of photoautotrophic Photoautotrophs are organisms that use light energy and inorganic carbon to produce organic materials. Eukaryotic photoautotrophs absorb energy through the chlorophyll molecules in their chloroplasts while prokaryotic photoautotrophs use chlorophyll ...
prokaryote
A prokaryote () is a single-celled organism that lacks a nucleus and other membrane-bound organelles. The word ''prokaryote'' comes from the Greek πρό (, 'before') and κάρυον (, 'nut' or 'kernel').Campbell, N. "Biology:Concepts & Conne ...
s. They are defined by their unique combination of pigments
A pigment is a colored material that is completely or nearly insoluble in water. In contrast, dyes are typically soluble, at least at some stage in their use. Generally dyes are often organic compounds whereas pigments are often inorganic compoun ...
and their ability to perform oxygenic photosynthesis
Photosynthesis is a process used by plants and other organisms to convert light energy into chemical energy that, through cellular respiration, can later be released to fuel the organism's activities. Some of this chemical energy is stored in ...
. They often live in colonial aggregates that can take on a multitude of forms. Of particular interest are the filamentous species, which often dominate the upper layers of microbial mat
A microbial mat is a multi-layered sheet of microorganisms, mainly bacteria and archaea, or bacteria alone. Microbial mats grow at interfaces between different types of material, mostly on submerged or moist surfaces, but a few survive in deserts ...
s found in extreme environments such as hot spring
A hot spring, hydrothermal spring, or geothermal spring is a spring produced by the emergence of geothermally heated groundwater onto the surface of the Earth. The groundwater is heated either by shallow bodies of magma (molten rock) or by c ...
s, hypersaline water, deserts and the polar regions, but are also widely distributed in more mundane environments as well. Creative Commons Attribution 4.0 International License
Cyanobacteria are a group of photosynthetic bacteria evolutionarily optimized for environmental conditions of low oxygen. Some species are
nitrogen-fixing
Nitrogen fixation is a chemical process by which molecular nitrogen (), with a strong triple covalent bond, in the air is converted into ammonia () or related nitrogenous compounds, typically in soil or aquatic systems but also in industry. Atm ...
and live in a wide variety of moist soils and water, either freely or in a symbiotic relationship with plants or lichen-forming fungi
A fungus ( : fungi or funguses) is any member of the group of eukaryotic organisms that includes microorganisms such as yeasts and molds, as well as the more familiar mushrooms. These organisms are classified as a kingdom, separately from ...
(as in the lichen genus ''Peltigera
''Peltigera'' is a genus of approximately 100 species of foliose lichens in the family Peltigeraceae. Commonly known as the dog or pelt lichens, species of ''Peltigera'' are often terricolous (growing on soil), but can also occur on moss, trees, ...
''). They range from unicellular
A unicellular organism, also known as a single-celled organism, is an organism that consists of a single cell, unlike a multicellular organism that consists of multiple cells. Organisms fall into two general categories: prokaryotic organisms and ...
to filamentous and include colonial
Colonial or The Colonial may refer to:
* Colonial, of, relating to, or characteristic of a colony or colony (biology)
Architecture
* American colonial architecture
* French Colonial
* Spanish Colonial architecture
Automobiles
* Colonial (1920 au ...
species. Colonies may form filaments, sheets, or even hollow spheres.
 Cyanobacteria are globally widespread photosynthetic prokaryotes and are major contributors to global biogeochemical cycles. They are the only oxygenic photosynthetic prokaryotes, and prosper in diverse and extreme habitats. They are among the oldest organisms on Earth with fossil records dating back 3.5 billion years. Since then, cyanobacteria have been essential players in the Earth's ecosystems. Planktonic cyanobacteria are a fundamental component of
Cyanobacteria are globally widespread photosynthetic prokaryotes and are major contributors to global biogeochemical cycles. They are the only oxygenic photosynthetic prokaryotes, and prosper in diverse and extreme habitats. They are among the oldest organisms on Earth with fossil records dating back 3.5 billion years. Since then, cyanobacteria have been essential players in the Earth's ecosystems. Planktonic cyanobacteria are a fundamental component of marine food web
Compared to terrestrial environments, marine environments have biomass pyramids which are inverted at the base. In particular, the biomass of consumers (copepods, krill, shrimp, forage fish) is larger than the biomass of primary producers. Th ...
s and are major contributors to global carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent—its atom making four electrons available to form covalent chemical bonds. It belongs to group 14 of the periodic table. Carbon mak ...
and nitrogen fluxes. Some cyanobacteria form harmful algal bloom
A harmful algal bloom (HAB) (or excessive algae growth) is an algal bloom that causes negative impacts to other organisms by production of natural phycotoxin, algae-produced toxins, mechanical damage to other organisms, or by other means. HABs are ...
s causing the disruption of aquatic ecosystem services and intoxication of wildlife and humans by the production of powerful toxins (cyanotoxin
Cyanotoxins are toxins produced by cyanobacteria (also known as blue-green algae). Cyanobacteria are found almost everywhere, but particularly in lakes and in the ocean where, under high concentration of phosphorus conditions, they reproduce exp ...
s) such as microcystin
Microcystins—or cyanoginosins—are a class of toxins produced by certain freshwater cyanobacteria, commonly known as blue-green algae. Over 250 different microcystins have been discovered so far, of which microcystin-LR is the most common. C ...
s, saxitoxin
Saxitoxin (STX) is a potent neurotoxin and the best-known paralytic shellfish toxin (PST). Ingestion of saxitoxin by humans, usually by consumption of shellfish contaminated by toxic algal blooms, is responsible for the illness known as paralyt ...
, and cylindrospermopsin
Cylindrospermopsin (abbreviated to CYN, or CYL) is a cyanotoxin produced by a variety of freshwater cyanobacteria. CYN is a polycyclic uracil derivative containing guanidino and sulfate groups. It is also zwitterionic, making it highly water ...
. Nowadays, cyanobacterial blooms pose a serious threat to aquatic environments and public health, and are increasing in frequency and magnitude globally.
Cyanobacteria are ubiquitous in marine environments and play important roles as primary producer
Primary or primaries may refer to:
Arts, entertainment, and media Music Groups and labels
* Primary (band), from Australia
* Primary (musician), hip hop musician and record producer from South Korea
* Primary Music, Israeli record label
Works
* ...
s. Marine phytoplankton today contribute almost half of the Earth's total primary production. Within the cyanobacteria, only a few lineages colonized the open-ocean (i.e., ''Crocosphaera
The Aphanothecaceae is a family of cyanobacteria.
References
Chroococcales
Cyanobacteria families
{{cyanobacteria-stub ...
'' and relatives, cyanobacterium UCYN-A
''Candidatus'' ''Atelocyanobacterium thalassa'', also referred to as UCYN-A, is a diazotrophic species of cyanobacteria commonly found in measurable quantities throughout the world's oceans and some seas. Members of ''A. thalassa'' are spheroid ...
, ''Trichodesmium
''Trichodesmium'', also called sea sawdust, is a genus of Filamentation, filamentous cyanobacteria. They are found in nutrient poor tropical and subtropical ocean waters (particularly around Australia and in the Red Sea, where they were first des ...
'', as well as ''Prochlorococcus
''Prochlorococcus'' is a genus of very small (0.6 μm) marine cyanobacteria with an unusual pigmentation ( chlorophyll ''a2'' and ''b2''). These bacteria belong to the photosynthetic picoplankton and are probably the most abundant photosynth ...
'' and ''Synechococcus
''Synechococcus'' (from the Greek ''synechos'', in succession, and the Greek ''kokkos'', granule) is a unicellular cyanobacterium that is very widespread in the marine environment. Its size varies from 0.8 to 1.5 µm. The photosynthetic c ...
''). From these lineages, nitrogen fixing cyanobacteria are particularly important because they exert a control on primary productivity
In ecology, primary production is the synthesis of organic compounds from atmospheric or aqueous carbon dioxide. It principally occurs through the process of photosynthesis, which uses light as its source of energy, but it also occurs through c ...
and the export of organic carbon to the deep ocean, by converting nitrogen gas into ammonium, which is later used to make amino acids and proteins. Marine picocyanobacteria (i.e., ''Prochlorococcus'' and ''Synechococcus'') numerically dominate most phytoplankton assemblages in modern oceans contributing importantly to primary productivity. While some planktonic cyanobacteria are unicellular and free living cells (e.g., ''Crocosphaera'', ''Prochlorococcus'', ''Synechococcus''), others have established symbiotic relationships with haptophyte algae, such as coccolithophore
Coccolithophores, or coccolithophorids, are single celled organisms which are part of the phytoplankton, the autotrophic (self-feeding) component of the plankton community. They form a group of about 200 species, and belong either to the king ...
s. Amongst the filamentous forms, ''Trichodesmium'' are free-living and form aggregates. However, filamentous heterocyst-forming cyanobacteria (e.g., ''Richelia
''Richelia'' is a genus of nitrogen-fixing, filamentous, heterocystous and cyanobacteria. It contains the single species ''Richelia intracellularis''. They exist as both free-living organisms as well as symbionts within potentially up to 13 ...
'', ''Calothrix
''Calothrix'' is a genus of cyanobacteria
Cyanobacteria (), also known as Cyanophyta, are a phylum of gram-negative bacteria that obtain energy via photosynthesis. The name ''cyanobacteria'' refers to their color (), which similarly forms ...
'') are found in association with diatoms such as ''Hemiaulus'', ''Rhizosolenia'' and ''Chaetoceros
''Chaetoceros'' is probably the largest genus of marine planktonic diatoms with approximately 400 species described, although many of these descriptions are no longer valid. It is often very difficult to distinguish between different ''Chaet ...
''.
Marine cyanobacteria include the smallest known photosynthetic organisms. The smallest of all, ''Prochlorococcus
''Prochlorococcus'' is a genus of very small (0.6 μm) marine cyanobacteria with an unusual pigmentation ( chlorophyll ''a2'' and ''b2''). These bacteria belong to the photosynthetic picoplankton and are probably the most abundant photosynth ...
'', is just 0.5 to 0.8 micrometres across. In terms of individual numbers, ''Prochlorococcus'' is possibly the most plentiful species on Earth: a single millilitre of surface seawater can contain 100,000 cells or more. Worldwide there are estimated to be several octillion
Two naming scales for large numbers have been used in English and other European languages since the early modern era: the long and short scales. Most English variants use the short scale today, but the long scale remains dominant in many non-Eng ...
(1027) individuals. ''Prochlorococcus'' is ubiquitous between 40°N and 40°S and dominates in the oligotroph
An oligotroph is an organism that can live in an environment that offers very low levels of nutrients. They may be contrasted with copiotrophs, which prefer nutritionally rich environments. Oligotrophs are characterized by slow growth, low rates o ...
ic (nutrient poor) regions of the oceans. The bacterium accounts for about 20% of the oxygen in the Earth's atmosphere.
Morphology
Cyanobacteria are variable in morphology, ranging fromunicellular
A unicellular organism, also known as a single-celled organism, is an organism that consists of a single cell, unlike a multicellular organism that consists of multiple cells. Organisms fall into two general categories: prokaryotic organisms and ...
and filamentous to colonial forms. Filamentous forms exhibit functional cell differentiation such as heterocyst
Heterocysts or heterocytes are specialized nitrogen-fixing cells formed during nitrogen starvation by some filamentous cyanobacteria, such as '' Nostoc punctiforme'', ''Cylindrospermum stagnale'', and ''Anabaena sphaerica''. They fix nitrogen fr ...
s (for nitrogen fixation), akinetes (resting stage cells), and hormogonia
Hormogonia are motile filaments of cells formed by some cyanobacteria in the order Nostocales and Stigonematales. They are formed during vegetative reproduction in unicellular, filamentous cyanobacteria, and some may contain heterocysts and ...
(reproductive, motile filaments). These, together with the intercellular connections they possess, are considered the first signs of multicellularity.
Many cyanobacteria form motile filaments of cells, called hormogonia
Hormogonia are motile filaments of cells formed by some cyanobacteria in the order Nostocales and Stigonematales. They are formed during vegetative reproduction in unicellular, filamentous cyanobacteria, and some may contain heterocysts and ...
, that travel away from the main biomass to bud and form new colonies elsewhere. The cells in a hormogonium are often thinner than in the vegetative state, and the cells on either end of the motile chain may be tapered. To break away from the parent colony, a hormogonium often must tear apart a weaker cell in a filament, called a necridium.
 Some filamentous species can differentiate into several different
Some filamentous species can differentiate into several different cell
Cell most often refers to:
* Cell (biology), the functional basic unit of life
Cell may also refer to:
Locations
* Monastic cell, a small room, hut, or cave in which a religious recluse lives, alternatively the small precursor of a monastery ...
types:
* Vegetative cells – the normal, photosynthetic cells that are formed under favorable growing conditions
* Akinete
An akinete is an enveloped, thick-walled, non-motile, dormant cell formed by filamentous, heterocyst-forming cyanobacteria under the order Nostocales and Stigonematales. Akinetes are resistant to cold and desiccation. They also accumulate and sto ...
s – climate-resistant spores that may form when environmental conditions become harsh
* Thick-walled heterocysts
Heterocysts or heterocytes are specialized nitrogen-fixing cells formed during nitrogen starvation by some filamentous cyanobacteria, such as '' Nostoc punctiforme'', ''Cylindrospermum stagnale'', and ''Anabaena sphaerica''. They fix nitrogen f ...
– which contain the enzyme nitrogenase
Nitrogenases are enzymes () that are produced by certain bacteria, such as cyanobacteria (blue-green bacteria) and rhizobacteria. These enzymes are responsible for the Organic redox reaction, reduction of nitrogen (N2) to ammonia (NH3). Nitrog ...
vital for nitrogen fixation
Nitrogen fixation is a chemical process by which molecular nitrogen (), with a strong triple covalent bond, in the air is converted into ammonia () or related nitrogenous compounds, typically in soil or aquatic systems but also in industry. Atmo ...
in an anaerobic environment due to its sensitivity to oxygen.
Each individual cell (each single cyanobacterium) typically has a thick, gelatinous cell wall
A cell wall is a structural layer surrounding some types of cells, just outside the cell membrane. It can be tough, flexible, and sometimes rigid. It provides the cell with both structural support and protection, and also acts as a filtering mech ...
. They lack flagella
A flagellum (; ) is a hairlike appendage that protrudes from certain plant and animal sperm cells, and from a wide range of microorganisms to provide motility. Many protists with flagella are termed as flagellates.
A microorganism may have f ...
, but hormogonia of some species can move about by gliding
Gliding is a recreational activity and competitive air sport in which pilots fly unpowered aircraft known as gliders or sailplanes using naturally occurring currents of rising air in the atmosphere to remain airborne. The word ''soaring'' is al ...
along surfaces. Many of the multicellular filamentous forms of ''Oscillatoria
''Oscillatoria'' is a genus of filamentous cyanobacterium which is often found in freshwater environments, such as hot springs, and appears blue-green. Its name refers to the oscillating motion of its filaments as they slide against each other ...
'' are capable of a waving motion; the filament oscillates back and forth. In water columns, some cyanobacteria float by forming gas vesicle
Gas vesicles, also known as gas vacuoles, are nanocompartments in certain prokaryotic organisms, which help in buoyancy. Gas vesicles are composed entirely of protein; no lipids or carbohydrates have been detected.
Function
Gas vesicles occur ...
s, as in archaea
Archaea ( ; singular archaeon ) is a domain of single-celled organisms. These microorganisms lack cell nuclei and are therefore prokaryotes. Archaea were initially classified as bacteria, receiving the name archaebacteria (in the Archaebac ...
. These vesicles are not organelles as such. They are not bounded by lipid membranes
The lipid bilayer (or phospholipid bilayer) is a thin polar membrane made of two layers of lipid molecules. These membranes are flat sheets that form a continuous barrier around all cells. The cell membranes of almost all organisms and many viru ...
, but by a protein sheath.
Nitrogen fixation
 Some cyanobacteria can fix atmospheric
Some cyanobacteria can fix atmospheric nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
in anaerobic conditions by means of specialized cells called heterocyst
Heterocysts or heterocytes are specialized nitrogen-fixing cells formed during nitrogen starvation by some filamentous cyanobacteria, such as '' Nostoc punctiforme'', ''Cylindrospermum stagnale'', and ''Anabaena sphaerica''. They fix nitrogen fr ...
s. Heterocysts may also form under the appropriate environmental conditions (anoxic) when fixed nitrogen is scarce. Heterocyst-forming species are specialized for nitrogen fixation and are able to fix nitrogen gas into ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogenous was ...
(), nitrites
The nitrite ion has the chemical formula . Nitrite (mostly sodium nitrite) is widely used throughout chemical and pharmaceutical industries. The nitrite anion is a pervasive intermediate in the nitrogen cycle in nature. The name nitrite also r ...
() or nitrates
Nitrate is a polyatomic ion with the chemical formula . Salts containing this ion are called nitrates. Nitrates are common components of fertilizers and explosives. Almost all inorganic nitrates are soluble in water. An example of an insoluble ...
(), which can be absorbed by plants and converted to protein and nucleic acids (atmospheric nitrogen is not bioavailable
In pharmacology, bioavailability is a subcategory of absorption and is the fraction (%) of an administered drug that reaches the systemic circulation.
By definition, when a medication is administered intravenously, its bioavailability is 100%. Ho ...
to plants, except for those having endosymbiotic nitrogen-fixing bacteria
Nitrogen fixation is a chemical process by which molecular nitrogen (), with a strong triple covalent bond, in the air is converted into ammonia () or related nitrogenous compounds, typically in soil or aquatic systems but also in industry. Atmo ...
, especially the family , among
others).
Free-living cyanobacteria are present in the water of rice paddies
A paddy field is a flooded field of arable land used for growing semiaquatic crops, most notably rice and taro. It originates from the Neolithic rice-farming cultures of the Yangtze River basin in southern China, associated with pre-Aust ...
, and cyanobacteria can be found growing as epiphyte
An epiphyte is an organism that grows on the surface of a plant and derives its moisture and nutrients from the air, rain, water (in marine environments) or from debris accumulating around it. The plants on which epiphytes grow are called phoroph ...
s on the surfaces of the green alga, '' Chara'', where they may fix nitrogen. Cyanobacteria such as ''Anabaena
''Anabaena'' is a genus of filamentous cyanobacteria that exist as plankton. They are known for nitrogen-fixing abilities, and they form symbiotic relationships with certain plants, such as the mosquito fern. They are one of four genera of cyanob ...
'' (a symbiont of the aquatic fern ''Azolla
''Azolla'' (mosquito fern, duckweed fern, fairy moss, water fern) is a genus of seven species of aquatic ferns in the family Salviniaceae. They are extremely reduced in form and specialized, looking nothing like other typical ferns but more rese ...
'') can provide rice plantations with biofertilizer
A biofertilizer is a substance which contains living micro-organisms which, when applied to seeds, plant surfaces, or soil, colonize the rhizosphere or the interior of the plant and promotes growth by increasing the supply or availability of prim ...
.
Photosynthesis

Carbon fixation
Cyanobacteria use the energy ofsunlight
Sunlight is a portion of the electromagnetic radiation given off by the Sun, in particular infrared, visible, and ultraviolet light. On Earth, sunlight is scattered and filtered through Earth's atmosphere, and is obvious as daylight when t ...
to drive photosynthesis
Photosynthesis is a process used by plants and other organisms to convert light energy into chemical energy that, through cellular respiration, can later be released to fuel the organism's activities. Some of this chemical energy is stored i ...
, a process where the energy of light is used to synthesize organic compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. The ...
s from carbon dioxide. Because they are aquatic organisms, they typically employ several strategies which are collectively known as a " concentrating mechanism" to aid in the acquisition of inorganic carbon ( or bicarbonate
In inorganic chemistry, bicarbonate (IUPAC-recommended nomenclature: hydrogencarbonate) is an intermediate form in the deprotonation of carbonic acid. It is a polyatomic anion with the chemical formula .
Bicarbonate serves a crucial biochemic ...
). Among the more specific strategies is the widespread prevalence of the bacterial microcompartments known as carboxysome
Carboxysomes are bacterial microcompartments (BMCs) consisting of polyhedral protein shells filled with the enzymes ribulose-1,5-bisphosphate carboxylase/oxygenase (RuBisCO)—the predominant enzyme in carbon fixation and the rate limiting e ...
s, which co-operate with active transporters of CO2 and bicarbonate, in order to accumulate bicarbonate into the cytoplasm of the cell. Carboxysomes are icosahedral
In geometry, an icosahedron ( or ) is a polyhedron with 20 faces. The name comes and . The plural can be either "icosahedra" () or "icosahedrons".
There are infinitely many non- similar shapes of icosahedra, some of them being more symmetrica ...
structures composed of hexameric shell proteins that assemble into cage-like structures that can be several hundreds of nanometres in diameter. It is believed that these structures tether the -fixing enzyme, RuBisCO
Ribulose-1,5-bisphosphate carboxylase-oxygenase, commonly known by the abbreviations RuBisCo, rubisco, RuBPCase, or RuBPco, is an enzyme () involved in the first major step of carbon fixation, a process by which atmospheric carbon dioxide is con ...
, to the interior of the shell, as well as the enzyme carbonic anhydrase
The carbonic anhydrases (or carbonate dehydratases) () form a family of enzymes that catalyze the interconversion between carbon dioxide and water and the dissociated ions of carbonic acid (i.e. bicarbonate and hydrogen ions). The active site ...
, using metabolic channeling to enhance the local concentrations and thus increase the efficiency of the RuBisCO enzyme.
Electron transport
In contrast to purple bacteria and other bacteria performing anoxygenic photosynthesis, thylakoid membranes of cyanobacteria are not continuous with the plasma membrane but are separate compartments. The photosynthetic machinery is embedded in the thylakoid membranes, with phycobilisomes acting as Light-harvesting complex, light-harvesting antennae attached to the membrane, giving the green pigmentation observed (with wavelengths from 450 nm to 660 nm) in most cyanobacteria. While most of the high-energy electrons derived from water are used by the cyanobacterial cells for their own needs, a fraction of these electrons may be donated to the external environment via electrogenic activity.Respiration
Cellular respiration, Respiration in cyanobacteria can occur in the thylakoid membrane alongside photosynthesis, with their photosynthetic Electron transport chain, electron transport sharing the same compartment as the components of respiratory electron transport. While the goal of photosynthesis is to store energy by building carbohydrates from CO2, respiration is the reverse of this, with carbohydrates turned back into CO2 accompanying energy release. Cyanobacteria appear to separate these two processes with their plasma membrane containing only components of the respiratory chain, while the thylakoid membrane hosts an interlinked respiratory and photosynthetic electron transport chain. Cyanobacteria use electrons from succinate dehydrogenase rather than from Nicotinamide adenine dinucleotide phosphate, NADPH for respiration. Cyanobacteria only respire during the night (or in the dark) because the facilities used for electron transport are used in reverse for photosynthesis while in the light.Electron transport chain
Many cyanobacteria are able to reduce nitrogen and carbon dioxide under Aerobic cellular respiration, aerobic conditions, a fact that may be responsible for their evolutionary and ecological success. The water-oxidizing photosynthesis is accomplished by coupling the activity of photosystem (PS) II and I (Z-scheme). In contrast to green sulfur bacteria which only use one photosystem, the use of water as an electron donor is energetically demanding, requiring two photosystems. Attached to the thylakoid membrane, phycobilisomes act as Light-harvesting complex, light-harvesting antennae for the photosystems. The phycobilisome components (phycobiliproteins) are responsible for the blue-green pigmentation of most cyanobacteria. The variations on this theme are due mainly to carotenoids and phycoerythrins that give the cells their red-brownish coloration. In some cyanobacteria, the color of light influences the composition of the phycobilisomes. In green light, the cells accumulate more phycoerythrin, which absorbs green light, whereas in red light they produce more phycocyanin which absorbs red. Thus, these bacteria can change from brick-red to bright blue-green depending on whether they are exposed to green light or to red light. This process of "complementary chromatic adaptation" is a way for the cells to maximize the use of available light for photosynthesis. A few genera lack phycobilisomes and have chlorophyll b instead (''Prochloron'', ''Prochlorococcus
''Prochlorococcus'' is a genus of very small (0.6 μm) marine cyanobacteria with an unusual pigmentation ( chlorophyll ''a2'' and ''b2''). These bacteria belong to the photosynthetic picoplankton and are probably the most abundant photosynth ...
'', ''Prochlorothrix''). These were originally grouped together as the Prochlorophyta, prochlorophytes or chloroxybacteria, but appear to have developed in several different lines of cyanobacteria. For this reason, they are now considered as part of the cyanobacterial group.
Metabolism
In general, photosynthesis in cyanobacteria uses water as an Redox, electron donor and produces oxygen as a byproduct, though some may also use hydrogen sulfide a process which occurs among other photosynthetic bacteria such as the purple sulfur bacteria. Carbon dioxide is reduced to form carbohydrates via the Calvin cycle. The large amounts of oxygen in the atmosphere are considered to have been first created by the activities of ancient cyanobacteria. They are often found as symbionts with a number of other groups of organisms such as fungi (lichens), corals, pteridophytes (''Azolla
''Azolla'' (mosquito fern, duckweed fern, fairy moss, water fern) is a genus of seven species of aquatic ferns in the family Salviniaceae. They are extremely reduced in form and specialized, looking nothing like other typical ferns but more rese ...
''), angiosperms (''Gunnera''), etc.
There are some groups capable of heterotrophic growth, while others are parasitic, causing diseases in invertebrates or algae (e.g., the black band disease).
Ecology
 Cyanobacteria can be found in almost every terrestrial and aquatic habitat – oceans, fresh water, damp soil, temporarily moistened rocks in deserts, bare rock and soil, and even Antarctic rocks. They can occur as planktonic cells or form phototrophic biofilms. They are found inside stones and shells (in endolithic ecosystems). A few are endosymbionts in lichens, plants, various protists, or Sea sponge, sponges and provide energy for the Host (biology), host. Some live in the fur of sloths, providing a form of camouflage.
Aquatic cyanobacteria are known for their extensive and highly visible Algal bloom, blooms that can form in both freshwater and marine environments. The blooms can have the appearance of blue-green paint or scum. These blooms can be toxic, and frequently lead to the closure of recreational waters when spotted. Marine bacteriophages are significant parasites of unicellular marine cyanobacteria.
Cyanobacterial growth is favoured in ponds and lakes where waters are calm and have little turbulent mixing. Their lifecycles are disrupted when the water naturally or artificially mixes from churning currents caused by the flowing water of streams or the churning water of fountains. For this reason blooms of cyanobacteria seldom occur in rivers unless the water is flowing slowly. Growth is also favoured at higher temperatures which enable ''Microcystis'' species to outcompete diatoms and green algae, and potentially allow development of toxins.
Based on environmental trends, models and observations suggest cyanobacteria will likely increase their dominance in aquatic environments. This can lead to serious consequences, particularly the contamination of sources of drinking water. Researchers including Linda Lawton at Robert Gordon University, have developed techniques to study these. Cyanobacteria can interfere with water treatment in various ways, primarily by plugging filters (often large beds of sand and similar media) and by producing
Cyanobacteria can be found in almost every terrestrial and aquatic habitat – oceans, fresh water, damp soil, temporarily moistened rocks in deserts, bare rock and soil, and even Antarctic rocks. They can occur as planktonic cells or form phototrophic biofilms. They are found inside stones and shells (in endolithic ecosystems). A few are endosymbionts in lichens, plants, various protists, or Sea sponge, sponges and provide energy for the Host (biology), host. Some live in the fur of sloths, providing a form of camouflage.
Aquatic cyanobacteria are known for their extensive and highly visible Algal bloom, blooms that can form in both freshwater and marine environments. The blooms can have the appearance of blue-green paint or scum. These blooms can be toxic, and frequently lead to the closure of recreational waters when spotted. Marine bacteriophages are significant parasites of unicellular marine cyanobacteria.
Cyanobacterial growth is favoured in ponds and lakes where waters are calm and have little turbulent mixing. Their lifecycles are disrupted when the water naturally or artificially mixes from churning currents caused by the flowing water of streams or the churning water of fountains. For this reason blooms of cyanobacteria seldom occur in rivers unless the water is flowing slowly. Growth is also favoured at higher temperatures which enable ''Microcystis'' species to outcompete diatoms and green algae, and potentially allow development of toxins.
Based on environmental trends, models and observations suggest cyanobacteria will likely increase their dominance in aquatic environments. This can lead to serious consequences, particularly the contamination of sources of drinking water. Researchers including Linda Lawton at Robert Gordon University, have developed techniques to study these. Cyanobacteria can interfere with water treatment in various ways, primarily by plugging filters (often large beds of sand and similar media) and by producing cyanotoxin
Cyanotoxins are toxins produced by cyanobacteria (also known as blue-green algae). Cyanobacteria are found almost everywhere, but particularly in lakes and in the ocean where, under high concentration of phosphorus conditions, they reproduce exp ...
s, which have the potential to cause serious illness if consumed. Consequences may also lie within fisheries and waste management practices. Anthropogenic eutrophication, rising temperatures, vertical stratification and increased atmospheric carbon dioxide are contributors to cyanobacteria increasing dominance of aquatic ecosystems.
 Cyanobacteria have been found to play an important role in terrestrial habitats. It has been widely reported that cyanobacteria soil crusts help to stabilize soil to prevent erosion and retain water. An example of a cyanobacterial species that does so is ''Microcoleus vaginatus''. ''M. vaginatus'' stabilizes soil using a polysaccharide sheath that binds to sand particles and absorbs water.
Some of these organisms contribute significantly to global ecology and the oxygen cycle. The tiny marine cyanobacterium ''
Cyanobacteria have been found to play an important role in terrestrial habitats. It has been widely reported that cyanobacteria soil crusts help to stabilize soil to prevent erosion and retain water. An example of a cyanobacterial species that does so is ''Microcoleus vaginatus''. ''M. vaginatus'' stabilizes soil using a polysaccharide sheath that binds to sand particles and absorbs water.
Some of these organisms contribute significantly to global ecology and the oxygen cycle. The tiny marine cyanobacterium ''Prochlorococcus
''Prochlorococcus'' is a genus of very small (0.6 μm) marine cyanobacteria with an unusual pigmentation ( chlorophyll ''a2'' and ''b2''). These bacteria belong to the photosynthetic picoplankton and are probably the most abundant photosynth ...
'' was discovered in 1986 and accounts for more than half of the photosynthesis of the open ocean. Circadian rhythms were once thought to only exist in eukaryotic cells but many cyanobacteria display a bacterial circadian rhythm.
"Cyanobacteria are arguably the most successful group of microorganisms on earth. They are the most genetically diverse; they occupy a broad range of habitats across all latitudes, widespread in freshwater, marine, and terrestrial ecosystems, and they are found in the most extreme niches such as hot springs, salt works, and hypersaline bays. Photoautotrophic, oxygen-producing cyanobacteria created the conditions in the planet's early atmosphere that directed the evolution of aerobic metabolism and eukaryotic photosynthesis. Cyanobacteria fulfill vital ecological functions in the world's oceans, being important contributors to global carbon and nitrogen budgets." – Stewart and Falconer
Cyanobionts
 Some cyanobacteria, the so-called cyanobionts (cyanobacterial symbionts), have a symbiotic relationship with other organisms, both unicellular and multicellular. As illustrated on the right, there are many examples of cyanobacteria interacting symbiotically with land plants. Cyanobacteria can enter the plant through the stomata and colonize the intercellular space, forming loops and intracellular coils. ''
Some cyanobacteria, the so-called cyanobionts (cyanobacterial symbionts), have a symbiotic relationship with other organisms, both unicellular and multicellular. As illustrated on the right, there are many examples of cyanobacteria interacting symbiotically with land plants. Cyanobacteria can enter the plant through the stomata and colonize the intercellular space, forming loops and intracellular coils. ''Anabaena
''Anabaena'' is a genus of filamentous cyanobacteria that exist as plankton. They are known for nitrogen-fixing abilities, and they form symbiotic relationships with certain plants, such as the mosquito fern. They are one of four genera of cyanob ...
'' spp. colonize the roots of wheat and cotton plants. ''Calothrix
''Calothrix'' is a genus of cyanobacteria
Cyanobacteria (), also known as Cyanophyta, are a phylum of gram-negative bacteria that obtain energy via photosynthesis. The name ''cyanobacteria'' refers to their color (), which similarly forms ...
'' sp. has also been found on the root system of wheat. Monocots, such as wheat and rice, have been colonised by ''Nostoc'' spp., In 1991, Ganther and others isolated diverse heterocystous nitrogen-fixing cyanobacteria, including ''Nostoc'', ''Anabaena'' and ''Cylindrospermum'', from plant root and soil. Assessment of wheat seedling roots revealed two types of association patterns: loose colonization of root hair by ''Anabaena'' and tight colonization of the root surface within a restricted zone by ''Nostoc''.
 The relationships between cyanobionts (cyanobacterial symbionts) and protistan hosts are particularly noteworthy, as some nitrogen-fixing cyanobacteria (diazotrophs) play an important role in Marine primary production, primary production, especially in nitrogen-limited oligotrophic oceans. Cyanobacteria, mostly pico-sized ''
The relationships between cyanobionts (cyanobacterial symbionts) and protistan hosts are particularly noteworthy, as some nitrogen-fixing cyanobacteria (diazotrophs) play an important role in Marine primary production, primary production, especially in nitrogen-limited oligotrophic oceans. Cyanobacteria, mostly pico-sized ''Synechococcus
''Synechococcus'' (from the Greek ''synechos'', in succession, and the Greek ''kokkos'', granule) is a unicellular cyanobacterium that is very widespread in the marine environment. Its size varies from 0.8 to 1.5 µm. The photosynthetic c ...
'' and ''Prochlorococcus
''Prochlorococcus'' is a genus of very small (0.6 μm) marine cyanobacteria with an unusual pigmentation ( chlorophyll ''a2'' and ''b2''). These bacteria belong to the photosynthetic picoplankton and are probably the most abundant photosynth ...
'', are ubiquitously distributed and are the most abundant photosynthetic organisms on Earth, accounting for a quarter of all carbon fixed in marine ecosystems. In contrast to free-living marine cyanobacteria, some cyanobionts are known to be responsible for nitrogen fixation rather than carbon fixation in the host. However, the physiological functions of most cyanobionts remain unknown. Cyanobionts have been found in numerous protist groups, including dinoflagellates, tintinnids, radiolarians, amoebae, diatoms, and haptophytes. Among these cyanobionts, little is known regarding the nature (e.g., genetic diversity, host or cyanobiont specificity, and cyanobiont seasonality) of the symbiosis involved, particularly in relation to dinoflagellate host.
Collective behaviour
 Some cyanobacteria – even single-celled ones – show striking collective behaviours and form colonies (or algal bloom, blooms) that can float on water and have important ecological roles. For instance, billions of years ago, communities of marine Paleoproterozoic cyanobacteria could have helped create the biosphere as we know it by burying carbon compounds and allowing the initial build-up of oxygen in the atmosphere. On the other hand, Harmful algal bloom, toxic cyanobacterial blooms are an increasing issue for society, as their toxins can be harmful to animals. Extreme blooms can also deplete water of oxygen and reduce the penetration of sunlight and visibility, thereby compromising the feeding and mating behaviour of light-reliant species.
As shown in the diagram on the right, bacteria can stay in suspension as individual cells, adhere collectively to surfaces to form biofilms, passively sediment, or flocculate to form suspended aggregates. Cyanobacteria are able to produce sulphated polysaccharides (yellow haze surrounding clumps of cells) that enable them to form floating aggregates. In 2021, Maeda et al. discovered that oxygen produced by cyanobacteria becomes trapped in the network of polysaccharides and cells, enabling the microorganisms to form buoyant blooms. It is thought that specific protein fibres known as Pilus, pili (represented as lines radiating from the cells) may act as an additional way to link cells to each other or onto surfaces. Some cyanobacteria also use sophisticated intracellular
Some cyanobacteria – even single-celled ones – show striking collective behaviours and form colonies (or algal bloom, blooms) that can float on water and have important ecological roles. For instance, billions of years ago, communities of marine Paleoproterozoic cyanobacteria could have helped create the biosphere as we know it by burying carbon compounds and allowing the initial build-up of oxygen in the atmosphere. On the other hand, Harmful algal bloom, toxic cyanobacterial blooms are an increasing issue for society, as their toxins can be harmful to animals. Extreme blooms can also deplete water of oxygen and reduce the penetration of sunlight and visibility, thereby compromising the feeding and mating behaviour of light-reliant species.
As shown in the diagram on the right, bacteria can stay in suspension as individual cells, adhere collectively to surfaces to form biofilms, passively sediment, or flocculate to form suspended aggregates. Cyanobacteria are able to produce sulphated polysaccharides (yellow haze surrounding clumps of cells) that enable them to form floating aggregates. In 2021, Maeda et al. discovered that oxygen produced by cyanobacteria becomes trapped in the network of polysaccharides and cells, enabling the microorganisms to form buoyant blooms. It is thought that specific protein fibres known as Pilus, pili (represented as lines radiating from the cells) may act as an additional way to link cells to each other or onto surfaces. Some cyanobacteria also use sophisticated intracellular gas vesicle
Gas vesicles, also known as gas vacuoles, are nanocompartments in certain prokaryotic organisms, which help in buoyancy. Gas vesicles are composed entirely of protein; no lipids or carbohydrates have been detected.
Function
Gas vesicles occur ...
s as floatation aids.

 The diagram on the left above shows a proposed model of microbial distribution, spatial organization, carbon and O2 cycling in clumps and adjacent areas. (a) Clumps contain denser cyanobacterial filaments and heterotrophic microbes. The initial differences in density depend on cyanobacterial motility and can be established over short timescales. Darker blue color outside of the clump indicates higher oxygen concentrations in areas adjacent to clumps. Oxic media increase the reversal frequencies of any filaments that begin to leave the clumps, thereby reducing the net migration away from the clump. This enables the persistence of the initial clumps over short timescales; (b) Spatial coupling between photosynthesis and respiration in clumps. Oxygen produced by cyanobacteria diffuses into the overlying medium or is used for aerobic respiration. Dissolved inorganic carbon (DIC) diffuses into the clump from the overlying medium and is also produced within the clump by respiration. In oxic solutions, high O2 concentrations reduce the efficiency of CO2 fixation and result in the excretion of glycolate. Under these conditions, clumping can be beneficial to cyanobacteria if it stimulates the retention of carbon and the assimilation of inorganic carbon by cyanobacteria within clumps. This effect appears to promote the accumulation of particulate organic carbon (cells, sheaths and heterotrophic organisms) in clumps.
It has been unclear why and how cyanobacteria form communities. Aggregation must divert resources away from the core business of making more cyanobacteria, as it generally involves the production of copious quantities of extracellular material. In addition, cells in the centre of dense aggregates can also suffer from both shading and shortage of nutrients. So, what advantage does this communal life bring for cyanobacteria?
The diagram on the left above shows a proposed model of microbial distribution, spatial organization, carbon and O2 cycling in clumps and adjacent areas. (a) Clumps contain denser cyanobacterial filaments and heterotrophic microbes. The initial differences in density depend on cyanobacterial motility and can be established over short timescales. Darker blue color outside of the clump indicates higher oxygen concentrations in areas adjacent to clumps. Oxic media increase the reversal frequencies of any filaments that begin to leave the clumps, thereby reducing the net migration away from the clump. This enables the persistence of the initial clumps over short timescales; (b) Spatial coupling between photosynthesis and respiration in clumps. Oxygen produced by cyanobacteria diffuses into the overlying medium or is used for aerobic respiration. Dissolved inorganic carbon (DIC) diffuses into the clump from the overlying medium and is also produced within the clump by respiration. In oxic solutions, high O2 concentrations reduce the efficiency of CO2 fixation and result in the excretion of glycolate. Under these conditions, clumping can be beneficial to cyanobacteria if it stimulates the retention of carbon and the assimilation of inorganic carbon by cyanobacteria within clumps. This effect appears to promote the accumulation of particulate organic carbon (cells, sheaths and heterotrophic organisms) in clumps.
It has been unclear why and how cyanobacteria form communities. Aggregation must divert resources away from the core business of making more cyanobacteria, as it generally involves the production of copious quantities of extracellular material. In addition, cells in the centre of dense aggregates can also suffer from both shading and shortage of nutrients. So, what advantage does this communal life bring for cyanobacteria?
 New insights into how cyanobacteria form blooms have come from a 2021 study on the cyanobacterium ''
New insights into how cyanobacteria form blooms have come from a 2021 study on the cyanobacterium ''Synechocystis
''Synechocystis'' is a genus of unicellular, freshwater cyanobacteria in the family Merismopediaceae. It includes a strain, ''Synechocystis'' sp. PCC 6803, which is a well studied model organism.
Like all cyanobacteria, ''Synechocystis'' ...
''. These use a set of genes that regulate the production and export of sulphated polysaccharides, chains of sugar molecules modified with sulphate groups that can often be found in marine algae and animal tissue. Many bacteria generate extracellular polysaccharides, but sulphated ones have only been seen in cyanobacteria. In ''Synechocystis'' these sulphated polysaccharide help the cyanobacterium form buoyant aggregates by trapping oxygen bubbles in the slimy web of cells and polysaccharides.
Previous studies on ''Synechocystis'' have shown type IV pili, which decorate the surface of cyanobacteria, also play a role in forming blooms. These retractable and adhesive protein fibres are important for motility, adhesion to substrates and DNA uptake. The formation of blooms may require both type IV pili and Synechan – for example, the pili may help to export the polysaccharide outside the cell. Indeed, the activity of these protein fibres may be connected to the production of extracellular polysaccharides in filamentous cyanobacteria. A more obvious answer would be that pili help to build the aggregates by binding the cells with each other or with the extracellular polysaccharide. As with other kinds of bacteria, certain components of the pili may allow cyanobacteria from the same species to recognise each other and make initial contacts, which are then stabilised by building a mass of extracellular polysaccharide.
The bubble flotation mechanism identified by Maeda et al. joins a range of known strategies that enable cyanobacteria to control their buoyancy, such as using gas vesicles or accumulating carbohydrate ballasts. Type IV pili on their own could also control the position of marine cyanobacteria in the water column by regulating viscous drag. Extracellular polysaccharide appears to be a multipurpose asset for cyanobacteria, from floatation device to food storage, defence mechanism and mobility aid.
Cellular death
One of the most critical processes determining cyanobacterial eco-physiology is cellular death. Evidence supports the existence of controlled cellular demise in cyanobacteria, and various forms of cell death have been described as a response to biotic and abiotic stresses. However, cell death research in cyanobacteria is a relatively young field and understanding of the underlying mechanisms and molecular machinery underpinning this fundamental process remains largely elusive. However, reports on cell death of marine and freshwater cyanobacteria indicate this process has major implications for the ecology of microbial communities/Agustí, S. (2004)"Viability and niche segregation of ''Prochlorococcus'' and ''Synechococcus'' cells across the Central Atlantic Ocean."
Accessed: 30 July 2021). Different forms of cell demise have been observed in cyanobacteria under several stressful conditions, and cell death has been suggested to play a key role in developmental processes, such as akinete and heterocyst differentiation.
Cyanophages
Cyanophages are viruses that infect cyanobacteria. Cyanophages can be found in both freshwater and marine environments. Marine and freshwater cyanophages have Regular icosahedron, icosahedral heads, which contain double-stranded DNA, attached to a tail by connector proteins. The size of the head and tail vary among species of cyanophages. Cyanophages like other bacteriophages rely on Brownian motion to collide with bacteria, and then use receptor binding proteins to recognize cell surface proteins, which leads to adherence. Viruses with contractile tails then rely on receptors found on their tails to recognize highly conserved proteins on the surface of the host cell. Cyanophages infect a wide range of cyanobacteria and are key regulators of the cyanobacterial populations in aquatic environments, and may aid in the prevention of cyanobacterial blooms in freshwater and marine ecosystems. These blooms can pose a danger to humans and other animals, particularly in eutrophic freshwater lakes. Infection by these viruses is highly prevalent in cells belonging to ''Synechococcus
''Synechococcus'' (from the Greek ''synechos'', in succession, and the Greek ''kokkos'', granule) is a unicellular cyanobacterium that is very widespread in the marine environment. Its size varies from 0.8 to 1.5 µm. The photosynthetic c ...
'' spp. in marine environments, where up to 5% of cells belonging to marine cyanobacterial cells have been reported to contain mature phage particles.
The first cyanophage, Cyanophage LPP-1, LPP-1, was discovered in 1963. Cyanophages are classified within the bacteriophage families ''Myoviridae'' (e.g. cyanophage AS-1, AS-1, cyanophage N-1, N-1), ''Podoviridae'' (e.g. LPP-1) and ''Siphoviridae'' (e.g. cyanophage S-1, S-1).Sarma TA. 'Cyanophages' in ''Handbook of Cyanobacteria'' (CRC Press; 2012) ()
Movement
 It has long been known that filamentous cyanobacteria perform surface motions, and that these movements result from type IV pili. Additionally, ''
It has long been known that filamentous cyanobacteria perform surface motions, and that these movements result from type IV pili. Additionally, ''Synechococcus
''Synechococcus'' (from the Greek ''synechos'', in succession, and the Greek ''kokkos'', granule) is a unicellular cyanobacterium that is very widespread in the marine environment. Its size varies from 0.8 to 1.5 µm. The photosynthetic c ...
'', a marine cyanobacteria, is known to swim at a speed of 25 μm/s by a mechanism different to that of bacterial flagella. Formation of waves on the cyanobacteria surface is thought to push surrounding water backwards. Creative Commons Attribution 4.0 International License
Cells are known to be motile by a gliding method and a novel uncharacterized, nonphototactic swimming method that does not involve flagellar motion. Many species of cyanobacteria are capable of gliding. Gliding motility, Gliding is a form of cell movement that differs from crawling or swimming in that it does not rely on any obvious external organ or change in cell shape and it occurs only in the presence of a substrate (biology), substrate. Gliding in filamentous cyanobacteria appears to be powered by a "slime jet" mechanism, in which the cells extrude a gel that expands quickly as it hydrates providing a propulsion force, although some
unicellular
A unicellular organism, also known as a single-celled organism, is an organism that consists of a single cell, unlike a multicellular organism that consists of multiple cells. Organisms fall into two general categories: prokaryotic organisms and ...
cyanobacteria use type IV pili for gliding.
Cyanobacteria have strict light requirements. Too little light can result in insufficient energy production, and in some species may cause the cells to resort to heterotrophic respiration. Too much light can inhibit the cells, decrease photosynthesis efficiency and cause damage by bleaching. UV radiation is especially deadly for cyanobacteria, with normal solar levels being significantly detrimental for these microorganisms in some cases.
Filamentous cyanobacteria that live in microbial mats often migrate vertically and horizontally within the mat in order to find an optimal niche that balances their light requirements for photosynthesis against their sensitivity to photodamage. For example, the filamentous cyanobacteria ''Oscillatoria
''Oscillatoria'' is a genus of filamentous cyanobacterium which is often found in freshwater environments, such as hot springs, and appears blue-green. Its name refers to the oscillating motion of its filaments as they slide against each other ...
'' sp. and ''Spirulina (genus), Spirulina subsalsa'' found in the hypersaline benthic mats of Guerrero Negro, Mexico migrate downwards into the lower layers during the day in order to escape the intense sunlight and then rise to the surface at dusk. In contrast, the population of ''Microcoleus, Microcoleus chthonoplastes'' found in hypersaline mats in Camargue, France migrate to the upper layer of the mat during the day and are spread homogenously through the mat at night. An in vitro experiment using P. uncinatum also demonstrated this species' tendency to migrate in order to avoid damaging radiation. These migrations are usually the result of some sort of photomovement, although other forms of taxis can also play a role.
Photomovement – the modulation of cell movement as a function of the incident light – is employed by the cyanoabacteria as a means to find optimal light conditions in their environment. There are three types of photomovement: photokinesis, phototaxis and photophobic responses.
Photokinetic microorganisms modulate their gliding speed according to the incident light intensity. For example, the speed with which Phormidium autumnale glides increases linearly with the incident light intensity.
Phototactic microorganisms move according to the direction of the light within the environment, such that positively phototactic species will tend to move roughly parallel to the light and towards the light source. Species such as ''Phormidium, Phormidium uncinatum'' cannot steer directly towards the light, but rely on random collisions to orient themselves in the right direction, after which they tend to move more towards the light source. Others, such as ''Anabaena variabilis'', can steer by bending the trichome.
Finally, photophobic microorganisms respond to spatial and temporal light gradients. A step-up photophobic reaction occurs when an organism enters a brighter area field from a darker one and then reverses direction, thus avoiding the bright light. The opposite reaction, called a step-down reaction, occurs when an organism enters a dark area from a bright area and then reverses direction, thus remaining in the light.
Evolution
Earth history
Stromatolites are layered biochemical accretion (geology), accretionary structures formed in shallow water by the trapping, binding, and cementation of sedimentary grains by biofilms (microbial mat
A microbial mat is a multi-layered sheet of microorganisms, mainly bacteria and archaea, or bacteria alone. Microbial mats grow at interfaces between different types of material, mostly on submerged or moist surfaces, but a few survive in deserts ...
s) of microorganisms, especially cyanobacteria.
During the Precambrian, stromatolite communities of microorganisms grew in most marine and non-marine environments in the photic zone. After the Cambrian explosion of marine animals, grazing on the stromatolite mats by herbivores greatly reduced the occurrence of the stromatolites in marine environments. Since then, they are found mostly in hypersaline conditions where grazing invertebrates cannot live (e.g. Shark Bay, Western Australia). Stromatolites provide ancient records of life on Earth by fossil remains which date from 3.5 annus#SI prefix multipliers, Ga ago. the oldest undisputed evidence of cyanobacteria is from 2.1 Ga ago, but there is some evidence for them as far back as 2.7 Ga ago. Oxygen concentrations in the atmosphere remained around or below 1% of today's level until 2.4 Ga ago (the Great Oxygenation Event). The rise in oxygen may have caused a fall in the concentration of atmospheric methane, and triggered the Huronian glaciation from around 2.4 to 2.1 Ga ago. In this way, cyanobacteria may have killed off much of the other bacteria of the time.
Oncolites are sedimentary structures composed of oncoids, which are layered structures formed by cyanobacterial growth. Oncolites are similar to stromatolites, but instead of forming columns, they form approximately spherical structures that were not attached to the underlying substrate as they formed. The oncoids often form around a central nucleus, such as a shell fragment, and a calcium carbonate structure is deposited by encrusting microbes. Oncolites are indicators of warm waters in the photic zone, but are also known in contemporary freshwater environments. These structures rarely exceed 10 cm in diameter.
One former classification scheme of cyanobacterial fossils divided them into the porostromata and the spongiostromata. These are now recognized as form taxa and considered taxonomically obsolete; however, some authors have advocated for the terms remaining informally to describe form and structure of bacterial fossils.
Origin of photosynthesis
As far as we can tell,oxygenic photosynthesis
Photosynthesis is a process used by plants and other organisms to convert light energy into chemical energy that, through cellular respiration, can later be released to fuel the organism's activities. Some of this chemical energy is stored in ...
only evolved once (in prokaryotic cyanobacteria), and all photosynthetic eukaryotes (including all plants and algae) have acquired this ability from them. In other words, all the oxygen that makes the atmosphere breathable for aerobic organisms originally comes from cyanobacteria or their later descendants.
Cyanobacteria remained principal primary producers throughout the Proterozoic Eon (2500–543 Ma), in part because the redox structure of the oceans favored photoautotrophs capable of nitrogen fixation
Nitrogen fixation is a chemical process by which molecular nitrogen (), with a strong triple covalent bond, in the air is converted into ammonia () or related nitrogenous compounds, typically in soil or aquatic systems but also in industry. Atmo ...
. Green algae joined blue-greens as major primary producers on Continental shelf, continental shelves near the end of the Proterozoic, but only with the Mesozoic (251–65 Ma) radiations of dinoflagellates, coccolithophorids, and diatoms did primary production in marine shelf waters take modern form. Cyanobacteria remain critical to marine ecosystems as primary producers in oceanic gyres, as agents of biological nitrogen fixation, and, in modified form, as the plastids of marine eukaryotic algae.
Origin of chloroplasts
Primary chloroplasts are cell organelles found in some eukaryote, eukaryotic lineages, where they are specialized in performing photosynthesis. They are considered to have evolved from endosymbiotic cyanobacteria. After some years of debate, it is now generally accepted that the three major groups of primary endosymbiotic eukaryotes (i.e. Viridiplantae, green plants, Rhodophytes, red algae and glaucophytes) form one large Monophyly, monophyletic group called Archaeplastida, which evolved after one unique endosymbiotic event. The Morphology (biology), morphological similarity between chloroplasts and cyanobacteria was first reported by German botanist Andreas Franz Wilhelm Schimper in the 19th century Chloroplasts are only found in plants and algae, thus paving the way for Russian biologist Konstantin Mereschkowski to suggest in 1905 the symbiogenic origin of the plastid. Lynn Margulis brought this hypothesis back to attention more than 60 years later but the idea did not become fully accepted until supplementary data started to accumulate. The cyanobacterial origin of plastids is now supported by various pieces of Phylogenetics, phylogenetic, Genomics, genomic, biochemical and structural evidence. The description of another independent and more recent primary endosymbiosis event between a cyanobacterium and a separate eukaryote lineage (the rhizarian ''Paulinella chromatophora'') also gives credibility to the endosymbiotic origin of the plastids. In addition to this primary endosymbiosis, many eukaryotic lineages have been subject to Secondary endosymbiosis, secondary or even tertiary endosymbiotic events, that is the "Matryoshka doll, Matryoshka-like" engulfment by a eukaryote of another plastid-bearing eukaryote. Chloroplasts have many similarities with cyanobacteria, including a circular chromosome, prokaryotic-type ribosomes, and similar proteins in the photosynthetic reaction center. The endosymbiotic theory suggests that photosynthetic bacteria were acquired (by endocytosis) by early Eukaryote, eukaryotic cells to form the first plant cells. Therefore, chloroplasts may be photosynthetic bacteria that adapted to life inside plant cells. Like mitochondrion, mitochondria, chloroplasts still possess their own DNA, separate from the nuclear DNA of their plant host cells and the genes in this chloroplast DNA resemble those in cyanobacteria. DNA in chloroplasts codes for redox proteins such as photosynthetic reaction centers. The CoRR hypothesis proposes this co-location is required for redox regulation.Marine origins
Cyanobacteria have fundamentally transformed the geochemistry of the planet. Multiple lines of geochemical evidence support the occurrence of intervals of profound global environmental change at the beginning and end of the Proterozoic (2,500–542 Mya). While it is widely accepted that the presence of molecular oxygen in the early fossil record was the result of cyanobacteria activity, little is known about how cyanobacteria evolution (e.g., habitat preference) may have contributed to changes in biogeochemical cycles through Earth history. Geochemical evidence has indicated that there was a first step-increase in the oxygenation of the Earth's surface, which is known as theGreat Oxidation Event
The Great Oxidation Event (GOE), also called the Great Oxygenation Event, the Oxygen Catastrophe, the Oxygen Revolution, the Oxygen Crisis, or the Oxygen Holocaust, was a time interval during the Paleoproterozoic era when the Earth's atmosphere ...
(GOE), in the early Paleoproterozoic (2,500–1,600 Mya). A second but much steeper increase in oxygen levels, known as the Neoproterozoic Oxygenation Event (NOE), occurred at around 800 to 500 Mya. Recent chromium isotope data point to low levels of atmospheric oxygen in the Earth's surface during the mid-Proterozoic, which is consistent with the late evolution of marine planktonic cyanobacteria during the Cryogenian; both types of evidence help explain the late emergence and diversification of animals.
Understanding the evolution of planktonic cyanobacteria is important because their origin fundamentally transformed the nitrogen cycle, nitrogen and carbon cycles towards the end of the Pre-Cambrian. It remains unclear, however, what evolutionary events led to the emergence of open-ocean planktonic forms within cyanobacteria and how these events relate to geochemical evidence during the Pre-Cambrian. So far, it seems that ocean geochemistry (e.g., euxinic conditions during the early- to mid-Proterozoic) and nutrient availability likely contributed to the apparent delay in diversification and widespread colonization of open ocean environments by planktonic cyanobacteria during the Neoproterozoic.
Genetics
Cyanobacteria are capable of natural genetic transformation (genetics), transformation. Natural genetic transformation is the genetic alteration of a cell resulting from the direct uptake and incorporation of exogenous DNA from its surroundings. For bacterial transformation to take place, the recipient bacteria must be in a state of natural competence, competence, which may occur in nature as a response to conditions such as starvation, high cell density or exposure to DNA damaging agents. In chromosomal transformation, homologous transforming DNA can be integrated into the recipient genome by homologous recombination, and this process appears to be an adaptation for DNA repair, repairing DNA damage.DNA repair
Cyanobacteria are challenged by environmental stresses and internally generated reactive oxygen species that cause DNA damage (naturally occurring), DNA damage. Cyanobacteria possess numerous ''Escherichia coli, E. coli''-like DNA repair genes. Several DNA repair genes are highly conserved in cyanobacteria, even in small genomes, suggesting that core DNA repair processes such as homologous recombination, recombinational repair, nucleotide excision repair and methyl-directed DNA mismatch repair are common among cyanobacteria.Classification
Phylogeny
Taxonomy
 Historically, bacteria were first classified as plants constituting the class Schizomycetes, which along with the Schizophyceae (blue-green algae/Cyanobacteria) formed the phylum Schizophyta, then in the phylum Monera in the kingdom Protista by Haeckel in 1866, comprising ''Protogens, Protamaeba, Vampyrella, Protomonae'', and ''Vibrio'', but not ''Nostoc'' and other cyanobacteria, which were classified with algae, later reclassified as the ''Prokaryotes'' by Édouard Chatton, Chatton.
The cyanobacteria were traditionally classified by morphology into five sections, referred to by the numerals I–V. The first three – Chroococcales, Pleurocapsales, and Oscillatoriales – are not supported by phylogenetic studies. The latter two – Nostocales and Stigonematales – are monophyletic, and make up the heterocystous cyanobacteria.
The members of Chroococales are unicellular and usually aggregate in colonies. The classic taxonomic criterion has been the cell morphology and the plane of cell division. In Pleurocapsales, the cells have the ability to form internal spores (baeocytes). The rest of the sections include filamentous species. In Oscillatoriales, the cells are uniseriately arranged and do not form specialized cells (akinetes and heterocysts). In Nostocales and Stigonematales, the cells have the ability to develop heterocysts in certain conditions. Stigonematales, unlike Nostocales, include species with truly branched trichomes.
Most taxa included in the phylum or division Cyanobacteria have not yet been validly published under ''The International Code of Nomenclature of Prokaryotes'' (ICNP) except:
*The classes Chroobacteria, Hormogoneae, and Gloeobacteria
*The orders Chroococcales, Gloeobacterales, Nostocales, Oscillatoriales, Pleurocapsales, and Stigonematales
*The families Prochloraceae and Prochlorotrichaceae
*The genera ''Halospirulina, Planktothricoides,
Historically, bacteria were first classified as plants constituting the class Schizomycetes, which along with the Schizophyceae (blue-green algae/Cyanobacteria) formed the phylum Schizophyta, then in the phylum Monera in the kingdom Protista by Haeckel in 1866, comprising ''Protogens, Protamaeba, Vampyrella, Protomonae'', and ''Vibrio'', but not ''Nostoc'' and other cyanobacteria, which were classified with algae, later reclassified as the ''Prokaryotes'' by Édouard Chatton, Chatton.
The cyanobacteria were traditionally classified by morphology into five sections, referred to by the numerals I–V. The first three – Chroococcales, Pleurocapsales, and Oscillatoriales – are not supported by phylogenetic studies. The latter two – Nostocales and Stigonematales – are monophyletic, and make up the heterocystous cyanobacteria.
The members of Chroococales are unicellular and usually aggregate in colonies. The classic taxonomic criterion has been the cell morphology and the plane of cell division. In Pleurocapsales, the cells have the ability to form internal spores (baeocytes). The rest of the sections include filamentous species. In Oscillatoriales, the cells are uniseriately arranged and do not form specialized cells (akinetes and heterocysts). In Nostocales and Stigonematales, the cells have the ability to develop heterocysts in certain conditions. Stigonematales, unlike Nostocales, include species with truly branched trichomes.
Most taxa included in the phylum or division Cyanobacteria have not yet been validly published under ''The International Code of Nomenclature of Prokaryotes'' (ICNP) except:
*The classes Chroobacteria, Hormogoneae, and Gloeobacteria
*The orders Chroococcales, Gloeobacterales, Nostocales, Oscillatoriales, Pleurocapsales, and Stigonematales
*The families Prochloraceae and Prochlorotrichaceae
*The genera ''Halospirulina, Planktothricoides, Prochlorococcus
''Prochlorococcus'' is a genus of very small (0.6 μm) marine cyanobacteria with an unusual pigmentation ( chlorophyll ''a2'' and ''b2''). These bacteria belong to the photosynthetic picoplankton and are probably the most abundant photosynth ...
, Prochloron'', and ''Prochlorothrix''
The remainder are validly published under the International Code of Nomenclature for algae, fungi, and plants.
Formerly, some bacteria, like ''Beggiatoa'', were thought to be colorless Cyanobacteria.
The currently accepted taxonomy is based on the List of Prokaryotic names with Standing in Nomenclature (LPSN) and National Center for Biotechnology Information (NCBI).
Class "Cyanobacteriia"
* Subclass Gloeobacteria
** Gloeobacterales Cavalier-Smith 2002
* Subclass Phycobacteria
** "Elainellales"
** "Eurycoccales"
** Gloeoemargaritales Moreira et al. 2016
** "Leptolyngbyales"
** "Neosynechococcales"
** "Phormidesmiales"
** Prochlorococcaceae Komárek & Strunecky 2020
** Pseudanabaenales Hoffmann, Komárek & Kastovsky 2005
** Thermostichales Komárek & Strunecký 2020
** "Thermosynechococcales"
** Nostocophycidae
*** Cyanobacteriales Rippka & Cohen-Bazire 1983 (Chamaesiphonales, Chroococcales, Chroococcidiopsidales, Nostocales, Oscillatoriales, Pleurocapsales, Spirulinales, Stigonematales)
** Synechococcophycidae
*** "Limnotrichales"
*** Prochlorotrichaceae Burger-Wiersma et al. 1989
*** Synechococcales Hoffmann, Komárek & Kastovsky 2005
Relation to humans
Biotechnology
 The unicellular cyanobacterium ''
The unicellular cyanobacterium ''Synechocystis
''Synechocystis'' is a genus of unicellular, freshwater cyanobacteria in the family Merismopediaceae. It includes a strain, ''Synechocystis'' sp. PCC 6803, which is a well studied model organism.
Like all cyanobacteria, ''Synechocystis'' ...
'' sp. PCC6803 was the third prokaryote and first photosynthetic organism whose genome was completely DNA sequencing, sequenced. It continues to be an important model organism. ''Cyanothece
''Cyanothece'' is a genus of unicellular, diazotrophic, oxygenic photosynthesizing cyanobacteria.
Modern organisms and cellular organization
In 1976, Jiří Komárek defined the prokaryotic cyanobacteria genus ''Cyanothece'' as distinct from ' ...
'' ATCC 51142 is an important diazotrophic model organism. The smallest genomes have been found in ''Prochlorococcus'' spp. (1.7 Mb) and the largest in ''Nostoc punctiforme'' (9 Mb). Those of ''Calothrix
''Calothrix'' is a genus of cyanobacteria
Cyanobacteria (), also known as Cyanophyta, are a phylum of gram-negative bacteria that obtain energy via photosynthesis. The name ''cyanobacteria'' refers to their color (), which similarly forms ...
'' spp. are estimated at 12–15 Mb, as large as yeast.
Recent research has suggested the potential application of cyanobacteria to the generation of renewable energy by directly converting sunlight into electricity. Internal photosynthetic pathways can be coupled to chemical mediators that transfer electrons to external electrodes. In the shorter term, efforts are underway to commercialize algae-based fuels such as diesel fuel, diesel, gasoline, and jet fuel. Cyanobacteria have been also engineered to produce ethanol and experiments have shown that when one or two CBB genes are being over expressed, the yield can be even higher.
Cyanobacteria may possess the ability to produce substances that could one day serve as anti-inflammatory agents and combat bacterial infections in humans. Cyanobacteria's photosynthetic output of sugar and oxygen has been demonstrated to have therapeutic value in rats with heart attacks. While cyanobacteria can naturally produce various secondary metabolites, they can serve as advantageous hosts for plant-derived metabolites production owing to biotechnological advances in systems biology and synthetic biology.
Spirulina's extracted blue color is used as a natural food coloring.
Researchers from several space agencies argue that cyanobacteria could be used for producing goods for human consumption in future crewed outposts on Mars, by transforming materials available on this planet.
Human nutrition
 Some cyanobacteria are sold as food, notably ''Arthrospira platensis (Spirulina (dietary supplement), Spirulina) and others'' (''Aphanizomenon flos-aquae'').
Some microalgae contain substances of high biological value, such as polyunsaturated fatty acids, amino acids, proteins, pigments, antioxidants, vitamins, and minerals. Edible blue-green algae reduce the production of pro-inflammatory cytokines by inhibiting NF-κB pathway in macrophages and splenocytes. Sulfate polysaccharides exhibit immunomodulatory, antitumor, antithrombotic, anticoagulant, anti-mutagenic, anti-inflammatory, antimicrobial, and even antiviral activity against HIV, herpes, and hepatitis.
Some cyanobacteria are sold as food, notably ''Arthrospira platensis (Spirulina (dietary supplement), Spirulina) and others'' (''Aphanizomenon flos-aquae'').
Some microalgae contain substances of high biological value, such as polyunsaturated fatty acids, amino acids, proteins, pigments, antioxidants, vitamins, and minerals. Edible blue-green algae reduce the production of pro-inflammatory cytokines by inhibiting NF-κB pathway in macrophages and splenocytes. Sulfate polysaccharides exhibit immunomodulatory, antitumor, antithrombotic, anticoagulant, anti-mutagenic, anti-inflammatory, antimicrobial, and even antiviral activity against HIV, herpes, and hepatitis.
Health risks
Some cyanobacteria can produce neurotoxins, cytotoxins, endotoxins, and hepatotoxins (e.g., themicrocystin
Microcystins—or cyanoginosins—are a class of toxins produced by certain freshwater cyanobacteria, commonly known as blue-green algae. Over 250 different microcystins have been discovered so far, of which microcystin-LR is the most common. C ...
-producing bacteria genus microcystis), which are collectively known as cyanotoxin
Cyanotoxins are toxins produced by cyanobacteria (also known as blue-green algae). Cyanobacteria are found almost everywhere, but particularly in lakes and in the ocean where, under high concentration of phosphorus conditions, they reproduce exp ...
s.
Specific toxins include anatoxin-a, guanitoxin, aplysiatoxin, cyanopeptolin, cylindrospermopsin
Cylindrospermopsin (abbreviated to CYN, or CYL) is a cyanotoxin produced by a variety of freshwater cyanobacteria. CYN is a polycyclic uracil derivative containing guanidino and sulfate groups. It is also zwitterionic, making it highly water ...
, domoic acid, nodularin R (from ''Nodularia''), neosaxitoxin, and saxitoxin
Saxitoxin (STX) is a potent neurotoxin and the best-known paralytic shellfish toxin (PST). Ingestion of saxitoxin by humans, usually by consumption of shellfish contaminated by toxic algal blooms, is responsible for the illness known as paralyt ...
. Cyanobacteria reproduce explosively under certain conditions. This results in algal blooms which can become Harmful algal blooms, harmful to other species and pose a danger to humans and animals if the cyanobacteria involved produce toxins. Several cases of human poisoning have been documented, but a lack of knowledge prevents an accurate assessment of the risks, and research by Linda Lawton, Fellowship of the Royal Society of Edinburgh, FRSE at Robert Gordon University, Aberdeen and collaborators has 30 years of examining the phenomenon and methods of improving water safety.
Recent studies suggest that significant exposure to high levels of cyanobacteria producing toxins such as beta-Methylamino-L-alanine, BMAA can cause amyotrophic lateral sclerosis (ALS). People living within half a mile of cyanobacterially contaminated lakes have had a 2.3 times greater risk of developing ALS than the rest of the population; people around New Hampshire's Lake Mascoma had an up to 25 times greater risk of ALS than the expected incidence. BMAA from desert crusts found throughout Qatar might have contributed to higher rates of ALS in Gulf War veterans.
Chemical control
Several chemicals can eliminate cyanobacterial blooms from smaller water-based systems such as swimming pools. They include calcium hypochlorite, Copper(II) sulfate, copper sulphate, cupricide, and simazine. The calcium hypochlorite amount needed varies depending on the cyanobacteria bloom, and treatment is needed periodically. According to the Department of Agriculture Australia, a rate of 12 g of 70% material in 1000 L of water is often effective to treat a bloom. Copper sulfate is also used commonly, but no longer recommended by the Australian Department of Agriculture, as it kills livestock, crustaceans, and fish. Cupricide is a chelated copper product that eliminates blooms with lower toxicity risks than copper sulfate. Dosage recommendations vary from 190 mL to 4.8 L per 1000 m2. Ferric alum treatments at the rate of 50 mg/L will reduce algae blooms. Simazine, which is also a herbicide, will continue to kill blooms for several days after an application. Simazine is marketed at different strengths (25, 50, and 90%), the recommended amount needed for one cubic meter of water per product is 25% product 8 mL; 50% product 4 mL; or 90% product 2.2 mL.Climate change
Climate change is likely to increase the frequency, intensity and duration of cyanobacterial blooms in many eutrophic lakes, reservoirs and estuaries. Bloom-forming cyanobacteria produce a variety of neurotoxins, hepatotoxins and dermatoxins, which can be fatal to birds and mammals (including waterfowl, cattle and dogs) and threaten the use of waters for recreation, drinking water production, agricultural irrigation and fisheries. Cyanotoxin, Toxic cyanobacteria have caused major water quality problems, for example in Lake Taihu (China), Lake Erie (USA), Lake Okeechobee (USA), Lake Victoria (Africa) and the Baltic Sea.Creative Commons Attribution 4.0 International License
Climate change favours cyanobacterial blooms both directly and indirectly. Many bloom-forming cyanobacteria can grow at relatively high temperatures. Increased thermal stratification of lakes and reservoirs enables buoyant cyanobacteria to float upwards and form dense surface blooms, which gives them better access to light and hence a selective advantage over nonbuoyant phytoplankton organisms. Protracted droughts during summer increase water residence times in reservoirs, rivers and estuaries, and these stagnant warm waters can provide ideal conditions for cyanobacterial bloom development. The capacity of the harmful cyanobacterial genus ''Microcystis'' to adapt to elevated CO2 levels was demonstrated in both laboratory and field experiments. ''Microcystis'' spp. take up CO2 and HCO3− and accumulate inorganic carbon in
carboxysome
Carboxysomes are bacterial microcompartments (BMCs) consisting of polyhedral protein shells filled with the enzymes ribulose-1,5-bisphosphate carboxylase/oxygenase (RuBisCO)—the predominant enzyme in carbon fixation and the rate limiting e ...
s, and strain competitiveness was found to depend on the concentration of inorganic carbon. As a result, climate change and increased CO2 levels are expected to affect the strain composition of cyanobacterial blooms.
Gallery
Oscillatoria
''Oscillatoria'' is a genus of filamentous cyanobacterium which is often found in freshwater environments, such as hot springs, and appears blue-green. Its name refers to the oscillating motion of its filaments as they slide against each other ...
'' and ''Gleocapsa'' – with oscillatory movement as filaments of ''Oscillatoria'' orient towards light
See also
* Archean Eon * Bacterial phyla, other major lineages of Bacteria * Biodiesel * Cyanobiont * Endosymbiotic theory * Geological history of oxygen * HypolithNotes
References
;AttributionFurther reading
* * * * * * * *External links
What are Cyanobacteria and What are its Types?
Webserver for Cyanobacteria Research
Diving an Antarctic Time Capsule Filled With Primordial Life
{{Authority control Cyanobacteria, Phototrophic bacteria Photosynthesis Gram-negative bacteria Environmental chemistry Bacteria phyla