
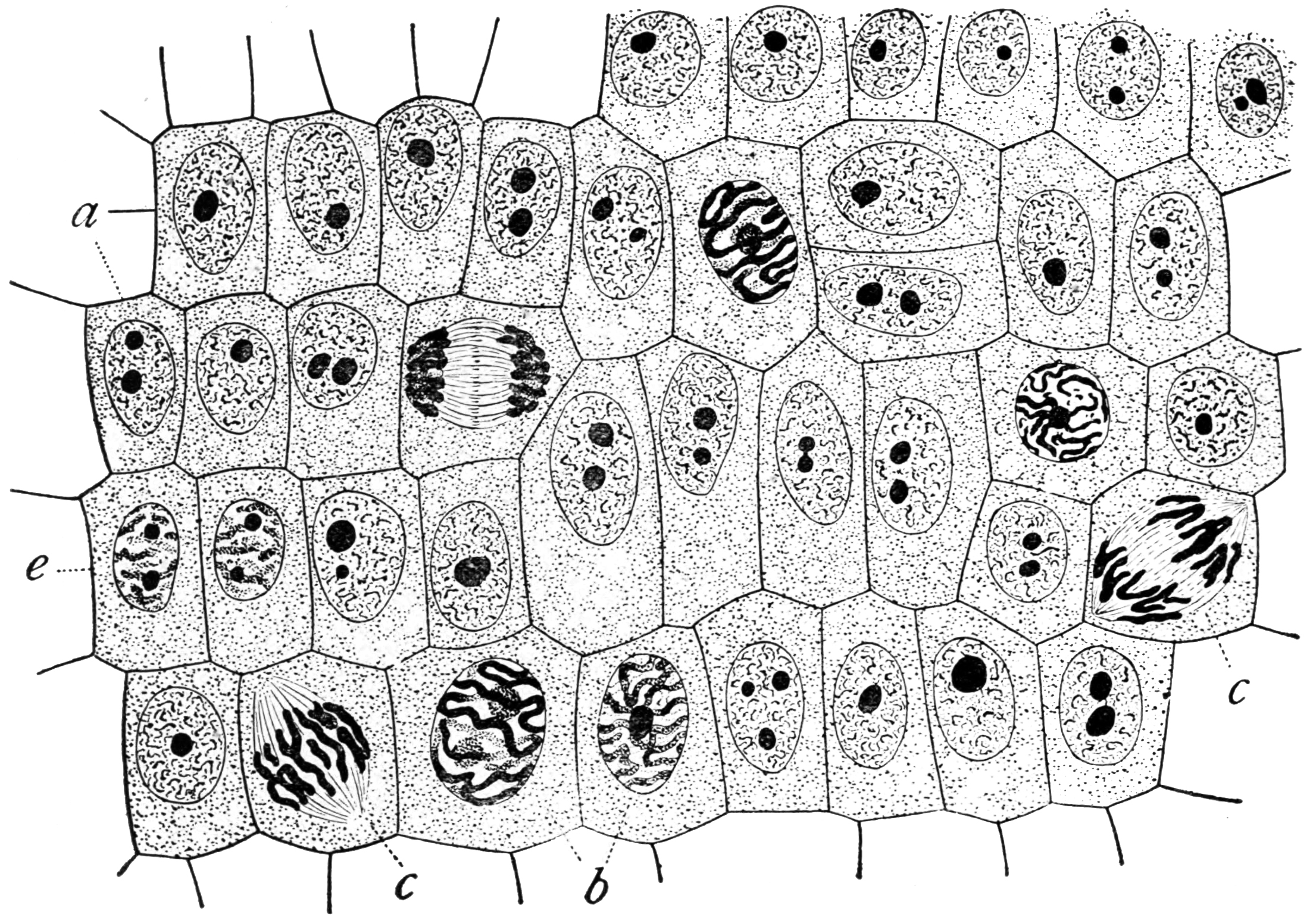
The cell cycle, or cell-division cycle, is the sequential series of events that take place in a
cell that causes it to divide into two daughter cells. These events include the growth of the cell, duplication of its DNA (
DNA replication
In molecular biology, DNA replication is the biological process of producing two identical replicas of DNA from one original DNA molecule. DNA replication occurs in all life, living organisms, acting as the most essential part of heredity, biolog ...
) and some of its
organelle
In cell biology, an organelle is a specialized subunit, usually within a cell (biology), cell, that has a specific function. The name ''organelle'' comes from the idea that these structures are parts of cells, as Organ (anatomy), organs are to th ...
s, and subsequently the partitioning of its cytoplasm, chromosomes and other components into two daughter cells in a process called
cell division
Cell division is the process by which a parent cell (biology), cell divides into two daughter cells. Cell division usually occurs as part of a larger cell cycle in which the cell grows and replicates its chromosome(s) before dividing. In eukar ...
.
In
eukaryotic cells (having a
cell nucleus
The cell nucleus (; : nuclei) is a membrane-bound organelle found in eukaryote, eukaryotic cell (biology), cells. Eukaryotic cells usually have a single nucleus, but a few cell types, such as mammalian red blood cells, have #Anucleated_cells, ...
) including
animal
Animals are multicellular, eukaryotic organisms in the Biology, biological Kingdom (biology), kingdom Animalia (). With few exceptions, animals heterotroph, consume organic material, Cellular respiration#Aerobic respiration, breathe oxygen, ...
,
plant
Plants are the eukaryotes that form the Kingdom (biology), kingdom Plantae; they are predominantly Photosynthesis, photosynthetic. This means that they obtain their energy from sunlight, using chloroplasts derived from endosymbiosis with c ...
,
fungal
A fungus (: fungi , , , or ; or funguses) is any member of the group of eukaryotic organisms that includes microorganisms such as yeasts and molds, as well as the more familiar mushrooms. These organisms are classified as one of the tradit ...
, and
protist
A protist ( ) or protoctist is any eukaryotic organism that is not an animal, land plant, or fungus. Protists do not form a natural group, or clade, but are a paraphyletic grouping of all descendants of the last eukaryotic common ancest ...
cells, the cell cycle is divided into two main stages:
interphase
Interphase is the active portion of the cell cycle that includes the G1, S, and G2 phases, where the cell grows, replicates its DNA, and prepares for mitosis, respectively. Interphase was formerly called the "resting phase," but the cell i ...
, and the
M phase
The cell cycle, or cell-division cycle, is the sequential series of events that take place in a cell that causes it to divide into two daughter cells. These events include the growth of the cell, duplication of its DNA (DNA replication) and ...
that includes mitosis and cytokinesis.
During interphase, the cell grows, accumulating nutrients needed for mitosis, and replicates its DNA and some of its organelles. During the M phase, the replicated
chromosomes
A chromosome is a package of DNA containing part or all of the genetic material of an organism. In most chromosomes, the very long thin DNA fibers are coated with nucleosome-forming packaging proteins; in eukaryotic cells, the most importa ...
, organelles, and cytoplasm separate into two new daughter cells. To ensure the proper replication of cellular components and division, there are control mechanisms known as
cell cycle checkpoint
Cell cycle checkpoints are control mechanisms in the eukaryotic cell cycle which ensure its proper progression. Each checkpoint serves as a potential termination point along the cell cycle, during which the conditions of the cell are assessed, wi ...
s after each of the key steps of the cycle that determine if the cell can progress to the next phase.
In cells without nuclei the
prokaryote
A prokaryote (; less commonly spelled procaryote) is a unicellular organism, single-celled organism whose cell (biology), cell lacks a cell nucleus, nucleus and other membrane-bound organelles. The word ''prokaryote'' comes from the Ancient Gree ...
s,
bacteria
Bacteria (; : bacterium) are ubiquitous, mostly free-living organisms often consisting of one Cell (biology), biological cell. They constitute a large domain (biology), domain of Prokaryote, prokaryotic microorganisms. Typically a few micr ...
and
archaea
Archaea ( ) is a Domain (biology), domain of organisms. Traditionally, Archaea only included its Prokaryote, prokaryotic members, but this has since been found to be paraphyletic, as eukaryotes are known to have evolved from archaea. Even thou ...
, the
cell cycle
The cell cycle, or cell-division cycle, is the sequential series of events that take place in a cell (biology), cell that causes it to divide into two daughter cells. These events include the growth of the cell, duplication of its DNA (DNA re ...
is divided into the B, C, and D periods. The B period extends from the end of cell division to the beginning of DNA replication. DNA replication occurs during the C period. The D period refers to the stage between the end of DNA replication and the splitting of the bacterial cell into two daughter cells.
In single-celled organisms, a single cell-division cycle is how the organism reproduces to ensure its survival. In multicellular organisms such as plants and animals, a series of cell-division cycles is how the organism develops from a single-celled
fertilized egg
A zygote (; , ) is a eukaryotic cell formed by a fertilization event between two gametes.
The zygote's genome is a combination of the DNA in each gamete, and contains all of the genetic information of a new individual organism.
The sexual ...
into a mature organism, and is also the process by which
hair
Hair is a protein filament that grows from follicles found in the dermis. Hair is one of the defining characteristics of mammals.
The human body, apart from areas of glabrous skin, is covered in follicles which produce thick terminal and ...
,
skin
Skin is the layer of usually soft, flexible outer tissue covering the body of a vertebrate animal, with three main functions: protection, regulation, and sensation.
Other animal coverings, such as the arthropod exoskeleton, have different ...
,
blood cell
A blood cell (also called a hematopoietic cell, hemocyte, or hematocyte) is a cell produced through hematopoiesis and found mainly in the blood. Major types of blood cells include red blood cells (erythrocytes), white blood cells (leukocytes), ...
s, and some
internal organs
In a multicellular organism, an organ is a collection of tissues joined in a structural unit to serve a common function. In the hierarchy of life, an organ lies between tissue and an organ system. Tissues are formed from same type cells to a ...
are
regenerated and
healed (with possible exception of
nerve
A nerve is an enclosed, cable-like bundle of nerve fibers (called axons). Nerves have historically been considered the basic units of the peripheral nervous system. A nerve provides a common pathway for the Electrochemistry, electrochemical nerv ...
s; see
nerve damage
Nerve injury is an injury to a nerve. There is no single classification system that can describe all the many variations of nerve injuries. In 1941, Herbert Seddon introduced a classification of nerve injuries based on three main types of nerve ...
). After cell division, each of the daughter cells begin the
interphase
Interphase is the active portion of the cell cycle that includes the G1, S, and G2 phases, where the cell grows, replicates its DNA, and prepares for mitosis, respectively. Interphase was formerly called the "resting phase," but the cell i ...
of a new cell cycle. Although the various stages of interphase are not usually morphologically distinguishable, each phase of the cell cycle has a distinct set of specialized biochemical processes that prepare the cell for initiation of the cell division.
Phases
The eukaryotic cell cycle consists of four distinct phases:
G1 phase,
S phase
S phase (Synthesis phase) is the phase of the cell cycle in which DNA is replicated, occurring between G1 phase and G2 phase. Since accurate duplication of the genome is critical to successful cell division, the processes that occur during S ...
(synthesis),
G2 phase (collectively known as
interphase
Interphase is the active portion of the cell cycle that includes the G1, S, and G2 phases, where the cell grows, replicates its DNA, and prepares for mitosis, respectively. Interphase was formerly called the "resting phase," but the cell i ...
) and
M phase
The cell cycle, or cell-division cycle, is the sequential series of events that take place in a cell that causes it to divide into two daughter cells. These events include the growth of the cell, duplication of its DNA (DNA replication) and ...
(mitosis and cytokinesis). M phase is itself composed of two tightly coupled processes: mitosis, in which the cell's nucleus divides, and
cytokinesis
Cytokinesis () is the part of the cell division process and part of mitosis during which the cytoplasm of a single eukaryotic cell divides into two daughter cells. Cytoplasmic division begins during or after the late stages of nuclear division ...
, in which the cell's
cytoplasm
The cytoplasm describes all the material within a eukaryotic or prokaryotic cell, enclosed by the cell membrane, including the organelles and excluding the nucleus in eukaryotic cells. The material inside the nucleus of a eukaryotic cell a ...
and cell membrane divides forming two daughter cells. Activation of each phase is dependent on the proper progression and completion of the previous one. Cells that have temporarily or reversibly stopped dividing are said to have entered a state of quiescence known as
G0 phase or ''resting phase''.
G0 phase (quiescence)


G
0 is a resting phase where the cell has left the cycle and has stopped dividing. The cell cycle starts with this phase. Non-proliferative (non-dividing) cells in multicellular
eukaryote
The eukaryotes ( ) constitute the Domain (biology), domain of Eukaryota or Eukarya, organisms whose Cell (biology), cells have a membrane-bound cell nucleus, nucleus. All animals, plants, Fungus, fungi, seaweeds, and many unicellular organisms ...
s generally enter the quiescent G
0 state from G
1 and may remain quiescent for long periods of time, possibly indefinitely (as is often the case for
neuron
A neuron (American English), neurone (British English), or nerve cell, is an membrane potential#Cell excitability, excitable cell (biology), cell that fires electric signals called action potentials across a neural network (biology), neural net ...
s). This is very common for cells that are fully
differentiated. Some cells enter the G
0 phase semi-permanently and are considered post-mitotic, e.g., some liver, kidney, and stomach cells. Many cells do not enter G
0 and continue to divide throughout an organism's life, e.g., epithelial cells.
The word "post-mitotic" is sometimes used to refer to both
quiescent and
senescent
Senescence () or biological aging is the gradual deterioration of functional characteristics in living organisms. Whole organism senescence involves an increase in death rates or a decrease in fecundity with increasing age, at least in the ...
cells. Cellular senescence occurs in response to DNA damage and external stress and usually constitutes an arrest in G
1. Cellular senescence may make a cell's progeny nonviable; it is often a biochemical alternative to the self-destruction of such a damaged cell by
apoptosis
Apoptosis (from ) is a form of programmed cell death that occurs in multicellular organisms and in some eukaryotic, single-celled microorganisms such as yeast. Biochemistry, Biochemical events lead to characteristic cell changes (Morphology (biol ...
.
Interphase
Interphase represents the phase between two successive M phases. Interphase is a series of changes that takes place in a newly formed cell and its nucleus before it becomes capable of division again. It is also called preparatory phase or intermitosis. Typically interphase lasts for at least 91% of the total time required for the cell cycle.
Interphase proceeds in three stages, G
1, S, and G
2, followed by the cycle of mitosis and cytokinesis. The cell's nuclear DNA contents are duplicated during S phase.
G1 phase (First growth phase or Post mitotic gap phase)

The first phase within interphase, from the end of the previous M phase until the beginning of DNA synthesis, is called
G1 (G indicating ''gap''). It is also called the growth phase. During this phase, the biosynthetic activities of the cell, which are considerably slowed down during M phase, resume at a high rate. The duration of G
1 is highly variable, even among different cells of the same species.
In this phase, the cell increases its supply of proteins, increases the number of organelles (such as mitochondria, ribosomes), and grows in size. In G
1 phase, a cell has three options.
*To continue cell cycle and enter
S phase
S phase (Synthesis phase) is the phase of the cell cycle in which DNA is replicated, occurring between G1 phase and G2 phase. Since accurate duplication of the genome is critical to successful cell division, the processes that occur during S ...
*Stop cell cycle and enter
G0 phase for undergoing
differentiation.
*Become arrested in G
1 phase hence it may enter G
0 phase or re-enter cell cycle.
The deciding point is called
check point
Check Point Software Technologies Ltd. is an
Israeli multinational provider of software and combined hardware and software products for IT security, including network security, endpoint security, cloud security, mobile security, data security ...
(
Restriction point
The restriction point (R), also known as the Start or G1/S checkpoint, is a cell cycle checkpoint in the G1 phase of the animal cell cycle at which the cell becomes "committed" to the cell cycle, and after which extracellular signals are no lon ...
). This
check point
Check Point Software Technologies Ltd. is an
Israeli multinational provider of software and combined hardware and software products for IT security, including network security, endpoint security, cloud security, mobile security, data security ...
is called the restriction point or START and is regulated by G
1/S cyclins, which cause transition from G
1 to S phase. Passage through the G
1 check point commits the cell to division.
S phase (DNA replication)
The ensuing
S phase
S phase (Synthesis phase) is the phase of the cell cycle in which DNA is replicated, occurring between G1 phase and G2 phase. Since accurate duplication of the genome is critical to successful cell division, the processes that occur during S ...
starts when
DNA synthesis
DNA synthesis is the natural or artificial creation of deoxyribonucleic acid (DNA) molecules. DNA is a macromolecule made up of nucleotide units, which are linked by covalent bonds and hydrogen bonds, in a repeating structure. DNA synthesis occu ...
commences; when it is complete, all of the
chromosome
A chromosome is a package of DNA containing part or all of the genetic material of an organism. In most chromosomes, the very long thin DNA fibers are coated with nucleosome-forming packaging proteins; in eukaryotic cells, the most import ...
s have been replicated, i.e., each chromosome consists of two sister
chromatid
A chromatid (Greek ''khrōmat-'' 'color' + ''-id'') is one half of a duplicated chromosome. Before replication, one chromosome is composed of one DNA molecule. In replication, the DNA molecule is copied, and the two molecules are known as chrom ...
s. Thus, during this phase, the amount of DNA in the cell has doubled, though the
ploidy
Ploidy () is the number of complete sets of chromosomes in a cell, and hence the number of possible alleles for autosomal and pseudoautosomal genes. Here ''sets of chromosomes'' refers to the number of maternal and paternal chromosome copies, ...
and number of chromosomes are unchanged. Rates of RNA
transcription and
protein synthesis
Protein biosynthesis, or protein synthesis, is a core biological process, occurring inside cells, balancing the loss of cellular proteins (via degradation or export) through the production of new proteins. Proteins perform a number of critica ...
are very low during this phase. An exception to this is
histone
In biology, histones are highly basic proteins abundant in lysine and arginine residues that are found in eukaryotic cell nuclei and in most Archaeal phyla. They act as spools around which DNA winds to create structural units called nucleosomes ...
production, most of which occurs during the S phase.
G2 phase (growth)
G
2 phase occurs after DNA replication and is a period of protein synthesis and rapid cell growth to prepare the cell for mitosis. During this phase microtubules begin to reorganize to form a spindle (preprophase). Before proceeding to
mitotic phase, cells must be checked at the G
2 checkpoint for any DNA damage within the chromosomes. The G
2 checkpoint is mainly regulated by the tumor protein
p53
p53, also known as tumor protein p53, cellular tumor antigen p53 (UniProt name), or transformation-related protein 53 (TRP53) is a regulatory transcription factor protein that is often mutated in human cancers. The p53 proteins (originally thou ...
. If the DNA is damaged, p53 will either repair the DNA or trigger the apoptosis of the cell. If p53 is dysfunctional or mutated, cells with damaged DNA may continue through the cell cycle, leading to the development of cancer.
Mitotic phase (chromosome separation)
The relatively brief ''M phase'' consists of nuclear division (
karyokinesis
Mitosis () is a part of the cell cycle in eukaryotic cells in which replicated chromosomes are separated into two new nuclei. Cell division by mitosis is an equational division which gives rise to genetically identical cells in which the ...
) and division of cytoplasm (
cytokinesis
Cytokinesis () is the part of the cell division process and part of mitosis during which the cytoplasm of a single eukaryotic cell divides into two daughter cells. Cytoplasmic division begins during or after the late stages of nuclear division ...
). M phase is complex and highly regulated. The sequence of events is divided into phases, corresponding to the completion of one set of activities and the start of the next. These phases are sequentially known as:
*
prophase
Prophase () is the first stage of cell division in both mitosis and meiosis. Beginning after interphase, DNA has already been replicated when the cell enters prophase. The main occurrences in prophase are the condensation of the chromatin retic ...
*
prometaphase
Prometaphase is the phase of mitosis following prophase and preceding metaphase in eukaryotic somatic cells. In prometaphase, the nuclear membrane breaks apart into numerous "membrane vesicles," and the chromosomes inside form protein structure ...
*
metaphase
Metaphase ( and ) is a stage of mitosis in the eukaryotic cell cycle in which chromosomes are at their second-most condensed and coiled stage (they are at their most condensed in anaphase). These chromosomes, carrying genetic information, alig ...
*
anaphase
Anaphase () is the stage of mitosis after the process of metaphase, when replicated chromosomes are split and the newly-copied chromosomes (daughter chromatids) are moved to opposite poles of the cell. Chromosomes also reach their overall maxim ...
*
telophase
Telophase () is the final stage in both meiosis and mitosis in a eukaryotic cell. During telophase, the effects of prophase and prometaphase (the nucleolus and nuclear membrane disintegrating) are reversed. As chromosomes reach the cell poles, ...
Mitosis is the process by which a
eukaryotic
The eukaryotes ( ) constitute the Domain (biology), domain of Eukaryota or Eukarya, organisms whose Cell (biology), cells have a membrane-bound cell nucleus, nucleus. All animals, plants, Fungus, fungi, seaweeds, and many unicellular organisms ...
cell separates the
chromosome
A chromosome is a package of DNA containing part or all of the genetic material of an organism. In most chromosomes, the very long thin DNA fibers are coated with nucleosome-forming packaging proteins; in eukaryotic cells, the most import ...
s in its
cell nucleus
The cell nucleus (; : nuclei) is a membrane-bound organelle found in eukaryote, eukaryotic cell (biology), cells. Eukaryotic cells usually have a single nucleus, but a few cell types, such as mammalian red blood cells, have #Anucleated_cells, ...
into two identical sets in two nuclei. During the process of mitosis the pairs of
chromosomes
A chromosome is a package of DNA containing part or all of the genetic material of an organism. In most chromosomes, the very long thin DNA fibers are coated with nucleosome-forming packaging proteins; in eukaryotic cells, the most importa ...
condense (
chromosome condensation
Chromosome condensation refers to the process by which dispersed interphase chromatin is transformed into a set of compact, rod-shaped structures during mitosis and meiosis (Figure 1).
The term "chromosome condensation" has long been used in biolo ...
) and attach to
microtubule
Microtubules are polymers of tubulin that form part of the cytoskeleton and provide structure and shape to eukaryotic cells. Microtubules can be as long as 50 micrometres, as wide as 23 to 27 nanometer, nm and have an inner diameter bet ...
s that pull the
sister chromatids
A sister chromatid refers to the identical copies ( chromatids) formed by the DNA replication of a chromosome, with both copies joined together by a common centromere. In other words, a sister chromatid may also be said to be 'one-half' of the du ...
to opposite sides of the cell.
Mitosis occurs exclusively in
eukaryotic
The eukaryotes ( ) constitute the Domain (biology), domain of Eukaryota or Eukarya, organisms whose Cell (biology), cells have a membrane-bound cell nucleus, nucleus. All animals, plants, Fungus, fungi, seaweeds, and many unicellular organisms ...
cells, but occurs in different ways in different species. For example, animal cells undergo an "open" mitosis, where the
nuclear envelope
The nuclear envelope, also known as the nuclear membrane, is made up of two lipid bilayer membranes that in eukaryotic cells surround the nucleus, which encloses the genetic material.
The nuclear envelope consists of two lipid bilayer membran ...
breaks down before the chromosomes separate, while
fungi
A fungus (: fungi , , , or ; or funguses) is any member of the group of eukaryotic organisms that includes microorganisms such as yeasts and mold (fungus), molds, as well as the more familiar mushrooms. These organisms are classified as one ...
such as ''
Aspergillus nidulans
''Aspergillus nidulans'' (also called '' Emericella nidulans'' when referring to its sexual form, or teleomorph) is one of many species of filamentous fungi in the phylum Ascomycota. It has been an important research organism for studying eukary ...
'' and ''
Saccharomyces cerevisiae
''Saccharomyces cerevisiae'' () (brewer's yeast or baker's yeast) is a species of yeast (single-celled fungal microorganisms). The species has been instrumental in winemaking, baking, and brewing since ancient times. It is believed to have be ...
'' (
yeast
Yeasts are eukaryotic, single-celled microorganisms classified as members of the fungus kingdom (biology), kingdom. The first yeast originated hundreds of millions of years ago, and at least 1,500 species are currently recognized. They are est ...
) undergo a "closed" mitosis, where chromosomes divide within an intact
cell nucleus
The cell nucleus (; : nuclei) is a membrane-bound organelle found in eukaryote, eukaryotic cell (biology), cells. Eukaryotic cells usually have a single nucleus, but a few cell types, such as mammalian red blood cells, have #Anucleated_cells, ...
.
Cytokinesis phase (separation of all cell components)

Mitosis is immediately followed by
cytokinesis
Cytokinesis () is the part of the cell division process and part of mitosis during which the cytoplasm of a single eukaryotic cell divides into two daughter cells. Cytoplasmic division begins during or after the late stages of nuclear division ...
, which divides the nuclei,
cytoplasm
The cytoplasm describes all the material within a eukaryotic or prokaryotic cell, enclosed by the cell membrane, including the organelles and excluding the nucleus in eukaryotic cells. The material inside the nucleus of a eukaryotic cell a ...
,
organelle
In cell biology, an organelle is a specialized subunit, usually within a cell (biology), cell, that has a specific function. The name ''organelle'' comes from the idea that these structures are parts of cells, as Organ (anatomy), organs are to th ...
s and
cell membrane
The cell membrane (also known as the plasma membrane or cytoplasmic membrane, and historically referred to as the plasmalemma) is a biological membrane that separates and protects the interior of a cell from the outside environment (the extr ...
into two cells containing roughly equal shares of these cellular components. Cytokinesis occurs differently in plant and animal cells. While the cell membrane forms a groove that gradually deepens to separate the cytoplasm in animal cells, a
cell plate
image:Phragmoplast.png, 300px, Phragmoplast and cell plate formation in a plant cell during cytokinesis. Left side: Phragmoplast forms and cell plate starts to assemble in the center of the cell. Towards the right: Phragmoplast enlarges in a donut- ...
is formed to separate it in plant cells. The position of the cell plate is determined by the position of a preprophase band of microtubules and
actin
Actin is a family of globular multi-functional proteins that form microfilaments in the cytoskeleton, and the thin filaments in muscle fibrils. It is found in essentially all eukaryotic cells, where it may be present at a concentration of ...
filaments. Mitosis and cytokinesis together define the
division of the parent cell into two daughter cells, genetically identical to each other and to their parent cell. This accounts for approximately 10% of the cell cycle.
Because cytokinesis usually occurs in conjunction with mitosis, "mitosis" is often used interchangeably with "M phase". However, there are many cells where mitosis and cytokinesis occur separately, forming single cells with multiple nuclei in a process called
endoreplication
Endoreduplication (also referred to as endoreplication or endocycling) is replication of the nuclear genome in the absence of mitosis, which leads to elevated nuclear gene content and polyploidy. Endoreduplication can be understood simply as a vari ...
. This occurs most notably among the
fungi
A fungus (: fungi , , , or ; or funguses) is any member of the group of eukaryotic organisms that includes microorganisms such as yeasts and mold (fungus), molds, as well as the more familiar mushrooms. These organisms are classified as one ...
and
slime mold
Slime mold or slime mould is an informal name given to a polyphyletic assemblage of unrelated eukaryotic organisms in the Stramenopiles, Rhizaria, Discoba, Amoebozoa and Holomycota clades. Most are near-microscopic; those in the Myxogastria ...
s, but is found in various groups. Even in animals, cytokinesis and mitosis may occur independently, for instance during certain stages of
fruit fly embryonic development.
Errors in mitosis can result in cell death through
apoptosis
Apoptosis (from ) is a form of programmed cell death that occurs in multicellular organisms and in some eukaryotic, single-celled microorganisms such as yeast. Biochemistry, Biochemical events lead to characteristic cell changes (Morphology (biol ...
or cause
mutation
In biology, a mutation is an alteration in the nucleic acid sequence of the genome of an organism, virus, or extrachromosomal DNA. Viral genomes contain either DNA or RNA. Mutations result from errors during DNA or viral replication, ...
s that may lead to
cancer
Cancer is a group of diseases involving Cell growth#Disorders, abnormal cell growth with the potential to Invasion (cancer), invade or Metastasis, spread to other parts of the body. These contrast with benign tumors, which do not spread. Po ...
.
Regulation of eukaryotic cell cycle
Regulation of the cell cycle involves processes crucial to the survival of a cell, including the detection and repair of genetic damage as well as the prevention of uncontrolled cell division. The molecular events that control the cell cycle are ordered and directional; that is, each process occurs in a sequential fashion and it is impossible to "reverse" the cycle.
Role of cyclins and CDKs
Two key classes of regulatory molecules,
cyclin
Cyclins are proteins that control the progression of a cell through the cell cycle by activating cyclin-dependent kinases (CDK).
Etymology
Cyclins were originally discovered by R. Timothy Hunt in 1982 while studying the cell cycle of sea urch ...
s and
cyclin-dependent kinase
Cyclin-dependent kinases (CDKs) are a predominant group of serine/threonine protein kinases involved in the regulation of the cell cycle and its progression, ensuring the integrity and functionality of cellular machinery. These regulatory enzym ...
s (CDKs), determine a cell's progress through the cell cycle.
Leland H. Hartwell
Leland Harrison "Lee" Hartwell (born October 30, 1939) is an American former president and director of the Fred Hutchinson Cancer Research Center in Seattle, Washington. He shared the 2001 Nobel Prize in Physiology or Medicine with Paul Nurse an ...
,
R. Timothy Hunt, and
Paul M. Nurse
Sir Paul Maxime Nurse (born 25 January 1949) is an English geneticist, former President of the Royal Society and Chief Executive and Director of the Francis Crick Institute. He was awarded the 2001 Nobel Prize in Physiology or Medicine, along w ...
won the 2001
Nobel Prize in Physiology or Medicine
The Nobel Prize in Physiology or Medicine () is awarded yearly by the Nobel Assembly at the Karolinska Institute for outstanding discoveries in physiology or medicine. The Nobel Prize is not a single prize, but five separate prizes that, acco ...
for their discovery of these central molecules. Many of the genes encoding cyclins and CDKs are
conserved among all eukaryotes, but in general, more complex organisms have more elaborate cell cycle control systems that incorporate more individual components. Many of the relevant genes were first identified by studying yeast, especially ''
Saccharomyces cerevisiae
''Saccharomyces cerevisiae'' () (brewer's yeast or baker's yeast) is a species of yeast (single-celled fungal microorganisms). The species has been instrumental in winemaking, baking, and brewing since ancient times. It is believed to have be ...
'';
genetic nomenclature in yeast dubs many of these genes ''cdc'' (for "cell division cycle") followed by an identifying number, e.g. ''
cdc25
Cdc25 is a dual-specificity phosphatase first isolated from the yeast ''Schizosaccharomyces pombe'' as a cell cycle defective mutant. As with other cell cycle proteins or genes such as Cdc2 and Cdc4, the "cdc" in its name refers to "cell divis ...
'' or ''
cdc20
The cell division cycle protein 20 homolog is an essential regulator of cell division that is encoded by the ''CDC20'' gene in humans. To the best of current knowledge its most important function is to activate the anaphase promoting complex (AP ...
''.
Cyclins form the regulatory subunits and CDKs the catalytic subunits of an activated
heterodimer
In biochemistry, a protein dimer is a macromolecular complex or multimer formed by two protein monomers, or single proteins, which are usually non-covalently bound. Many macromolecules, such as proteins or nucleic acids, form dimers. The word ...
; cyclins have no catalytic activity and CDKs are inactive in the absence of a partner cyclin. When activated by a bound cyclin, CDKs perform a common biochemical reaction called
phosphorylation
In biochemistry, phosphorylation is described as the "transfer of a phosphate group" from a donor to an acceptor. A common phosphorylating agent (phosphate donor) is ATP and a common family of acceptor are alcohols:
:
This equation can be writ ...
that activates or inactivates target proteins to orchestrate coordinated entry into the next phase of the cell cycle. Different cyclin-CDK combinations determine the downstream proteins targeted. CDKs are constitutively expressed in cells whereas cyclins are synthesised at specific stages of the cell cycle, in response to various molecular signals.
General mechanism of cyclin-CDK interaction
Upon receiving a pro-mitotic extracellular signal, G
1 cyclin-CDK
A cyclin-dependent kinase complex (CDKC, cyclin-CDK) is a protein complex formed by the association of an inactive catalytic subunit of a protein kinase, cyclin-dependent kinase (CDK), with a regulatory subunit, cyclin.Malumbres M, Barbacid M. Ma ...
complexes become active to prepare the cell for S phase, promoting the expression of
transcription factor
In molecular biology, a transcription factor (TF) (or sequence-specific DNA-binding factor) is a protein that controls the rate of transcription (genetics), transcription of genetics, genetic information from DNA to messenger RNA, by binding t ...
s that in turn promote the expression of S cyclins and of enzymes required for
DNA replication
In molecular biology, DNA replication is the biological process of producing two identical replicas of DNA from one original DNA molecule. DNA replication occurs in all life, living organisms, acting as the most essential part of heredity, biolog ...
. The G
1 cyclin-CDK complexes also promote the degradation of molecules that function as S phase inhibitors by targeting them for
ubiquitination
Ubiquitin is a small (8.6 kDa) regulatory protein found in most tissues of eukaryotic organisms, i.e., it is found ''ubiquitously''. It was discovered in 1975 by Gideon Goldstein and further characterized throughout the late 1970s and 19 ...
. Once a protein has been ubiquitinated, it is targeted for proteolytic degradation by the
proteasome
Proteasomes are essential protein complexes responsible for the degradation of proteins by proteolysis, a chemical reaction that breaks peptide bonds. Enzymes that help such reactions are called proteases. Proteasomes are found inside all e ...
. Results from a study of E2F transcriptional dynamics at the single-cell level argue that the role of G1 cyclin-CDK activities, in particular cyclin D-CDK4/6, is to tune the timing rather than the commitment of cell cycle entry.
Active S cyclin-CDK complexes phosphorylate proteins that make up the
pre-replication complex
A pre-replication complex (pre-RC) is a protein complex that forms at the origin of replication during the initiation step of DNA replication. Formation of the pre-RC is required for DNA replication to occur. Complete and faithful replication of ...
es assembled during G
1 phase on DNA
replication origins. The phosphorylation serves two purposes: to activate each already-assembled pre-replication complex, and to prevent new complexes from forming. This ensures that every portion of the cell's
genome
A genome is all the genetic information of an organism. It consists of nucleotide sequences of DNA (or RNA in RNA viruses). The nuclear genome includes protein-coding genes and non-coding genes, other functional regions of the genome such as ...
will be replicated once and only once. The reason for prevention of gaps in replication is fairly clear, because daughter cells that are missing all or part of crucial genes will die. However, for reasons related to
gene copy number effects, possession of extra copies of certain genes is also deleterious to the daughter cells.
Mitotic cyclin-CDK complexes, which are synthesized but inactivated during S and G
2 phases, promote the initiation of
mitosis
Mitosis () is a part of the cell cycle in eukaryote, eukaryotic cells in which replicated chromosomes are separated into two new Cell nucleus, nuclei. Cell division by mitosis is an equational division which gives rise to genetically identic ...
by stimulating downstream proteins involved in chromosome condensation and
mitotic spindle
In cell biology, the spindle apparatus is the cytoskeletal structure of eukaryotic cells that forms during cell division to separate sister chromatids between daughter cells. It is referred to as the mitotic spindle during mitosis, a process ...
assembly. A critical complex activated during this process is a
ubiquitin ligase
A ubiquitin ligase (also called an E3 ubiquitin ligase) is a protein that recruits an E2 ubiquitin-conjugating enzyme that has been loaded with ubiquitin, recognizes a protein substrate, and assists or directly catalyzes the transfer of ubiquitin ...
known as the
anaphase-promoting complex
Anaphase-promoting complex (also called the cyclosome or APC/C) is an E3 ubiquitin ligase that marks target cell cycle proteins for degradation by the 26S proteasome. The APC/C is a large complex of 11–13 subunit proteins, including a cullin ...
(APC), which promotes degradation of structural proteins associated with the chromosomal
kinetochore
A kinetochore (, ) is a flared oblique-shaped protein structure associated with duplicated chromatids in eukaryotic cells where the spindle fibers, which can be thought of as the ropes pulling chromosomes apart, attach during cell division to ...
. APC also targets the mitotic cyclins for degradation, ensuring that telophase and cytokinesis can proceed.
Specific action of cyclin-CDK complexes
Cyclin D
Cyclin D is a member of the cyclin protein family that is involved in regulating cell cycle progression. The synthesis of cyclin D is initiated during G1 and drives the G1/S transition, G1/S phase transition. Cyclin D protein is anywhere fro ...
is the first cyclin produced in the cells that enter the cell cycle, in response to extracellular signals (e.g.
growth factor
A growth factor is a naturally occurring substance capable of stimulating cell proliferation, wound healing, and occasionally cellular differentiation. Usually it is a secreted protein or a steroid hormone. Growth factors are important for ...
s). Cyclin D levels stay low in resting cells that are not proliferating. Additionally,
CDK4/6 and
CDK2
Cyclin-dependent kinase 2, also known as cell division protein kinase 2, or Cdk2, is an enzyme that in humans is encoded by the ''CDK2'' gene. The protein encoded by this gene is a member of the cyclin-dependent kinase family of serine/threonine ...
are also inactive because CDK4/6 are bound by
INK4 family members (e.g., p16), limiting kinase activity. Meanwhile, CDK2 complexes are inhibited by the CIP/KIP proteins such as p21 and p27, When it is time for a cell to enter the cell cycle, which is triggered by a mitogenic stimuli, levels of cyclin D increase. In response to this trigger, cyclin D binds to existing
CDK4
Cyclin-dependent kinase 4 (CDK4), also known as cell division protein kinase 4, is an enzyme that is encoded by the ''CDK4'' gene in humans. CDK4 is a member of the cyclin-dependent kinase family, a group of serine/threonine kinases which regula ...
/6, forming the active cyclin D-CDK4/6 complex. Cyclin D-CDK4/6 complexes in turn mono-phosphorylates the
retinoblastoma
Retinoblastoma (Rb) is a rare form of cancer that rapidly develops from the immature cells of a retina, the light-detecting tissue of the eye. It is the most common primary malignant intraocular cancer in children, and 80% of retinoblastoma cas ...
susceptibility protein (
Rb) to pRb. The un-phosphorylated Rb tumour suppressor functions in inducing cell cycle exit and maintaining G0 arrest (senescence).
In the last few decades, a model has been widely accepted whereby pRB proteins are inactivated by cyclin D-Cdk4/6-mediated phosphorylation. Rb has 14+ potential phosphorylation sites. Cyclin D-Cdk 4/6 progressively phosphorylates Rb to hyperphosphorylated state, which triggers dissociation of pRB–
E2F complexes, thereby inducing G1/S cell cycle gene expression and progression into S phase.
Scientific observations from a study have shown that Rb is present in three types of isoforms: (1) un-phosphorylated Rb in G0 state; (2) mono-phosphorylated Rb, also referred to as "hypo-phosphorylated' or 'partially' phosphorylated Rb in early G1 state; and (3) inactive hyper-phosphorylated Rb in late G1 state.
In early G1 cells, mono-phosphorylated Rb exists as 14 different isoforms, one of each has distinct
E2F binding affinity.
Rb has been found to associate with hundreds of different proteins and the idea that different mono-phosphorylated Rb isoforms have different protein partners was very appealing.
A later report confirmed that mono-phosphorylation controls Rb's association with other proteins and generates functional distinct forms of Rb.
All different mono-phosphorylated Rb isoforms inhibit E2F transcriptional program and are able to arrest cells in G1-phase. Different mono-phosphorylated forms of Rb have distinct transcriptional outputs that are extended beyond E2F regulation.
In general, the binding of pRb to E2F inhibits the E2F target gene expression of certain G1/S and S transition genes including
E-type cyclins. The partial phosphorylation of Rb de-represses the Rb-mediated suppression of E2F target gene expression, begins the expression of cyclin E. The molecular mechanism that causes the cell switched to cyclin E activation is currently not known, but as cyclin E levels rise, the active cyclin E-CDK2 complex is formed, bringing Rb to be inactivated by hyper-phosphorylation.
Hyperphosphorylated Rb is completely dissociated from E2F, enabling further expression of a wide range of E2F target genes are required for driving cells to proceed into S phase
It has been identified that cyclin D-Cdk4/6 binds to a C-terminal alpha-helix region of Rb that is only distinguishable to cyclin D rather than other cyclins,
cyclin E
Cyclin E is a member of the cyclin family.
Cyclin E binds to G1 phase Cdk2, which is required for the transition from G1 to S phase of the cell cycle that determines initiation of DNA duplication. The Cyclin E/CDK2 complex phosphorylates p27 ...
,
A and
B.
This observation based on the structural analysis of Rb phosphorylation supports that Rb is phosphorylated in a different level through multiple Cyclin-Cdk complexes. This also makes feasible the current model of a simultaneous switch-like inactivation of all mono-phosphorylated Rb isoforms through one type of Rb hyper-phosphorylation mechanism. In addition, mutational analysis of the cyclin D- Cdk 4/6 specific Rb C-terminal helix shows that disruptions of cyclin D-Cdk 4/6 binding to Rb prevents Rb phosphorylation, arrests cells in G1, and bolsters Rb's functions in tumor suppressor.
This cyclin-Cdk driven cell cycle transitional mechanism governs a cell committed to the cell cycle that allows cell proliferation. A cancerous cell growth often accompanies with deregulation of Cyclin D-Cdk 4/6 activity.
The hyperphosphorylated Rb dissociates from the E2F/DP1/Rb complex (which was bound to the
E2F responsive genes, effectively "blocking" them from transcription), activating E2F. Activation of E2F results in transcription of various genes like
cyclin E
Cyclin E is a member of the cyclin family.
Cyclin E binds to G1 phase Cdk2, which is required for the transition from G1 to S phase of the cell cycle that determines initiation of DNA duplication. The Cyclin E/CDK2 complex phosphorylates p27 ...
,
cyclin A
Cyclin A is a member of the cyclin family, a group of proteins that function in regulating progression through the cell cycle. The stages that a cell passes through that culminate in its division and replication are collectively known as the cel ...
,
DNA polymerase
A DNA polymerase is a member of a family of enzymes that catalyze the synthesis of DNA molecules from nucleoside triphosphates, the molecular precursors of DNA. These enzymes are essential for DNA replication and usually work in groups to create t ...
,
thymidine kinase
Thymidine kinase is an enzyme, a phosphotransferase (a kinase): 2'-deoxythymidine kinase, ATP-thymidine 5'-phosphotransferase, EC 2.7.1.21. It can be found in most living cells. It is present in two forms in mammalian cells, TK1 and TK2. Cert ...
, etc. Cyclin E thus produced binds to
CDK2
Cyclin-dependent kinase 2, also known as cell division protein kinase 2, or Cdk2, is an enzyme that in humans is encoded by the ''CDK2'' gene. The protein encoded by this gene is a member of the cyclin-dependent kinase family of serine/threonine ...
, forming the cyclin E-CDK2 complex, which pushes the cell from G
1 to S phase (G
1/S, which initiates the G
2/M transition).
Cyclin B
Cyclin B is a member of the cyclin family. Cyclin B is a mitotic cyclin. The amount of cyclin B (which binds to Cdk1) and the activity of the cyclin B-Cdk complex rise through the cell cycle until mitosis, where they fall abruptly due to degr ...
-cdk1 complex activation causes breakdown of
nuclear envelope
The nuclear envelope, also known as the nuclear membrane, is made up of two lipid bilayer membranes that in eukaryotic cells surround the nucleus, which encloses the genetic material.
The nuclear envelope consists of two lipid bilayer membran ...
and initiation of
prophase
Prophase () is the first stage of cell division in both mitosis and meiosis. Beginning after interphase, DNA has already been replicated when the cell enters prophase. The main occurrences in prophase are the condensation of the chromatin retic ...
, and subsequently, its deactivation causes the cell to exit mitosis.
A quantitative study of E2F transcriptional dynamics at the single-cell level by using engineered fluorescent reporter cells provided a quantitative framework for understanding the control logic of cell cycle entry, challenging the canonical textbook model. Genes that regulate the amplitude of E2F accumulation, such as Myc, determine the commitment in cell cycle and S phase entry. G1 cyclin-CDK activities are not the driver of cell cycle entry. Instead, they primarily tune the timing of E2F increase, thereby modulating the pace of cell cycle progression.
Inhibitors
Endogenous
Two families of genes, the ''cip/kip'' (''CDK interacting protein/Kinase inhibitory protein'') family and the INK4a/ARF (''In''hibitor of ''K''inase 4/''A''lternative ''R''eading ''F''rame) family, prevent the progression of the cell cycle. Because these genes are instrumental in prevention of
tumor
A neoplasm () is a type of abnormal and excessive growth of tissue. The process that occurs to form or produce a neoplasm is called neoplasia. The growth of a neoplasm is uncoordinated with that of the normal surrounding tissue, and persists ...
formation, they are known as
tumor suppressor
A tumor suppressor gene (TSG), or anti-oncogene, is a gene that regulates a cell (biology), cell during cell division and replication. If the cell grows uncontrollably, it will result in cancer. When a tumor suppressor gene is mutated, it results ...
s.
The ''cip/kip'' family includes the genes
p21
p21Cip1 (alternatively p21Waf1), also known as cyclin-dependent kinase inhibitor 1 or CDK-interacting protein 1, is a cyclin-dependent kinase inhibitor (CKI) that is capable of inhibiting all cyclin/ CDK complexes, though is primarily associ ...
,
p27 and
p57. They halt the cell cycle in G
1 phase by binding to and inactivating cyclin-CDK complexes. p21 is activated by
p53
p53, also known as tumor protein p53, cellular tumor antigen p53 (UniProt name), or transformation-related protein 53 (TRP53) is a regulatory transcription factor protein that is often mutated in human cancers. The p53 proteins (originally thou ...
(which, in turn, is triggered by DNA damage e.g. due to radiation). p27 is activated by Transforming Growth Factor β (
TGF β), a growth inhibitor.
The INK4a/ARF family includes
p16INK4a, which binds to CDK4 and arrests the cell cycle in G
1 phase, and
p14ARF which prevents p53 degradation.
Synthetic
Synthetic inhibitors of
Cdc25
Cdc25 is a dual-specificity phosphatase first isolated from the yeast ''Schizosaccharomyces pombe'' as a cell cycle defective mutant. As with other cell cycle proteins or genes such as Cdc2 and Cdc4, the "cdc" in its name refers to "cell divis ...
could also be useful for the arrest of cell cycle and therefore be useful as antineoplastic and anticancer agents.
Many human cancers possess the hyper-activated Cdk 4/6 activities. Given the observations of cyclin D-Cdk 4/6 functions, inhibition of Cdk 4/6 should result in preventing a malignant tumor from proliferating. Consequently, scientists have tried to invent the synthetic Cdk4/6 inhibitor as Cdk4/6 has been characterized to be a therapeutic target for anti-tumor effectiveness. Three Cdk4/6 inhibitors –
palbociclib
Palbociclib, sold under the brand name Ibrance among others, is a medication developed by Pfizer for the treatment of HR-positive and HER2-negative breast cancer. It is a selective inhibitor of the cyclin-dependent kinases CDK4 and CDK6. Palb ...
,
ribociclib
Ribociclib, sold under the brand name Kisqali, is a medication used for the treatment of certain kinds of breast cancer. Ribociclib is a kinase inhibitor. It was developed by Novartis and Astex Pharmaceuticals.
The most common side effects inc ...
, and
abemaciclib – currently received FDA approval for clinical use to treat advanced-stage or
metastatic
Metastasis is a pathogenic agent's spreading from an initial or primary site to a different or secondary site within the host's body; the term is typically used when referring to metastasis by a cancerous tumor. The newly pathological sites, ...
,
hormone-receptor-positive (HR-positive, HR+),
HER2-negative (HER2-) breast cancer.
For example, palbociclib is an orally active CDK4/6 inhibitor which has demonstrated improved outcomes for ER-positive/HER2-negative advanced breast cancer. The main side effect is
neutropenia
Neutropenia is an abnormally low concentration of neutrophils (a type of white blood cell) in the blood. Neutrophils make up the majority of circulating white blood cells and serve as the primary defense against infections by destroying bacteria ...
which can be managed by dose reduction.
Cdk4/6 targeted therapy will only treat cancer types where Rb is expressed. Cancer cells with loss of Rb have primary resistance to Cdk4/6 inhibitors.
Transcriptional regulatory network
Current evidence suggests that a semi-autonomous transcriptional network acts in concert with the CDK-cyclin machinery to regulate the cell cycle. Several gene expression studies in ''
Saccharomyces cerevisiae
''Saccharomyces cerevisiae'' () (brewer's yeast or baker's yeast) is a species of yeast (single-celled fungal microorganisms). The species has been instrumental in winemaking, baking, and brewing since ancient times. It is believed to have be ...
'' have identified 800–1200 genes that change expression over the course of the cell cycle.
They are transcribed at high levels at specific points in the cell cycle, and remain at lower levels throughout the rest of the cycle. While the set of identified genes differs between studies due to the computational methods and criteria used to identify them, each study indicates that a large portion of yeast genes are temporally regulated.
Many periodically expressed genes are driven by
transcription factor
In molecular biology, a transcription factor (TF) (or sequence-specific DNA-binding factor) is a protein that controls the rate of transcription (genetics), transcription of genetics, genetic information from DNA to messenger RNA, by binding t ...
s that are also periodically expressed. One screen of single-gene knockouts identified 48 transcription factors (about 20% of all non-essential transcription factors) that show cell cycle progression defects.
Genome-wide studies using high throughput technologies have identified the transcription factors that bind to the promoters of yeast genes, and correlating these findings with temporal expression patterns have allowed the identification of transcription factors that drive phase-specific gene expression.
The expression profiles of these transcription factors are driven by the transcription factors that peak in the prior phase, and computational models have shown that a CDK-autonomous network of these transcription factors is sufficient to produce steady-state oscillations in gene expression).
Experimental evidence also suggests that gene expression can oscillate with the period seen in dividing wild-type cells independently of the CDK machinery. Orlando ''et al.'' used
microarray
A microarray is a multiplex (assay), multiplex lab-on-a-chip. Its purpose is to simultaneously detect the expression of thousands of biological interactions. It is a two-dimensional array on a Substrate (materials science), solid substrate—usu ...
s to measure the expression of a set of 1,271 genes that they identified as periodic in both wild type cells and cells lacking all S-phase and mitotic cyclins (''clb1,2,3,4,5,6''). Of the 1,271 genes assayed, 882 continued to be expressed in the cyclin-deficient cells at the same time as in the wild type cells, despite the fact that the cyclin-deficient cells arrest at the border between
G1 and
S phase
S phase (Synthesis phase) is the phase of the cell cycle in which DNA is replicated, occurring between G1 phase and G2 phase. Since accurate duplication of the genome is critical to successful cell division, the processes that occur during S ...
. However, 833 of the genes assayed changed behavior between the wild type and mutant cells, indicating that these genes are likely directly or indirectly regulated by the CDK-cyclin machinery. Some genes that continued to be expressed on time in the mutant cells were also expressed at different levels in the mutant and wild type cells. These findings suggest that while the transcriptional network may oscillate independently of the CDK-cyclin oscillator, they are coupled in a manner that requires both to ensure the proper timing of cell cycle events.
Other work indicates that
phosphorylation
In biochemistry, phosphorylation is described as the "transfer of a phosphate group" from a donor to an acceptor. A common phosphorylating agent (phosphate donor) is ATP and a common family of acceptor are alcohols:
:
This equation can be writ ...
, a post-translational modification, of cell cycle transcription factors by
Cdk1
Cyclin-dependent kinase 1 also known as CDK1 or cell division cycle protein 2 homolog is a highly conserved protein that functions as a serine/threonine protein kinase, and is a key player in cell cycle regulation. It has been highly studied in ...
may alter the localization or activity of the transcription factors in order to tightly control timing of target genes.
While oscillatory transcription plays a key role in the progression of the yeast cell cycle, the CDK-cyclin machinery operates independently in the early embryonic cell cycle. Before the
midblastula transition,
zygotic
A zygote (; , ) is a eukaryotic cell formed by a fertilization event between two gametes.
The zygote's genome is a combination of the DNA in each gamete, and contains all of the genetic information of a new individual organism.
The sexual ...
transcription does not occur and all needed proteins, such as the B-type cyclins, are translated from maternally loaded
mRNA
In molecular biology, messenger ribonucleic acid (mRNA) is a single-stranded molecule of RNA that corresponds to the genetic sequence of a gene, and is read by a ribosome in the process of Protein biosynthesis, synthesizing a protein.
mRNA is ...
.
DNA replication and DNA replication origin activity
Analyses of synchronized cultures of ''Saccharomyces cerevisiae'' under conditions that prevent DNA replication initiation without delaying cell cycle progression showed that origin licensing decreases the expression of genes with origins near their 3' ends, revealing that downstream origins can regulate the expression of upstream genes. This confirms previous predictions from mathematical modeling of a global causal coordination between DNA replication origin activity and mRNA expression, and shows that mathematical modeling of DNA microarray data can be used to correctly predict previously unknown biological modes of regulation.
Checkpoints
Cell cycle checkpoint
Cell cycle checkpoints are control mechanisms in the eukaryotic cell cycle which ensure its proper progression. Each checkpoint serves as a potential termination point along the cell cycle, during which the conditions of the cell are assessed, wi ...
s are used by the cell to monitor and regulate the progress of the cell cycle. Checkpoints prevent cell cycle progression at specific points, allowing verification of necessary phase processes and repair of
DNA damage
DNA repair is a collection of processes by which a cell identifies and corrects damage to the DNA molecules that encode its genome. A weakened capacity for DNA repair is a risk factor for the development of cancer. DNA is constantly modified ...
. The cell cannot proceed to the next phase until checkpoint requirements have been met. Checkpoints typically consist of a network of regulatory proteins that monitor and dictate the progression of the cell through the different stages of the cell cycle.

It is estimated that in normal human cells about 1% of
single-strand DNA damages are converted to about 50 endogenous DNA double-strand breaks per cell per cell cycle.
Although such double-strand breaks are usually
repaired with high fidelity, errors in their repair are considered to contribute significantly to the rate of cancer in humans.
[
There are several checkpoints to ensure that damaged or incomplete DNA is not passed on to daughter cells. Three main checkpoints exist: the G1/S checkpoint, the G2/M checkpoint and the metaphase (mitotic) checkpoint. Another checkpoint is the Go checkpoint, in which the cells are checked for maturity. If the cells fail to pass this checkpoint by not being ready yet, they will be discarded from dividing.
G1/S transition is a rate-limiting step in the cell cycle and is also known as ]restriction point
The restriction point (R), also known as the Start or G1/S checkpoint, is a cell cycle checkpoint in the G1 phase of the animal cell cycle at which the cell becomes "committed" to the cell cycle, and after which extracellular signals are no lon ...
.p53
p53, also known as tumor protein p53, cellular tumor antigen p53 (UniProt name), or transformation-related protein 53 (TRP53) is a regulatory transcription factor protein that is often mutated in human cancers. The p53 proteins (originally thou ...
plays an important role in triggering the control mechanisms at both G1/S and G2/M checkpoints. In addition to p53, checkpoint regulators are being heavily researched for their roles in cancer growth and proliferation.
Fluorescence imaging of the cell cycle
Pioneering work by Atsushi Miyawaki and coworkers developed the fluorescent ubiquitination-based cell cycle indicator
FUCCI
, which enables fluorescence
Fluorescence is one of two kinds of photoluminescence, the emission of light by a substance that has absorbed light or other electromagnetic radiation. When exposed to ultraviolet radiation, many substances will glow (fluoresce) with colore ...
imaging of the cell cycle. Originally, a green fluorescent protein
The green fluorescent protein (GFP) is a protein that exhibits green fluorescence when exposed to light in the blue to ultraviolet range. The label ''GFP'' traditionally refers to the protein first isolated from the jellyfish ''Aequorea victo ...
, mAG, was fused to hGem(1/110) and an orange fluorescent protein Fluorescent proteins include:
* Green fluorescent protein
The green fluorescent protein (GFP) is a protein that exhibits green fluorescence when exposed to light in the blue to ultraviolet range. The label ''GFP'' traditionally refers to the ...
(mKO2) was fused to hCdt1(30/120). Note, these fusions are fragments that contain a nuclear localization signal
A nuclear localization signal ''or'' sequence (NLS) is an amino acid sequence that 'tags' a protein for import into the cell nucleus by nuclear transport. Typically, this signal consists of one or more short sequences of positively charged lysin ...
and ubiquitination
Ubiquitin is a small (8.6 kDa) regulatory protein found in most tissues of eukaryotic organisms, i.e., it is found ''ubiquitously''. It was discovered in 1975 by Gideon Goldstein and further characterized throughout the late 1970s and 19 ...
sites for degradation, but are not functional proteins. The green fluorescent protein
The green fluorescent protein (GFP) is a protein that exhibits green fluorescence when exposed to light in the blue to ultraviolet range. The label ''GFP'' traditionally refers to the protein first isolated from the jellyfish ''Aequorea victo ...
is made during the S, G2, or M phase and degraded during the G0 or G1 phase, while the orange fluorescent protein Fluorescent proteins include:
* Green fluorescent protein
The green fluorescent protein (GFP) is a protein that exhibits green fluorescence when exposed to light in the blue to ultraviolet range. The label ''GFP'' traditionally refers to the ...
is made during the G0 or G1 phase and destroyed during the S, G2, or M phase. A far-red and near-infrared FUCCI was developed using a cyanobacteria
Cyanobacteria ( ) are a group of autotrophic gram-negative bacteria that can obtain biological energy via oxygenic photosynthesis. The name "cyanobacteria" () refers to their bluish green (cyan) color, which forms the basis of cyanobacteri ...
-derived fluorescent protein Fluorescent proteins include:
* Green fluorescent protein
The green fluorescent protein (GFP) is a protein that exhibits green fluorescence when exposed to light in the blue to ultraviolet range. The label ''GFP'' traditionally refers to the ...
( smURFP) and a bacteriophytochrome-derived fluorescent protein Fluorescent proteins include:
* Green fluorescent protein
The green fluorescent protein (GFP) is a protein that exhibits green fluorescence when exposed to light in the blue to ultraviolet range. The label ''GFP'' traditionally refers to the ...
movie found at this link
.
Several modifications have been made to the original FUCCI system to improve its usability in several in vitro systems and model organisms. These advancements have increased the sensitivity and accuracy of cell cycle phase detection, enabling more precise assessments of cellular proliferation
Role in tumor formation
A disregulation of the cell cycle components may lead to tumor
A neoplasm () is a type of abnormal and excessive growth of tissue. The process that occurs to form or produce a neoplasm is called neoplasia. The growth of a neoplasm is uncoordinated with that of the normal surrounding tissue, and persists ...
formation. As mentioned above, when some genes like the cell cycle inhibitors, RB, p53
p53, also known as tumor protein p53, cellular tumor antigen p53 (UniProt name), or transformation-related protein 53 (TRP53) is a regulatory transcription factor protein that is often mutated in human cancers. The p53 proteins (originally thou ...
etc. mutate, they may cause the cell to multiply uncontrollably, forming a tumor. Although the duration of cell cycle in tumor cells is equal to or longer than that of normal cell cycle, the proportion of cells that are in active cell division (versus quiescent cells in G0 phase) in tumors is much higher than that in normal tissue.drugs
A drug is any chemical substance other than a nutrient or an essential dietary ingredient, which, when administered to a living organism, produces a biological effect. Consumption of drugs can be via inhalation, injection, smoking, ingestio ...
or radiation
In physics, radiation is the emission or transmission of energy in the form of waves or particles through space or a material medium. This includes:
* ''electromagnetic radiation'' consisting of photons, such as radio waves, microwaves, infr ...
. This fact is made use of in cancer treatment; by a process known as debulking
Debulking is the reduction of as much of the bulk (volume) of a tumour without the intention of a complete eradication. It is usually achieved by surgical removal. When performed for curative intent, it is a different procedure, which is called sur ...
, a significant mass of the tumor is removed which pushes a significant number of the remaining tumor cells from G0 to G1 phase (due to increased availability of nutrients, oxygen, growth factors etc.). Radiation or chemotherapy following the debulking procedure kills these cells which have newly entered the cell cycle.Homologous recombination
Homologous recombination is a type of genetic recombination in which genetic information is exchanged between two similar or identical molecules of double-stranded or single-stranded nucleic acids (usually DNA as in Cell (biology), cellular organi ...
(HR) is an accurate process for repairing DNA double-strand breaks. HR is nearly absent in G1 phase
The G1 phase, gap 1 phase, or growth 1 phase, is the first of four phases of the cell cycle that takes place in eukaryotic cell division. In this part of interphase, the cell synthesizes Messenger RNA, mRNA and proteins in preparation for subsequ ...
, is most active in S phase
S phase (Synthesis phase) is the phase of the cell cycle in which DNA is replicated, occurring between G1 phase and G2 phase. Since accurate duplication of the genome is critical to successful cell division, the processes that occur during S ...
, and declines in G2/M.Non-homologous end joining
Non-homologous end joining (NHEJ) is a pathway that repairs double-strand breaks in DNA. It is called "non-homologous" because the break ends are directly ligated without the need for a homologous template, in contrast to homology directed repair ...
, a less accurate and more mutagenic process for repairing double strand breaks, is active throughout the cell cycle.
Cell cycle evolution
Evolution of the genome
The cell cycle must duplicate all cellular constituents and equally partition them into two daughter cells. Many constituents, such as proteins and ribosome
Ribosomes () are molecular machine, macromolecular machines, found within all cell (biology), cells, that perform Translation (biology), biological protein synthesis (messenger RNA translation). Ribosomes link amino acids together in the order s ...
s, are produced continuously throughout the cell cycle (except during M-phase
Mitosis () is a part of the cell cycle in eukaryotic cells in which replicated chromosomes are separated into two new nuclei. Cell division by mitosis is an equational division which gives rise to genetically identical cells in which the ...
). However, the chromosomes and other associated elements like MTOCs, are duplicated just once during the cell cycle. A central component of the cell cycle is its ability to coordinate the continuous and periodic duplications of different cellular elements, which evolved with the formation of the genome.
The pre-cellular environment contained functional and self-replicating RNA
Ribonucleic acid (RNA) is a polymeric molecule that is essential for most biological functions, either by performing the function itself (non-coding RNA) or by forming a template for the production of proteins (messenger RNA). RNA and deoxyrib ...
s.DNA
Deoxyribonucleic acid (; DNA) is a polymer composed of two polynucleotide chains that coil around each other to form a double helix. The polymer carries genetic instructions for the development, functioning, growth and reproduction of al ...
, which is a more stable molecule, allowed for larger genomes. The transition from self-catalysis enzyme synthesis to genome-directed enzyme synthesis was a critical step in cell evolution, and had lasting implications on the cell cycle, which must regulate functional synthesis and genomic duplication in very different ways.
Cyclin-dependent kinase and cyclin evolution
Cell-cycle progression is controlled by the oscillating concentrations of different cyclin
Cyclins are proteins that control the progression of a cell through the cell cycle by activating cyclin-dependent kinases (CDK).
Etymology
Cyclins were originally discovered by R. Timothy Hunt in 1982 while studying the cell cycle of sea urch ...
s and the resulting molecular interactions from the various cyclin-dependent kinase
Cyclin-dependent kinases (CDKs) are a predominant group of serine/threonine protein kinases involved in the regulation of the cell cycle and its progression, ensuring the integrity and functionality of cellular machinery. These regulatory enzym ...
s (CDKs). In yeast, just one CDK (Cdc28 in '' S. cerevisiae'' and Cdc2 in ''S. pombe
''Schizosaccharomyces pombe'', also called "fission yeast", is a species of yeast used in traditional brewing and as a model organism in molecular and cell biology. It is a unicellular eukaryote, whose cells are rod-shaped. Cells typically measu ...
'') controls the cell cycle. However, in animals, whole families of CDKs have evolved. Cdk1 controls entry to mitosis and Cdk2, Cdk4, and Cdk6 regulate entry into S phase. Despite the evolution of the CDK family in animals, these proteins have related or redundant functions. For example, ''cdk2 cdk4 cdk6'' triple knockout mice cells can still progress through the basic cell cycle.Arabidopsis thaliana
''Arabidopsis thaliana'', the thale cress, mouse-ear cress or arabidopsis, is a small plant from the mustard family (Brassicaceae), native to Eurasia and Africa. Commonly found along the shoulders of roads and in disturbed land, it is generally ...
'' has a Cdk1 homolog called CDKA;1, however ''cdka;1'' ''A. thaliana'' mutants are still viable, running counter to the opisthokont
The opisthokonts () are a broad group of eukaryotes, including both the animal and fungus kingdoms. The opisthokonts, previously called the "Fungi/Metazoa group", are generally recognized as a clade. Opisthokonts together with Apusomonadida and ...
pattern of CDK1-type kinases as essential regulators controlling the cell cycle.
G1/S checkpoint evolution
 The G1/S checkpoint is the point at which the cell commits to division through the cell cycle. Complex regulatory networks lead to the G1/S transition decision. Across opisthokonts, there are both highly diverged protein sequences as well as strikingly similar network topologies.
The G1/S checkpoint is the point at which the cell commits to division through the cell cycle. Complex regulatory networks lead to the G1/S transition decision. Across opisthokonts, there are both highly diverged protein sequences as well as strikingly similar network topologies.transcription factor
In molecular biology, a transcription factor (TF) (or sequence-specific DNA-binding factor) is a protein that controls the rate of transcription (genetics), transcription of genetics, genetic information from DNA to messenger RNA, by binding t ...
s are double-negative feedback loops and positive feedback loops in both yeast and animals.phosphorylation
In biochemistry, phosphorylation is described as the "transfer of a phosphate group" from a donor to an acceptor. A common phosphorylating agent (phosphate donor) is ATP and a common family of acceptor are alcohols:
:
This equation can be writ ...
/de-phosphorylation of CDK-cyclin complexes. The sum of these regulatory networks creates a hysteretic
Hysteresis is the dependence of the state of a system on its history. For example, a magnet may have more than one possible magnetic moment in a given magnetic field, depending on how the field changed in the past. Plots of a single component of ...
and bistable scheme, despite the specific proteins being highly diverged. For yeast, Whi5
Whi5 is a transcriptional regulator in the budding yeast, notably in the G1 phase. It plays an important role in cell size control in G1 phase, similarly with Retinoblastoma (Rb) protein in human, although the two have no similarity in sequenc ...
must be suppressed by Cln3 phosphorylation for SBF to be expressed, while in animals Rb must be suppressed by the Cdk4/6-cyclin D complex for E2F to be expressed.eukaryote
The eukaryotes ( ) constitute the Domain (biology), domain of Eukaryota or Eukarya, organisms whose Cell (biology), cells have a membrane-bound cell nucleus, nucleus. All animals, plants, Fungus, fungi, seaweeds, and many unicellular organisms ...
s as a whole. Plants also share a number of conserved network features with opisthokonts, and many plant regulators have direct animal homologs. For example, plants also need to suppress Rb for E2F translation in the network.
See also
* Cellular model
* Eukaryotic DNA replication
Eukaryotic DNA replication is a conserved mechanism that restricts DNA replication to once per cell cycle. Eukaryotic DNA replication of chromosomal DNA is central for the duplication of a Cell (biology), cell and is necessary for the maintenance ...
* Mitotic catastrophe
Mitotic catastrophe has been defined as either a cellular mechanism to prevent potentially cancerous cells from proliferating or as a mode of cellular death that occurs following improper cell cycle progression or entrance. Mitotic catastrophe can ...
* Origin recognition complex
In molecular biology, origin recognition complex (ORC) is a multi-subunit DNA binding complex (6 subunits) that binds in all eukaryotes and archaea in an Adenosine triphosphate, ATP-dependent manner to origins of replication. The subunits of this ...
* Retinoblastoma protein
Retinoblastoma (Rb) is a rare form of cancer that rapidly develops from the immature cells of a retina, the light-detecting tissue of the eye. It is the most common primary malignant intraocular cancer in children, and 80% of retinoblastoma cas ...
* Synchronous culture – synchronization of cell cultures
* Wee1
References
Notes
Citations
Further reading
*
*
*
*
External links
*
David Morgan's Seminar: Controlling the Cell Cycle
The cell cycle & Cell death
Transcriptional program of the cell cycle: high-resolution timing
Cell cycle and metabolic cycle regulated transcription in yeast
Cell Cycle Animation
1Lec.com
Cell Cycle
Science Creative Quarterly's overview of the cell cycle
{{DEFAULTSORT:Cell Cycle
Cellular senescence

 The cell cycle, or cell-division cycle, is the sequential series of events that take place in a cell that causes it to divide into two daughter cells. These events include the growth of the cell, duplication of its DNA (
The cell cycle, or cell-division cycle, is the sequential series of events that take place in a cell that causes it to divide into two daughter cells. These events include the growth of the cell, duplication of its DNA ( The first phase within interphase, from the end of the previous M phase until the beginning of DNA synthesis, is called G1 (G indicating ''gap''). It is also called the growth phase. During this phase, the biosynthetic activities of the cell, which are considerably slowed down during M phase, resume at a high rate. The duration of G1 is highly variable, even among different cells of the same species. In this phase, the cell increases its supply of proteins, increases the number of organelles (such as mitochondria, ribosomes), and grows in size. In G1 phase, a cell has three options.
*To continue cell cycle and enter
The first phase within interphase, from the end of the previous M phase until the beginning of DNA synthesis, is called G1 (G indicating ''gap''). It is also called the growth phase. During this phase, the biosynthetic activities of the cell, which are considerably slowed down during M phase, resume at a high rate. The duration of G1 is highly variable, even among different cells of the same species. In this phase, the cell increases its supply of proteins, increases the number of organelles (such as mitochondria, ribosomes), and grows in size. In G1 phase, a cell has three options.
*To continue cell cycle and enter  Mitosis is immediately followed by
Mitosis is immediately followed by  It is estimated that in normal human cells about 1% of single-strand DNA damages are converted to about 50 endogenous DNA double-strand breaks per cell per cell cycle. Although such double-strand breaks are usually repaired with high fidelity, errors in their repair are considered to contribute significantly to the rate of cancer in humans.
There are several checkpoints to ensure that damaged or incomplete DNA is not passed on to daughter cells. Three main checkpoints exist: the G1/S checkpoint, the G2/M checkpoint and the metaphase (mitotic) checkpoint. Another checkpoint is the Go checkpoint, in which the cells are checked for maturity. If the cells fail to pass this checkpoint by not being ready yet, they will be discarded from dividing.
G1/S transition is a rate-limiting step in the cell cycle and is also known as
It is estimated that in normal human cells about 1% of single-strand DNA damages are converted to about 50 endogenous DNA double-strand breaks per cell per cell cycle. Although such double-strand breaks are usually repaired with high fidelity, errors in their repair are considered to contribute significantly to the rate of cancer in humans.
There are several checkpoints to ensure that damaged or incomplete DNA is not passed on to daughter cells. Three main checkpoints exist: the G1/S checkpoint, the G2/M checkpoint and the metaphase (mitotic) checkpoint. Another checkpoint is the Go checkpoint, in which the cells are checked for maturity. If the cells fail to pass this checkpoint by not being ready yet, they will be discarded from dividing.
G1/S transition is a rate-limiting step in the cell cycle and is also known as  The G1/S checkpoint is the point at which the cell commits to division through the cell cycle. Complex regulatory networks lead to the G1/S transition decision. Across opisthokonts, there are both highly diverged protein sequences as well as strikingly similar network topologies.
Entry into S-phase in both yeast and animals is controlled by the levels of two opposing regulators. The networks regulating these
The G1/S checkpoint is the point at which the cell commits to division through the cell cycle. Complex regulatory networks lead to the G1/S transition decision. Across opisthokonts, there are both highly diverged protein sequences as well as strikingly similar network topologies.
Entry into S-phase in both yeast and animals is controlled by the levels of two opposing regulators. The networks regulating these