The second law of thermodynamics is a
physical law
Scientific laws or laws of science are statements, based on repeated experiments or observations, that describe or predict a range of natural phenomena. The term ''law'' has diverse usage in many cases (approximate, accurate, broad, or narrow) a ...
based on universal experience concerning
heat
In thermodynamics, heat is defined as the form of energy crossing the boundary of a thermodynamic system by virtue of a temperature difference across the boundary. A thermodynamic system does not ''contain'' heat. Nevertheless, the term is al ...
and
energy interconversions. One simple statement of the law is that heat always moves from hotter objects to colder objects (or "downhill"), unless energy in some form is supplied to reverse the direction of
heat flow
Heat transfer is a discipline of thermal engineering that concerns the generation, use, conversion, and exchange of thermal energy (heat) between physical systems. Heat transfer is classified into various mechanisms, such as thermal conduction, ...
. Another definition is: "Not all heat energy can be converted into
work
Work may refer to:
* Work (human activity), intentional activity people perform to support themselves, others, or the community
** Manual labour, physical work done by humans
** House work, housework, or homemaking
** Working animal, an animal tr ...
in a
cyclic process
A thermodynamic cycle consists of a linked sequence of thermodynamic processes that involve transfer of heat and work into and out of the system, while varying pressure, temperature, and other state variables within the system, and that eventual ...
."
[Young, H. D; Freedman, R. A. (2004). ''University Physics'', 11th edition. Pearson. p. 764.]
The second law of thermodynamics in other versions establishes the concept of
entropy
Entropy is a scientific concept, as well as a measurable physical property, that is most commonly associated with a state of disorder, randomness, or uncertainty. The term and the concept are used in diverse fields, from classical thermodynam ...
as a physical property of a
thermodynamic system
A thermodynamic system is a body of matter and/or radiation, confined in space by walls, with defined permeabilities, which separate it from its surroundings. The surroundings may include other thermodynamic systems, or physical systems that are ...
. It can be used to predict whether processes are forbidden despite obeying the requirement of
conservation of energy
In physics and chemistry, the law of conservation of energy states that the total energy of an isolated system remains constant; it is said to be ''conserved'' over time. This law, first proposed and tested by Émilie du Châtelet, means th ...
as expressed in the
first law of thermodynamics
The first law of thermodynamics is a formulation of the law of conservation of energy, adapted for thermodynamic processes. It distinguishes in principle two forms of energy transfer, heat and thermodynamic work for a system of a constant amoun ...
and provides necessary criteria for
spontaneous process In thermodynamics, a spontaneous process is a process which occurs without any external input to the system. A more technical definition is the time-evolution of a system in which it releases free energy and it moves to a lower, more thermodynamica ...
es. The second law may be formulated by the observation that the entropy of
isolated system
In physical science, an isolated system is either of the following:
# a physical system so far removed from other systems that it does not interact with them.
# a thermodynamic system enclosed by rigid immovable walls through which neither m ...
s left to spontaneous evolution cannot decrease, as they always arrive at a state of
thermodynamic equilibrium
Thermodynamic equilibrium is an axiomatic concept of thermodynamics. It is an internal state of a single thermodynamic system, or a relation between several thermodynamic systems connected by more or less permeable or impermeable walls. In thermod ...
where the entropy is highest at the given internal energy. An increase in the combined entropy of system and surroundings accounts for the
irreversibility
In science, a process that is not reversible is called irreversible. This concept arises frequently in thermodynamics. All complex natural processes are irreversible, although a phase transition at the coexistence temperature (e.g. melting of i ...
of natural processes, often referred to in the concept of the
arrow of time
The arrow of time, also called time's arrow, is the concept positing the "one-way direction" or " asymmetry" of time. It was developed in 1927 by the British astrophysicist Arthur Eddington, and is an unsolved general physics question. This ...
.
Historically, the second law was an
empirical finding that was accepted as an
axiom
An axiom, postulate, or assumption is a statement that is taken to be true, to serve as a premise or starting point for further reasoning and arguments. The word comes from the Ancient Greek word (), meaning 'that which is thought worthy or f ...
of
thermodynamic theory.
Statistical mechanics
In physics, statistical mechanics is a mathematical framework that applies statistical methods and probability theory to large assemblies of microscopic entities. It does not assume or postulate any natural laws, but explains the macroscopic be ...
provides a microscopic explanation of the law in terms of
probability distribution
In probability theory and statistics, a probability distribution is the mathematical function that gives the probabilities of occurrence of different possible outcomes for an experiment. It is a mathematical description of a random phenomenon i ...
s of the states of large assemblies of
atom
Every atom is composed of a nucleus and one or more electrons bound to the nucleus. The nucleus is made of one or more protons and a number of neutrons. Only the most common variety of hydrogen has no neutrons.
Every solid, liquid, gas, and ...
s or
molecule
A molecule is a group of two or more atoms held together by attractive forces known as chemical bonds; depending on context, the term may or may not include ions which satisfy this criterion. In quantum physics, organic chemistry, and bioch ...
s. The second law has been expressed in many ways. Its first formulation, which preceded the proper definition of entropy and was based on
caloric theory
The caloric theory is an obsolete scientific theory that heat consists of a self-repellent fluid called caloric that flows from hotter bodies to colder bodies. Caloric was also thought of as a weightless gas that could pass in and out of pores i ...
, is
Carnot's theorem, formulated by the French scientist
Sadi Carnot, who in 1824 showed that the efficiency of conversion of heat to work in a heat engine has an upper limit. The first rigorous definition of the second law based on the concept of entropy came from German scientist
Rudolf Clausius
Rudolf Julius Emanuel Clausius (; 2 January 1822 – 24 August 1888) was a German physicist and mathematician and is considered one of the central founding fathers of the science of thermodynamics. By his restatement of Sadi Carnot's principle ...
in the 1850s and included his statement that heat can never pass from a colder to a warmer body without some other change, connected therewith, occurring at the same time.
The second law of thermodynamics allows the definition of the concept of
thermodynamic temperature
Thermodynamic temperature is a quantity defined in thermodynamics as distinct from kinetic theory or statistical mechanics.
Historically, thermodynamic temperature was defined by Kelvin in terms of a macroscopic relation between thermodynamic wor ...
, relying also on the
zeroth law of thermodynamics
The zeroth law of thermodynamics is one of the four principal laws of thermodynamics. It provides an independent definition of temperature without reference to entropy, which is defined in the second law. The law was established by Ralph H. Fowl ...
.
Introduction
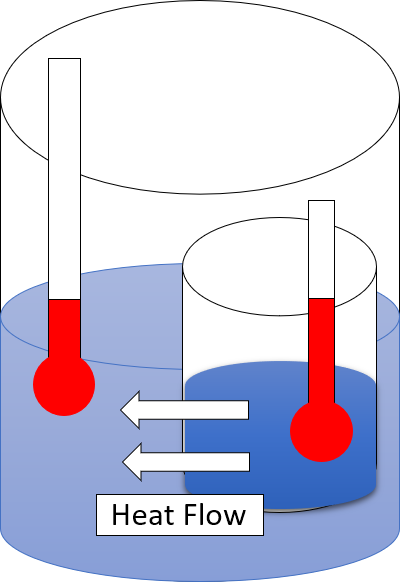
The
first law of thermodynamics
The first law of thermodynamics is a formulation of the law of conservation of energy, adapted for thermodynamic processes. It distinguishes in principle two forms of energy transfer, heat and thermodynamic work for a system of a constant amoun ...
provides the definition of the
internal energy
The internal energy of a thermodynamic system is the total energy contained within it. It is the energy necessary to create or prepare the system in its given internal state, and includes the contributions of potential energy and internal kinet ...
of a
thermodynamic system
A thermodynamic system is a body of matter and/or radiation, confined in space by walls, with defined permeabilities, which separate it from its surroundings. The surroundings may include other thermodynamic systems, or physical systems that are ...
, and expresses its change for a
closed system
A closed system is a natural physical system that does not allow transfer of matter in or out of the system, although — in contexts such as physics, chemistry or engineering — the transfer of energy (''e.g.'' as work or heat) is allowed.
In ...
in terms of
work
Work may refer to:
* Work (human activity), intentional activity people perform to support themselves, others, or the community
** Manual labour, physical work done by humans
** House work, housework, or homemaking
** Working animal, an animal tr ...
and
heat
In thermodynamics, heat is defined as the form of energy crossing the boundary of a thermodynamic system by virtue of a temperature difference across the boundary. A thermodynamic system does not ''contain'' heat. Nevertheless, the term is al ...
. It can be linked to the law of
conservation of energy
In physics and chemistry, the law of conservation of energy states that the total energy of an isolated system remains constant; it is said to be ''conserved'' over time. This law, first proposed and tested by Émilie du Châtelet, means th ...
. The second law is concerned with the direction of natural processes. It asserts that a natural process runs only in one sense, and is not reversible. For example, when a path for conduction or radiation is made available, heat always flows spontaneously from a hotter to a colder body. Such
phenomena
A phenomenon ( : phenomena) is an observable event. The term came into its modern philosophical usage through Immanuel Kant, who contrasted it with the noumenon, which ''cannot'' be directly observed. Kant was heavily influenced by Gottfried W ...
are accounted for in terms of
entropy change. If an isolated system containing distinct subsystems is held initially in internal thermodynamic equilibrium by internal partitioning by impermeable walls between the subsystems, and then some operation makes the walls more permeable, then the system spontaneously evolves to reach a final new internal thermodynamic equilibrium, and its total entropy,
, increases.
In a
reversible or
quasi-static, idealized process of transfer of energy as heat to a
closed thermodynamic system of interest, (which allows the entry or exit of energy – but not transfer of matter), from an auxiliary thermodynamic system, an infinitesimal increment (
) in the entropy of the system of interest is defined to result from an infinitesimal transfer of heat (
) to the system of interest, divided by the common thermodynamic temperature
of the system of interest and the auxiliary thermodynamic system:
:
Different notations are used for an infinitesimal amount of heat
and infinitesimal change of entropy
because entropy is a
function of state
In the thermodynamics of equilibrium, a state function, function of state, or point function for a thermodynamic system is a mathematical function relating several state variables or state quantities (that describe equilibrium states of a syste ...
, while heat, like work, is not.
For an actually possible infinitesimal process without exchange of mass with the surroundings, the second law requires that the increment in system entropy fulfills the
inequality
Inequality may refer to:
Economics
* Attention inequality, unequal distribution of attention across users, groups of people, issues in etc. in attention economy
* Economic inequality, difference in economic well-being between population groups
* ...
:
This is because a general process for this case (no mass exchange between the system and its surroundings) may include work being done on the system by its surroundings, which can have frictional or viscous effects inside the system, because a chemical reaction may be in progress, or because heat transfer actually occurs only irreversibly, driven by a finite difference between the system temperature () and the temperature of the surroundings ().
[Adkins, C.J. (1968/1983), p. 75.]
Note that the equality still applies for pure heat flow (only heat flow, no change in chemical composition and mass),
:
which is the basis of the accurate determination of the absolute entropy of pure substances from measured heat capacity curves and entropy changes at phase transitions, i.e. by calorimetry.
[Oxtoby, D. W; Gillis, H.P., Butler, L. J. (2015).''Principles of Modern Chemistry'', Brooks Cole. p. 617. ]
Introducing a set of internal variables
to describe the deviation of a thermodynamic system from a chemical equilibrium state in physical equilibrium (with the required well-defined uniform pressure ''P'' and temperature ''T''), one can record the equality
:
The second term represents work of internal variables that can be perturbed by external influences, but the system cannot perform any positive work via internal variables. This statement introduces the impossibility of the reversion of evolution of the thermodynamic system in time and can be considered as a formulation of ''the second principle of thermodynamics'' – the formulation, which is, of course, equivalent to the formulation of the principle in terms of entropy.
The
zeroth law of thermodynamics
The zeroth law of thermodynamics is one of the four principal laws of thermodynamics. It provides an independent definition of temperature without reference to entropy, which is defined in the second law. The law was established by Ralph H. Fowl ...
in its usual short statement allows recognition that two bodies in a relation of thermal equilibrium have the same temperature, especially that a test body has the same temperature as a reference thermometric body.
For a body in thermal equilibrium with another, there are indefinitely many empirical temperature scales, in general respectively depending on the properties of a particular reference thermometric body. The second law allows a distinguished temperature scale, which defines an absolute,
thermodynamic temperature
Thermodynamic temperature is a quantity defined in thermodynamics as distinct from kinetic theory or statistical mechanics.
Historically, thermodynamic temperature was defined by Kelvin in terms of a macroscopic relation between thermodynamic wor ...
, independent of the properties of any particular reference thermometric body.
Various statements of the law
The second law of thermodynamics may be expressed in many specific ways,
the most prominent classical statements being the statement by
Rudolf Clausius
Rudolf Julius Emanuel Clausius (; 2 January 1822 – 24 August 1888) was a German physicist and mathematician and is considered one of the central founding fathers of the science of thermodynamics. By his restatement of Sadi Carnot's principle ...
(1854), the statement by
Lord Kelvin
William Thomson, 1st Baron Kelvin, (26 June 182417 December 1907) was a British mathematician, Mathematical physics, mathematical physicist and engineer born in Belfast. Professor of Natural Philosophy (Glasgow), Professor of Natural Philoso ...
(1851), and the statement in axiomatic thermodynamics by
Constantin Carathéodory
Constantin Carathéodory ( el, Κωνσταντίνος Καραθεοδωρή, Konstantinos Karatheodori; 13 September 1873 – 2 February 1950) was a Greek mathematician who spent most of his professional career in Germany. He made significant ...
(1909). These statements cast the law in general physical terms citing the impossibility of certain processes. The Clausius and the Kelvin statements have been shown to be equivalent.
Carnot's principle
The historical origin of the second law of thermodynamics was in
Sadi Carnot's theoretical analysis of the flow of heat in steam engines (1824). The centerpiece of that analysis, now known as a
Carnot engine
A Carnot heat engine is a heat engine that operates on the Carnot cycle. The basic model for this engine was developed by Nicolas Léonard Sadi Carnot in 1824. The Carnot engine model was graphically expanded by Benoît Paul Émile Clapeyron in ...
, is an ideal
heat engine
In thermodynamics and engineering, a heat engine is a system that converts heat to mechanical energy, which can then be used to do mechanical work. It does this by bringing a working substance from a higher state temperature to a lower state ...
fictively operated in the limiting mode of extreme slowness known as quasi-static, so that the heat and work transfers are between subsystems that are always in their own internal states of thermodynamic equilibrium. It represents the theoretical maximum efficiency of a heat engine operating between any two given thermal or heat reservoirs at different temperatures. Carnot's principle was recognized by Carnot at a time when the
caloric theory
The caloric theory is an obsolete scientific theory that heat consists of a self-repellent fluid called caloric that flows from hotter bodies to colder bodies. Caloric was also thought of as a weightless gas that could pass in and out of pores i ...
represented the dominant understanding of the nature of heat, before the recognition of the
first law of thermodynamics
The first law of thermodynamics is a formulation of the law of conservation of energy, adapted for thermodynamic processes. It distinguishes in principle two forms of energy transfer, heat and thermodynamic work for a system of a constant amoun ...
, and before the mathematical expression of the concept of entropy. Interpreted in the light of the first law, Carnot's analysis is physically equivalent to the second law of thermodynamics, and remains valid today. Some samples from his book are:
::...''wherever there exists a difference of temperature, motive power can be produced.''
::The production of motive power is then due in steam engines not to an actual consumption of caloric, but ''to its transportation from a warm body to a cold body ...''
::''The motive power of heat is independent of the agents employed to realize it; its quantity is fixed solely by the temperatures of the bodies between which is effected, finally, the transfer of caloric.''
In modern terms, Carnot's principle may be stated more precisely:
::The efficiency of a quasi-static or reversible Carnot cycle depends only on the temperatures of the two heat reservoirs, and is the same, whatever the working substance. A Carnot engine operated in this way is the most efficient possible heat engine using those two temperatures.
Clausius statement
The German scientist
Rudolf Clausius
Rudolf Julius Emanuel Clausius (; 2 January 1822 – 24 August 1888) was a German physicist and mathematician and is considered one of the central founding fathers of the science of thermodynamics. By his restatement of Sadi Carnot's principle ...
laid the foundation for the second law of thermodynamics in 1850 by examining the relation between heat transfer and work. His formulation of the second law, which was published in German in 1854, is known as the ''Clausius statement'':
Heat can never pass from a colder to a warmer body without some other change, connected therewith, occurring at the same time.
The statement by Clausius uses the concept of 'passage of heat'. As is usual in thermodynamic discussions, this means 'net transfer of energy as heat', and does not refer to contributory transfers one way and the other.
Heat cannot spontaneously flow from cold regions to hot regions without external work being performed on the system, which is evident from ordinary experience of
refrigeration
The term refrigeration refers to the process of removing heat from an enclosed space or substance for the purpose of lowering the temperature.International Dictionary of Refrigeration, http://dictionary.iifiir.org/search.phpASHRAE Terminology, ht ...
, for example. In a refrigerator, heat is transferred from cold to hot, but only when forced by an external agent, the refrigeration system.
Kelvin statements
Lord Kelvin
William Thomson, 1st Baron Kelvin, (26 June 182417 December 1907) was a British mathematician, Mathematical physics, mathematical physicist and engineer born in Belfast. Professor of Natural Philosophy (Glasgow), Professor of Natural Philoso ...
expressed the second law in several wordings.
::It is impossible for a self-acting machine, unaided by any external agency, to convey heat from one body to another at a higher temperature.
::It is impossible, by means of inanimate material agency, to derive mechanical effect from any portion of matter by cooling it below the temperature of the coldest of the surrounding objects.
Equivalence of the Clausius and the Kelvin statements
Suppose there is an engine violating the Kelvin statement: i.e., one that drains heat and converts it completely into work (The drained heat is fully converted to work.) in a cyclic fashion without any other result. Now pair it with a reversed
Carnot engine
A Carnot heat engine is a heat engine that operates on the Carnot cycle. The basic model for this engine was developed by Nicolas Léonard Sadi Carnot in 1824. The Carnot engine model was graphically expanded by Benoît Paul Émile Clapeyron in ...
as shown by the right figure. The
efficiency
Efficiency is the often measurable ability to avoid wasting materials, energy, efforts, money, and time in doing something or in producing a desired result. In a more general sense, it is the ability to do things well, successfully, and without ...
of a normal heat engine is ''η'' and so the efficiency of the reversed heat engine is 1/''η''. The net and sole effect of the combined pair of engines is to transfer heat
from the cooler reservoir to the hotter one, which violates the Clausius statement. This is a consequence of the
first law of thermodynamics
The first law of thermodynamics is a formulation of the law of conservation of energy, adapted for thermodynamic processes. It distinguishes in principle two forms of energy transfer, heat and thermodynamic work for a system of a constant amoun ...
, as for the total system's energy to remain the same;
, so therefore
, where (1) the sign convention of heat is used in which heat entering into (leaving from) an engine is positive (negative) and (2)
is obtained by
the definition of efficiency of the engine when the engine operation is not reversed. Thus a violation of the Kelvin statement implies a violation of the Clausius statement, i.e. the Clausius statement implies the Kelvin statement. We can prove in a similar manner that the Kelvin statement implies the Clausius statement, and hence the two are equivalent.
Planck's proposition
Planck offered the following proposition as derived directly from experience. This is sometimes regarded as his statement of the second law, but he regarded it as a starting point for the derivation of the second law.
::It is impossible to construct an engine which will work in a complete cycle, and produce no effect except the raising of a weight and cooling of a heat reservoir.
Relation between Kelvin's statement and Planck's proposition
It is almost customary in textbooks to speak of the "Kelvin–Planck statement" of the law, as for example in the text by
ter Haar and
Wergeland. This version, also known as the heat engine statement, of the second law states that
::It is impossible to devise a
cyclically operating device, the sole effect of which is to absorb energy in the form of heat from a single
thermal reservoir and to deliver an equivalent amount of
work
Work may refer to:
* Work (human activity), intentional activity people perform to support themselves, others, or the community
** Manual labour, physical work done by humans
** House work, housework, or homemaking
** Working animal, an animal tr ...
.
Planck's statement
Planck stated the second law as follows.
::Every process occurring in nature proceeds in the sense in which the sum of the entropies of all bodies taking part in the process is increased. In the limit, i.e. for reversible processes, the sum of the entropies remains unchanged.
[ Planck, M. (1897/1903), p. 100.][ Planck, M. (1926), p. 463, translation by Uffink, J. (2003), p. 131.][Roberts, J.K., Miller, A.R. (1928/1960), p. 382. This source is partly verbatim from Planck's statement, but does not cite Planck. This source calls the statement the principle of the increase of entropy.]
Rather like Planck's statement is that of Uhlenbeck and Ford for ''irreversible phenomena''.
::... in an irreversible or spontaneous change from one equilibrium state to another (as for example the equalization of temperature of two bodies A and B, when brought in contact) the entropy always increases.
Principle of Carathéodory
Constantin Carathéodory
Constantin Carathéodory ( el, Κωνσταντίνος Καραθεοδωρή, Konstantinos Karatheodori; 13 September 1873 – 2 February 1950) was a Greek mathematician who spent most of his professional career in Germany. He made significant ...
formulated thermodynamics on a purely mathematical axiomatic foundation. His statement of the second law is known as the Principle of Carathéodory, which may be formulated as follows:
In every neighborhood of any state S of an adiabatically enclosed system there are states inaccessible from S.
With this formulation, he described the concept of
adiabatic accessibility
Adiabatic accessibility denotes a certain relation between two equilibrium states of a thermodynamic system (or of different such systems). The concept was coined by Constantin Carathéodory in 1909 ("adiabatische Erreichbarkeit") and taken up 90 ...
for the first time and provided the foundation for a new subfield of classical thermodynamics, often called
geometrical thermodynamics. It follows from Carathéodory's principle that quantity of energy quasi-statically transferred as heat is a holonomic
process function
In thermodynamics, a quantity that is well defined so as to describe the path of a process through the equilibrium state space of a thermodynamic system is termed a process function, or, alternatively, a process quantity, or a path function. As ...
, in other words,
.
Though it is almost customary in textbooks to say that Carathéodory's principle expresses the second law and to treat it as equivalent to the Clausius or to the Kelvin-Planck statements, such is not the case. To get all the content of the second law, Carathéodory's principle needs to be supplemented by Planck's principle, that isochoric work always increases the internal energy of a closed system that was initially in its own internal thermodynamic equilibrium.
[Münster, A. (1970), p. 45.][ Planck, M. (1926).]
Planck's principle
In 1926,
Max Planck
Max Karl Ernst Ludwig Planck (, ; 23 April 1858 – 4 October 1947) was a German theoretical physicist whose discovery of energy quanta won him the Nobel Prize in Physics in 1918.
Planck made many substantial contributions to theoretical p ...
wrote an important paper on the basics of thermodynamics.
He indicated the principle
::The internal energy of a closed system is increased by an adiabatic process, throughout the duration of which, the volume of the system remains constant.
This formulation does not mention heat and does not mention temperature, nor even entropy, and does not necessarily implicitly rely on those concepts, but it implies the content of the second law. A closely related statement is that "Frictional pressure never does positive work." Planck wrote: "The production of heat by friction is irreversible."
Not mentioning entropy, this principle of Planck is stated in physical terms. It is very closely related to the Kelvin statement given just above. It is relevant that for a system at constant volume and
mole numbers, the entropy is a monotonic function of the internal energy. Nevertheless, this principle of Planck is not actually Planck's preferred statement of the second law, which is quoted above, in a previous sub-section of the present section of this present article, and relies on the concept of entropy.
A statement that in a sense is complementary to Planck's principle is made by Borgnakke and Sonntag. They do not offer it as a full statement of the second law:
::... there is only one way in which the entropy of a
losedsystem can be decreased, and that is to transfer heat from the system.
Differing from Planck's just foregoing principle, this one is explicitly in terms of entropy change. Removal of matter from a system can also decrease its entropy.
Statement for a system that has a known expression of its internal energy as a function of its extensive state variables
The second law has been shown to be equivalent to the
internal energy
The internal energy of a thermodynamic system is the total energy contained within it. It is the energy necessary to create or prepare the system in its given internal state, and includes the contributions of potential energy and internal kinet ...
''U'' being a weakly
convex function
In mathematics, a real-valued function is called convex if the line segment between any two points on the graph of a function, graph of the function lies above the graph between the two points. Equivalently, a function is convex if its epigra ...
, when written as a function of extensive properties (mass, volume, entropy, ...).
Corollaries
Perpetual motion of the second kind
Before the establishment of the second law, many people who were interested in inventing a perpetual motion machine had tried to circumvent the restrictions of
first law of thermodynamics
The first law of thermodynamics is a formulation of the law of conservation of energy, adapted for thermodynamic processes. It distinguishes in principle two forms of energy transfer, heat and thermodynamic work for a system of a constant amoun ...
by extracting the massive internal energy of the environment as the power of the machine. Such a machine is called a "perpetual motion machine of the second kind". The second law declared the impossibility of such machines.
Carnot theorem
Carnot's theorem (1824) is a principle that limits the maximum efficiency for any possible engine. The efficiency solely depends on the temperature difference between the hot and cold thermal reservoirs. Carnot's theorem states:
*All irreversible heat engines between two heat reservoirs are less efficient than a
Carnot engine
A Carnot heat engine is a heat engine that operates on the Carnot cycle. The basic model for this engine was developed by Nicolas Léonard Sadi Carnot in 1824. The Carnot engine model was graphically expanded by Benoît Paul Émile Clapeyron in ...
operating between the same reservoirs.
*All reversible heat engines between two heat reservoirs are equally efficient with a Carnot engine operating between the same reservoirs.
In his ideal model, the heat of caloric converted into work could be reinstated by reversing the motion of the cycle, a concept subsequently known as
thermodynamic reversibility
In thermodynamics, a reversible process is a process, involving a system and its surroundings, whose direction can be reversed by infinitesimal changes in some properties of the surroundings, such as pressure or temperature.
Throughout an ent ...
. Carnot, however, further postulated that some caloric is lost, not being converted to mechanical work. Hence, no real heat engine could realize the
Carnot cycle
A Carnot cycle is an ideal thermodynamic cycle proposed by French physicist Sadi Carnot in 1824 and expanded upon by others in the 1830s and 1840s. By Carnot's theorem, it provides an upper limit on the efficiency of any classical thermodynam ...
's reversibility and was condemned to be less efficient.
Though formulated in terms of caloric (see the obsolete
caloric theory
The caloric theory is an obsolete scientific theory that heat consists of a self-repellent fluid called caloric that flows from hotter bodies to colder bodies. Caloric was also thought of as a weightless gas that could pass in and out of pores i ...
), rather than
entropy
Entropy is a scientific concept, as well as a measurable physical property, that is most commonly associated with a state of disorder, randomness, or uncertainty. The term and the concept are used in diverse fields, from classical thermodynam ...
, this was an early insight into the second law.
Clausius inequality
The
Clausius theorem
The Clausius theorem (1855), also known as the ''Clausius inequality'', states that for a thermodynamic system (e.g. heat engine or heat pump) exchanging heat with external thermal reservoirs and undergoing a thermodynamic cycle,
:-\oint dS_\te ...
(1854) states that in a cyclic process
:
The equality holds in the reversible case and the strict inequality holds in the irreversible case, with ''T''
surr as the temperature of the heat bath (surroundings) here. The reversible case is used to introduce the state function
entropy
Entropy is a scientific concept, as well as a measurable physical property, that is most commonly associated with a state of disorder, randomness, or uncertainty. The term and the concept are used in diverse fields, from classical thermodynam ...
. This is because in cyclic processes the variation of a state function is zero from state functionality.
Thermodynamic temperature
For an arbitrary heat engine, the efficiency is:
where ''W''
n is the net work done by the engine per cycle, ''q''
''H'' > 0 is the heat added to the engine from a hot reservoir, and ''q''
''C'' = - , ''q''
''C'', < 0
[.] is waste
heat given off to a cold reservoir from the engine. Thus the efficiency depends only on the ratio , ''q''
''C'', / , ''q''
''H'', .
Carnot's theorem states that all reversible engines operating between the same heat reservoirs are equally efficient. Thus, any reversible heat engine operating between temperatures ''T''
H and ''T''
C must have the same efficiency, that is to say, the efficiency is a function of temperatures only:
In addition, a reversible heat engine operating between temperatures ''T''
1 and ''T''
3 must have the same efficiency as one consisting of two cycles, one between ''T''
1 and another (intermediate) temperature ''T''
2, and the second between ''T''
2 and ''T''
3, where ''T
1'' > ''T
2'' > ''T
3''. This is because, if a part of the two cycle engine is hidden such that it is recognized as an engine between the reservoirs at the temperatures ''T''
1 and ''T''
3, then the efficiency of this engine must be same to the other engine at the same reservoirs. If we choose engines such that work done by the one cycle engine and the two cycle engine are same, then the efficiency of each heat engine is written as the below.
:
,
:
,
:
.
Here, the engine 1 is the one cycle engine, and the engines 2 and 3 make the two cycle engine where there is the intermediate reservoir at ''T''
2. We also have used the fact that the heat
passes through the intermediate thermal reservoir at
without losing its energy. (I.e.,
is not lost during its passage through the reservoir at
.) This fact can be proved by the following.
:
In order to have the consistency in the last equation, the heat
flown from the engine 2 to the intermediate reservoir must be equal to the heat
flown out from the reservoir to the engine 3.
Then
:
Now consider the case where
is a fixed reference temperature: the temperature of the
triple point
In thermodynamics, the triple point of a substance is the temperature and pressure at which the three phases (gas, liquid, and solid) of that substance coexist in thermodynamic equilibrium.. It is that temperature and pressure at which the subli ...
of water as 273.16 Kelvin;
. Then for any ''T''
2 and ''T''
3,
:
Therefore, if thermodynamic temperature ''T''* is defined by
:
then the function ''f'', viewed as a function of thermodynamic temperatures, is simply
:
and the reference temperature ''T''
1* = 273.16 K × ''f''(''T''
1,''T''
1) = 273.16 K. (Any reference temperature and any positive numerical value could be usedthe choice here corresponds to the
Kelvin
The kelvin, symbol K, is the primary unit of temperature in the International System of Units (SI), used alongside its prefixed forms and the degree Celsius. It is named after the Belfast-born and University of Glasgow-based engineer and phys ...
scale.)
Entropy
According to the
Clausius equality, for a ''reversible process''
:
That means the line integral
is path independent for reversible processes.
So we can define a state function S called entropy, which for a reversible process or for pure heat transfer satisfies
:
With this we can only obtain the difference of entropy by integrating the above formula. To obtain the absolute value, we need the
third law of thermodynamics
The third law of thermodynamics states, regarding the properties of closed systems in thermodynamic equilibrium: This constant value cannot depend on any other parameters characterizing the closed system, such as pressure or applied magnetic fiel ...
, which states that ''S'' = 0 at
absolute zero
Absolute zero is the lowest limit of the thermodynamic temperature scale, a state at which the enthalpy and entropy of a cooled ideal gas reach their minimum value, taken as zero kelvin. The fundamental particles of nature have minimum vibration ...
for perfect crystals.
For any irreversible process, since entropy is a state function, we can always connect the initial and terminal states with an imaginary reversible process and integrating on that path to calculate the difference in entropy.
Now reverse the reversible process and combine it with the said irreversible process. Applying the
Clausius inequality
The Clausius theorem (1855), also known as the ''Clausius inequality'', states that for a thermodynamic system (e.g. heat engine or heat pump) exchanging heat with external thermal reservoirs and undergoing a thermodynamic cycle,
:-\oint dS_\te ...
on this loop, with ''T''
surr as the temperature of the surroundings,
:
Thus,
:
where the equality holds if the transformation is reversible.
Notice that if the process is an
adiabatic process
In thermodynamics, an adiabatic process (Greek: ''adiábatos'', "impassable") is a type of thermodynamic process that occurs without transferring heat or mass between the thermodynamic system and its environment. Unlike an isothermal process, ...
, then
, so
.
Energy, available useful work
An important and revealing idealized special case is to consider applying the second law to the scenario of an isolated system (called the total system or universe), made up of two parts: a sub-system of interest, and the sub-system's surroundings. These surroundings are imagined to be so large that they can be considered as an ''unlimited'' heat reservoir at temperature ''T
R'' and pressure ''P
R'' so that no matter how much heat is transferred to (or from) the sub-system, the temperature of the surroundings will remain ''T
R''; and no matter how much the volume of the sub-system expands (or contracts), the pressure of the surroundings will remain ''P
R''.
Whatever changes to ''dS'' and ''dS
R'' occur in the entropies of the sub-system and the surroundings individually, the entropy ''S''
tot of the isolated total system must not decrease according to the second law of thermodynamics:
:
According to the
first law of thermodynamics
The first law of thermodynamics is a formulation of the law of conservation of energy, adapted for thermodynamic processes. It distinguishes in principle two forms of energy transfer, heat and thermodynamic work for a system of a constant amoun ...
, the change ''dU'' in the internal energy of the sub-system is the sum of the heat ''δq'' added to the sub-system, ''less'' any work ''δw'' done ''by'' the sub-system, ''plus'' any net chemical energy entering the sub-system ''d'' Σ''μ
iRN
i'', so that:
:
where ''μ''
''iR'' are the
chemical potential
In thermodynamics, the chemical potential of a species is the energy that can be absorbed or released due to a change of the particle number of the given species, e.g. in a chemical reaction or phase transition. The chemical potential of a species ...
s of chemical species in the external surroundings.
Now the heat leaving the reservoir and entering the sub-system is
:
where we have first used the definition of entropy in classical thermodynamics (alternatively, in statistical thermodynamics, the relation between entropy change, temperature and absorbed heat can be derived); and then the Second Law inequality from above.
It therefore follows that any net work ''δw'' done by the sub-system must obey
:
It is useful to separate the work ''δw'' done by the subsystem into the ''useful'' work ''δw
u'' that can be done ''by'' the sub-system, over and beyond the work ''p
R dV'' done merely by the sub-system expanding against the surrounding external pressure, giving the following relation for the useful work (exergy) that can be done:
:
It is convenient to define the right-hand-side as the exact derivative of a thermodynamic potential, called the ''availability'' or ''
exergy
In thermodynamics, the exergy of a system is the maximum useful work possible during a process that brings the system into equilibrium with a heat reservoir, reaching maximum entropy. When the surroundings are the reservoir, exergy is the potent ...
'' ''E'' of the subsystem,
:
The Second Law therefore implies that for any process which can be considered as divided simply into a subsystem, and an unlimited temperature and pressure reservoir with which it is in contact,
:
i.e. the change in the subsystem's exergy plus the useful work done ''by'' the subsystem (or, the change in the subsystem's exergy less any work, additional to that done by the pressure reservoir, done ''on'' the system) must be less than or equal to zero.
In sum, if a proper ''infinite-reservoir-like'' reference state is chosen as the system surroundings in the real world, then the second law predicts a decrease in ''E'' for an irreversible process and no change for a reversible process.
:
is equivalent to
This expression together with the associated reference state permits a
design engineer
A design engineer is an engineer focused on the engineering design process in any of the various engineering disciplines (including civil, mechanical, electrical, chemical, textiles, aerospace, nuclear, manufacturing, systems, and structural /buil ...
working at the macroscopic scale (above the
thermodynamic limit
In statistical mechanics, the thermodynamic limit or macroscopic limit, of a system is the limit for a large number of particles (e.g., atoms or molecules) where the volume is taken to grow in proportion with the number of particles.S.J. Blundel ...
) to utilize the second law without directly measuring or considering entropy change in a total isolated system. (''Also, see
process engineer
Process engineering is the understanding and application of the fundamental principles and laws of nature that allow humans to transform raw material and energy into products that are useful to society, at an industrial level. By taking advantage ...
''). Those changes have already been considered by the assumption that the system under consideration can reach equilibrium with the reference state without altering the reference state. An efficiency for a process or collection of processes that compares it to the reversible ideal may also be found (''See
second law efficiency Exergy efficiency (also known as the second-law efficiency or rational efficiency) computes the effectiveness of a system relative to its performance in reversible conditions. It is defined as the ratio of the thermal efficiency of an actual system ...
''.)
This approach to the second law is widely utilized in
engineering
Engineering is the use of scientific method, scientific principles to design and build machines, structures, and other items, including bridges, tunnels, roads, vehicles, and buildings. The discipline of engineering encompasses a broad rang ...
practice,
environmental accounting Environmental accounting is a subset of accounting proper, its target being to incorporate both economic and environmental information. It can be conducted at the corporate level or at the level of a national economy through the System of Integrated ...
,
systems ecology
Systems ecology is an interdisciplinary field of ecology, a subset of Earth system science, that takes a holism, holistic approach to the study of ecological systems, especially ecosystems. Systems ecology can be seen as an application of general ...
, and other disciplines.
Direction of spontaneous processes
The second law determines whether a proposed physical or chemical process is forbidden or may occur spontaneously. For
isolated system
In physical science, an isolated system is either of the following:
# a physical system so far removed from other systems that it does not interact with them.
# a thermodynamic system enclosed by rigid immovable walls through which neither m ...
s, no energy is provided by the surroundings and the second law requires that the entropy of the system alone must increase: Δ''S'' > 0. Examples of spontaneous physical processes in isolated systems include the following:
* 1)
Heat can be transferred from a region of higher temperature to a lower temperature (but not the reverse).
* 2) Mechanical energy can be converted to thermal energy (but not the reverse).
* 3) A solute can move from a region of higher concentration to a region of lower concentration (but not the reverse).
However, for some non-isolated systems which can exchange energy with their surroundings, the surroundings exchange enough heat with the system, or do sufficient work on the system, so that the processes occur in the opposite direction. This is possible provided the total entropy change of the system plus the surroundings is positive as required by the second law: Δ''S''
tot = Δ''S'' + Δ''S''
R > 0. For the three examples given above:
* 1) Heat can be transferred from a region of lower temperature to a higher temperature in a
refrigerator
A refrigerator, colloquially fridge, is a commercial and home appliance consisting of a thermally insulated compartment and a heat pump (mechanical, electronic or chemical) that transfers heat from its inside to its external environment so th ...
or in a
heat pump
A heat pump is a device that can heat a building (or part of a building) by transferring thermal energy from the outside using a refrigeration cycle. Many heat pumps can also operate in the opposite direction, cooling the building by removing h ...
. These machines must provide sufficient work to the system.
* 2) Thermal energy can be converted to mechanical work in a
heat engine
In thermodynamics and engineering, a heat engine is a system that converts heat to mechanical energy, which can then be used to do mechanical work. It does this by bringing a working substance from a higher state temperature to a lower state ...
, if sufficient heat is also expelled to the surroundings.
* 3) A solute can move from a region of lower concentration to a region of higher concentration in the biochemical process of
active transport
In cellular biology, ''active transport'' is the movement of molecules or ions across a cell membrane from a region of lower concentration to a region of higher concentration—against the concentration gradient. Active transport requires cellul ...
, if sufficient work is provided by a concentration gradient of a chemical such as
ATP or by an
electrochemical gradient
An electrochemical gradient is a gradient of electrochemical potential, usually for an ion that can move across a membrane. The gradient consists of two parts, the chemical gradient, or difference in solute concentration across a membrane, and th ...
.
The second law in chemical thermodynamics
For a
spontaneous chemical process in a closed system at constant temperature and pressure without non-''PV'' work, the Clausius inequality Δ''S'' > ''Q/T''
surr transforms into a condition for the change in
Gibbs free energy
In thermodynamics, the Gibbs free energy (or Gibbs energy; symbol G) is a thermodynamic potential that can be used to calculate the maximum amount of work that may be performed by a thermodynamically closed system at constant temperature and pr ...
:
or d''G'' < 0. For a similar process at constant temperature and volume, the change in
Helmholtz free energy
In thermodynamics, the Helmholtz free energy (or Helmholtz energy) is a thermodynamic potential that measures the useful work obtainable from a closed thermodynamic system at a constant temperature (isothermal
In thermodynamics, an isotherma ...
must be negative,
. Thus, a negative value of the change in free energy (''G'' or ''A'') is a necessary condition for a process to be spontaneous. This is the most useful form of the second law of thermodynamics in chemistry, where free-energy changes can be calculated from tabulated enthalpies of formation and standard molar entropies of reactants and products.
The chemical equilibrium condition at constant ''T'' and ''p'' without electrical work is d''G'' = 0.
History

The first theory of the conversion of heat into mechanical work is due to
Nicolas Léonard Sadi Carnot
''Sous-lieutenant'' Nicolas Léonard Sadi Carnot (; 1 June 1796 – 24 August 1832) was a French mechanical engineer in the French Army, military scientist and physicist, and often described as the "father of thermodynamics". He published onl ...
in 1824. He was the first to realize correctly that the efficiency of this conversion depends on the difference of temperature between an engine and its surroundings.
Recognizing the significance of
James Prescott Joule
James Prescott Joule (; 24 December 1818 11 October 1889) was an English physicist, mathematician and brewer, born in Salford, Lancashire. Joule studied the nature of heat, and discovered its relationship to mechanical work (see energy). Th ...
's work on the conservation of energy,
Rudolf Clausius
Rudolf Julius Emanuel Clausius (; 2 January 1822 – 24 August 1888) was a German physicist and mathematician and is considered one of the central founding fathers of the science of thermodynamics. By his restatement of Sadi Carnot's principle ...
was the first to formulate the second law during 1850, in this form: heat does not flow ''spontaneously'' from cold to hot bodies. While common knowledge now, this was contrary to the
caloric theory
The caloric theory is an obsolete scientific theory that heat consists of a self-repellent fluid called caloric that flows from hotter bodies to colder bodies. Caloric was also thought of as a weightless gas that could pass in and out of pores i ...
of heat popular at the time, which considered heat as a fluid. From there he was able to infer the principle of Sadi Carnot and the definition of entropy (1865).
Established during the 19th century, the
Kelvin-Planck statement of the Second Law says, "It is impossible for any device that operates on a
cycle to receive heat from a single
reservoir
A reservoir (; from French ''réservoir'' ) is an enlarged lake behind a dam. Such a dam may be either artificial, built to store fresh water or it may be a natural formation.
Reservoirs can be created in a number of ways, including contro ...
and produce a net amount of work." This was shown to be equivalent to the statement of Clausius.
The
ergodic hypothesis
In physics and thermodynamics, the ergodic hypothesis says that, over long periods of time, the time spent by a system in some region of the phase space of microstates with the same energy is proportional to the volume of this region, i.e., th ...
is also important for the
Boltzmann
Ludwig Eduard Boltzmann (; 20 February 1844 – 5 September 1906) was an Austrian physicist and philosopher. His greatest achievements were the development of statistical mechanics, and the statistical explanation of the second law of thermodyn ...
approach. It says that, over long periods of time, the time spent in some region of the phase space of microstates with the same energy is proportional to the volume of this region, i.e. that all accessible microstates are equally probable over a long period of time. Equivalently, it says that time average and average over the statistical ensemble are the same.
There is a traditional doctrine, starting with Clausius, that entropy can be understood in terms of molecular 'disorder' within a
macroscopic system. This doctrine is obsolescent.
[Entropy Sites — A Guide]
Content selected by Frank L. Lambert
Frank L. Lambert (July 10, 1918 – December 28, 2018) was an American academic who was Professor Emeritus of Chemistry at Occidental College, Los Angeles. He is known for his advocacy of changing the definition of thermodynamic entropy as "dis ...
Account given by Clausius

In 1865, the German physicist
Rudolf Clausius
Rudolf Julius Emanuel Clausius (; 2 January 1822 – 24 August 1888) was a German physicist and mathematician and is considered one of the central founding fathers of the science of thermodynamics. By his restatement of Sadi Carnot's principle ...
stated what he called the "second fundamental theorem in the
mechanical theory of heat
The history of thermodynamics is a fundamental strand in the history of physics, the history of chemistry, and the history of science in general. Owing to the relevance of thermodynamics in much of science and technology, its history is finely wov ...
" in the following form:
:
where ''Q'' is heat, ''T'' is temperature and ''N'' is the "equivalence-value" of all uncompensated transformations involved in a cyclical process. Later, in 1865, Clausius would come to define "equivalence-value" as entropy. On the heels of this definition, that same year, the most famous version of the second law was read in a presentation at the Philosophical Society of Zurich on April 24, in which, in the end of his presentation, Clausius concludes:
The entropy of the universe tends to a maximum.
This statement is the best-known phrasing of the second law. Because of the looseness of its language, e.g.
universe
The universe is all of space and time and their contents, including planets, stars, galaxies, and all other forms of matter and energy. The Big Bang theory is the prevailing cosmological description of the development of the universe. Acc ...
, as well as lack of specific conditions, e.g. open, closed, or isolated, many people take this simple statement to mean that the second law of thermodynamics applies virtually to every subject imaginable. This is not true; this statement is only a simplified version of a more extended and precise description.
In terms of time variation, the mathematical statement of the second law for an
isolated system
In physical science, an isolated system is either of the following:
# a physical system so far removed from other systems that it does not interact with them.
# a thermodynamic system enclosed by rigid immovable walls through which neither m ...
undergoing an arbitrary transformation is:
:
where
: ''S'' is the entropy of the system and
: ''t'' is
time
Time is the continued sequence of existence and events that occurs in an apparently irreversible succession from the past, through the present, into the future. It is a component quantity of various measurements used to sequence events, to ...
.
The equality sign applies after equilibration. An alternative way of formulating of the second law for isolated systems is:
:
with
with
the sum of the rate of
entropy production
Entropy production (or generation) is the amount of entropy which is produced in any irreversible processes such as heat and mass transfer processes including motion of bodies, heat exchange, fluid flow, substances expanding or mixing, anelastic d ...
by all processes inside the system. The advantage of this formulation is that it shows the effect of the entropy production. The rate of entropy production is a very important concept since it determines (limits) the efficiency of thermal machines. Multiplied with ambient temperature
it gives the so-called dissipated energy
.
The expression of the second law for closed systems (so, allowing heat exchange and moving boundaries, but not exchange of matter) is:
:
with
Here
:
is the heat flow into the system
:
is the temperature at the point where the heat enters the system.
The equality sign holds in the case that only reversible processes take place inside the system. If irreversible processes take place (which is the case in real systems in operation) the >-sign holds. If heat is supplied to the system at several places we have to take the algebraic sum of the corresponding terms.
For open systems (also allowing exchange of matter):
:
with
Here
is the flow of entropy into the system associated with the flow of matter entering the system. It should not be confused with the time derivative of the entropy. If matter is supplied at several places we have to take the algebraic sum of these contributions.
Statistical mechanics
Statistical mechanics
In physics, statistical mechanics is a mathematical framework that applies statistical methods and probability theory to large assemblies of microscopic entities. It does not assume or postulate any natural laws, but explains the macroscopic be ...
gives an explanation for the second law by postulating that a material is composed of atoms and molecules which are in constant motion. A particular set of positions and velocities for each particle in the system is called a
microstate
A microstate or ministate is a sovereign state having a very small population or very small land area, usually both. However, the meanings of "state" and "very small" are not well-defined in international law.Warrington, E. (1994). "Lilliputs ...
of the system and because of the constant motion, the system is constantly changing its microstate. Statistical mechanics postulates that, in equilibrium, each microstate that the system might be in is equally likely to occur, and when this assumption is made, it leads directly to the conclusion that the second law must hold in a statistical sense. That is, the second law will hold on average, with a statistical variation on the order of 1/ where ''N'' is the number of particles in the system. For everyday (macroscopic) situations, the probability that the second law will be violated is practically zero. However, for systems with a small number of particles, thermodynamic parameters, including the entropy, may show significant statistical deviations from that predicted by the second law. Classical thermodynamic theory does not deal with these statistical variations.
Derivation from statistical mechanics
The first mechanical argument of the
Kinetic theory of gases
Kinetic (Ancient Greek: κίνησις “kinesis”, movement or to move) may refer to:
* Kinetic theory, describing a gas as particles in random motion
* Kinetic energy, the energy of an object that it possesses due to its motion
Art and enter ...
that molecular collisions entail an equalization of temperatures and hence a tendency towards equilibrium was due to
James Clerk Maxwell
James Clerk Maxwell (13 June 1831 – 5 November 1879) was a Scottish mathematician and scientist responsible for the classical theory of electromagnetic radiation, which was the first theory to describe electricity, magnetism and ligh ...
in 1860;
Ludwig Boltzmann
Ludwig Eduard Boltzmann (; 20 February 1844 – 5 September 1906) was an Austrian physicist and philosopher. His greatest achievements were the development of statistical mechanics, and the statistical explanation of the second law of thermodyn ...
with his
H-theorem
In classical statistical mechanics, the ''H''-theorem, introduced by Ludwig Boltzmann in 1872, describes the tendency to decrease in the quantity ''H'' (defined below) in a nearly-ideal gas of molecules.
L. Boltzmann,Weitere Studien über das Wä ...
of 1872 also argued that due to collisions gases should over time tend toward the Maxwell–Boltzmann distribution.
Due to Loschmidt's paradox, derivations of the Second Law have to make an assumption regarding the past, namely that the system is Correlation and dependence, uncorrelated at some time in the past; this allows for simple probabilistic treatment. This assumption is usually thought as a boundary condition, and thus the second Law is ultimately a consequence of the initial conditions somewhere in the past, probably at the beginning of the universe (the Big Bang), though Boltzmann brain, other scenarios have also been suggested.
Given these assumptions, in statistical mechanics, the Second Law is not a postulate, rather it is a consequence of the Statistical mechanics#Fundamental postulate, fundamental postulate, also known as the equal prior probability postulate, so long as one is clear that simple probability arguments are applied only to the future, while for the past there are auxiliary sources of information which tell us that it was low entropy. The first part of the second law, which states that the entropy of a thermally isolated system can only increase, is a trivial consequence of the equal prior probability postulate, if we restrict the notion of the entropy to systems in thermal equilibrium. The entropy of an isolated system in thermal equilibrium containing an amount of energy of
is:
:
where
is the number of quantum states in a small interval between
and
. Here
is a macroscopically small energy interval that is kept fixed. Strictly speaking this means that the entropy depends on the choice of
. However, in the thermodynamic limit (i.e. in the limit of infinitely large system size), the specific entropy (entropy per unit volume or per unit mass) does not depend on
.
Suppose we have an isolated system whose macroscopic state is specified by a number of variables. These macroscopic variables can, e.g., refer to the total volume, the positions of pistons in the system, etc. Then
will depend on the values of these variables. If a variable is not fixed, (e.g. we do not clamp a piston in a certain position), then because all the accessible states are equally likely in equilibrium, the free variable in equilibrium will be such that
is maximized at the given energy of the isolated system
[Young, H. D; Freedman, R. A. (2004). ''University Physics'', 11th edition. Pearson. p. 731.] as that is the most probable situation in equilibrium.
If the variable was initially fixed to some value then upon release and when the new equilibrium has been reached, the fact the variable will adjust itself so that
is maximized, implies that the entropy will have increased or it will have stayed the same (if the value at which the variable was fixed happened to be the equilibrium value).
Suppose we start from an equilibrium situation and we suddenly remove a constraint on a variable. Then right after we do this, there are a number
of accessible microstates, but equilibrium has not yet been reached, so the actual probabilities of the system being in some accessible state are not yet equal to the prior probability of
. We have already seen that in the final equilibrium state, the entropy will have increased or have stayed the same relative to the previous equilibrium state. Boltzmann's
H-theorem
In classical statistical mechanics, the ''H''-theorem, introduced by Ludwig Boltzmann in 1872, describes the tendency to decrease in the quantity ''H'' (defined below) in a nearly-ideal gas of molecules.
L. Boltzmann,Weitere Studien über das Wä ...
, however, proves that the quantity increases monotonically as a function of time during the intermediate out of equilibrium state.
Derivation of the entropy change for reversible processes
The second part of the Second Law states that the entropy change of a system undergoing a reversible process is given by:
:
where the temperature is defined as:
:
See Microcanonical ensemble, here for the justification for this definition. Suppose that the system has some external parameter, ''x'', that can be changed. In general, the energy eigenstates of the system will depend on ''x''. According to the adiabatic theorem of quantum mechanics, in the limit of an infinitely slow change of the system's Hamiltonian, the system will stay in the same energy eigenstate and thus change its energy according to the change in energy of the energy eigenstate it is in.
The generalized force, ''X'', corresponding to the external variable ''x'' is defined such that
is the work performed by the system if ''x'' is increased by an amount ''dx''. For example, if ''x'' is the volume, then ''X'' is the pressure. The generalized force for a system known to be in energy eigenstate
is given by:
:
Since the system can be in any energy eigenstate within an interval of
, we define the generalized force for the system as the expectation value of the above expression:
:
To evaluate the average, we partition the
energy eigenstates by counting how many of them have a value for
within a range between
and
. Calling this number
, we have:
:
The average defining the generalized force can now be written:
:
We can relate this to the derivative of the entropy with respect to ''x'' at constant energy ''E'' as follows. Suppose we change ''x'' to ''x'' + ''dx''. Then
will change because the energy eigenstates depend on ''x'', causing energy eigenstates to move into or out of the range between
and
. Let's focus again on the energy eigenstates for which
lies within the range between
and
. Since these energy eigenstates increase in energy by ''Y dx'', all such energy eigenstates that are in the interval ranging from ''E'' – ''Y'' ''dx'' to ''E'' move from below ''E'' to above ''E''. There are
:
such energy eigenstates. If
, all these energy eigenstates will move into the range between
and
and contribute to an increase in
. The number of energy eigenstates that move from below
to above
is given by
. The difference
:
is thus the net contribution to the increase in
. Note that if ''Y dx'' is larger than
there will be the energy eigenstates that move from below ''E'' to above
. They are counted in both
and
, therefore the above expression is also valid in that case.
Expressing the above expression as a derivative with respect to ''E'' and summing over ''Y'' yields the expression:
:
The logarithmic derivative of
with respect to ''x'' is thus given by:
:
The first term is intensive, i.e. it does not scale with system size. In contrast, the last term scales as the inverse system size and will thus vanish in the thermodynamic limit. We have thus found that:
:
Combining this with
:
gives:
:
Derivation for systems described by the canonical ensemble
If a system is in thermal contact with a heat bath at some temperature ''T'' then, in equilibrium, the probability distribution over the energy eigenvalues are given by the canonical ensemble:
:
Here ''Z'' is a factor that normalizes the sum of all the probabilities to 1, this function is known as the Partition function (statistical mechanics), partition function. We now consider an infinitesimal reversible change in the temperature and in the external parameters on which the energy levels depend. It follows from the general formula for the entropy:
:
that
:
Inserting the formula for
for the canonical ensemble in here gives:
:
Initial conditions at the Big Bang
As elaborated above, it is thought that the second law of thermodynamics is a result of the very low-entropy initial conditions at the Big Bang. From a statistical point of view, these were very special conditions. On the other hand, they were quite simple, as the universe - or at least the part thereof from which the observable universe developed - seems to have been extremely uniform.
This may seem somewhat paradoxical, since in many physical systems uniform conditions (e.g. mixed rather than separated gases) have high entropy. The paradox is solved once realizing that gravitational systems have Heat capacity#Negative heat capacity, negative heat capacity, so that when gravity is important, uniform conditions (e.g. gas of uniform density) in fact have lower entropy compared to non-uniform ones (e.g. black holes in empty space). Yet another approach is that the universe had high (or even maximal) entropy given its size, but as the universe grew it rapidly came out of thermodynamic equilibrium, its entropy only slightly increased compared to the increase in maximal possible entropy, and thus it has arrived at a very low entropy when compared to the much larger possible maximum given its later size.
As for the reason why initial conditions were such, one suggestion is that cosmological inflation was enough to wipe off non-smoothness, while another is that the universe was Hartle–Hawking state, created spontaneously where the mechanism of creation implies low-entropy initial conditions.
Living organisms
There are two principal ways of formulating thermodynamics, (a) through passages from one state of thermodynamic equilibrium to another, and (b) through cyclic processes, by which the system is left unchanged, while the total entropy of the surroundings is increased. These two ways help to understand the processes of life. The thermodynamics of living organisms has been considered by many authors, such as What is Life?, Erwin Schrödinger, Léon Brillouin
and Life and Energy, Isaac Asimov.
To a fair approximation, living organisms may be considered as examples of (b). Approximately, an animal's physical state cycles by the day, leaving the animal nearly unchanged. Animals take in food, water, and oxygen, and, as a result of metabolism, give out breakdown products and heat. Plants Photosynthesis, take in radiative energy from the sun, which may be regarded as heat, and carbon dioxide and water. They give out oxygen. In this way they grow. Eventually they die, and their remains rot away, turning mostly back into carbon dioxide and water. This can be regarded as a cyclic process. Overall, the sunlight is from a high temperature source, the sun, and its energy is passed to a lower temperature sink, i.e. radiated into space. This is an increase of entropy of the surroundings of the plant. Thus animals and plants obey the second law of thermodynamics, considered in terms of cyclic processes.
Furthermore, the ability of living organisms to grow and increase in complexity, as well as to form correlations with their environment in the form of adaption and memory, is not opposed to the second law – rather, it is akin to general results following from it: Under some definitions, an increase in entropy also results in an increase in complexity,
and for a finite system interacting with finite reservoirs, an increase in entropy is equivalent to an increase in correlations between the system and the reservoirs.
Living organisms may be considered as open systems, because matter passes into and out from them. Thermodynamics of open systems is currently often considered in terms of passages from one state of thermodynamic equilibrium to another, or in terms of flows in the approximation of local thermodynamic equilibrium. The problem for living organisms may be further simplified by the approximation of assuming a steady state with unchanging flows. General principles of entropy production for such approximations are subject to an Non-equilibrium thermodynamics, unsettled current debate or research.
Gravitational systems
Commonly, systems for which gravity is not important have a positive heat capacity, meaning that their temperature rises with their internal energy. Therefore, when energy flows from a high-temperature object to a low-temperature object, the source temperature decreases while the sink temperature is increased; hence temperature differences tend to diminish over time.
This is not always the case for systems in which the gravitational force is important: systems that are bound by their own gravity, such as stars, can have negative heat capacities. As they contract, both their total energy and their entropy decrease but Kelvin-Helmholtz mechanism, their internal temperature may increase. This can be significant for protostars and even gas giant planets such as Jupiter.
As gravity is the most important force operating on cosmological scales, it may be difficult or impossible to apply the second law to the universe as a whole.
[Grandy, W.T. (Jr) (2008), p. 151.]
Non-equilibrium states
The theory of classical or thermodynamic equilibrium, equilibrium thermodynamics is idealized. A main postulate or assumption, often not even explicitly stated, is the existence of systems in their own internal states of thermodynamic equilibrium. In general, a region of space containing a physical system at a given time, that may be found in nature, is not in thermodynamic equilibrium, read in the most stringent terms. In looser terms, nothing in the entire universe is or has ever been truly in exact thermodynamic equilibrium.
For purposes of physical analysis, it is often enough convenient to make an assumption of
thermodynamic equilibrium
Thermodynamic equilibrium is an axiomatic concept of thermodynamics. It is an internal state of a single thermodynamic system, or a relation between several thermodynamic systems connected by more or less permeable or impermeable walls. In thermod ...
. Such an assumption may rely on trial and error for its justification. If the assumption is justified, it can often be very valuable and useful because it makes available the theory of thermodynamics. Elements of the equilibrium assumption are that a system is observed to be unchanging over an indefinitely long time, and that there are so many particles in a system, that its particulate nature can be entirely ignored. Under such an equilibrium assumption, in general, there are no macroscopically detectable thermal fluctuations, fluctuations. There is an exception, the case of critical point (thermodynamics), critical states, which exhibit to the naked eye the phenomenon of critical opalescence. For laboratory studies of critical states, exceptionally long observation times are needed.
In all cases, the assumption of thermodynamic equilibrium, once made, implies as a consequence that no putative candidate "fluctuation" alters the entropy of the system.
It can easily happen that a physical system exhibits internal macroscopic changes that are fast enough to invalidate the assumption of the constancy of the entropy. Or that a physical system has so few particles that the particulate nature is manifest in observable fluctuations. Then the assumption of thermodynamic equilibrium is to be abandoned. There is no unqualified general definition of entropy for non-equilibrium states.
There are intermediate cases, in which the assumption of local thermodynamic equilibrium is a very good approximation, but strictly speaking it is still an approximation, not theoretically ideal.
For non-equilibrium situations in general, it may be useful to consider statistical mechanical definitions of other quantities that may be conveniently called 'entropy', but they should not be confused or conflated with thermodynamic entropy properly defined for the second law. These other quantities indeed belong to statistical mechanics, not to thermodynamics, the primary realm of the second law.
The physics of macroscopically observable fluctuations is beyond the scope of this article.
Arrow of time
The second law of thermodynamics is a physical law that is not symmetric to reversal of the time direction. This does not conflict with symmetries observed in the fundamental laws of physics (particularly CPT symmetry) since the second law applies statistically on time-asymmetric boundary conditions. The second law has been related to the difference between moving forwards and backwards in time, or to the principle that cause precedes effect (Arrow of time#Causal arrow of time, the causal arrow of time, or causality).
Irreversibility
Irreversibility in thermodynamic processes is a consequence of the asymmetric character of thermodynamic operations, and not of any internally irreversible microscopic properties of the bodies. Thermodynamic operations are macroscopic external interventions imposed on the participating bodies, not derived from their internal properties. There are reputed "paradoxes" that arise from failure to recognize this.
Loschmidt's paradox
Loschmidt's paradox, also known as the reversibility paradox, is the objection that it should not be possible to deduce an irreversible process from the time-symmetric dynamics that describe the microscopic evolution of a macroscopic system.
In the opinion of Erwin Schrödinger, Schrödinger, "It is now quite obvious in what manner you have to reformulate the law of entropyor for that matter, all other irreversible statementsso that they be capable of being derived from reversible models. You must not speak of one isolated system but at least of two, which you may for the moment consider isolated from the rest of the world, but not always from each other."
[Erwin Schrödinger, Schrödinger, E. (1950), p. 192.] The two systems are isolated from each other by the wall, until it is removed by the thermodynamic operation, as envisaged by the law. The thermodynamic operation is externally imposed, not subject to the reversible microscopic dynamical laws that govern the constituents of the systems. It is the cause of the irreversibility. The statement of the law in this present article complies with Schrödinger's advice. The cause–effect relation is logically prior to the second law, not derived from it.
Poincaré recurrence theorem
The Poincaré recurrence theorem considers a theoretical microscopic description of an isolated physical system. This may be considered as a model of a thermodynamic system after a thermodynamic operation has removed an internal wall. The system will, after a sufficiently long time, return to a microscopically defined state very close to the initial one. The Poincaré recurrence time is the length of time elapsed until the return. It is exceedingly long, likely longer than the life of the universe, and depends sensitively on the geometry of the wall that was removed by the thermodynamic operation. The recurrence theorem may be perceived as apparently contradicting the second law of thermodynamics. More obviously, however, it is simply a microscopic model of thermodynamic equilibrium in an isolated system formed by removal of a wall between two systems. For a typical thermodynamical system, the recurrence time is so large (many many times longer than the lifetime of the universe) that, for all practical purposes, one cannot observe the recurrence. One might wish, nevertheless, to imagine that one could wait for the Poincaré recurrence, and then re-insert the wall that was removed by the thermodynamic operation. It is then evident that the appearance of irreversibility is due to the utter unpredictability of the Poincaré recurrence given only that the initial state was one of thermodynamic equilibrium, as is the case in macroscopic thermodynamics. Even if one could wait for it, one has no practical possibility of picking the right instant at which to re-insert the wall. The Poincaré recurrence theorem provides a solution to Loschmidt's paradox. If an isolated thermodynamic system could be monitored over increasingly many multiples of the average Poincaré recurrence time, the thermodynamic behavior of the system would become invariant under time reversal.

Maxwell's demon
James Clerk Maxwell
James Clerk Maxwell (13 June 1831 – 5 November 1879) was a Scottish mathematician and scientist responsible for the classical theory of electromagnetic radiation, which was the first theory to describe electricity, magnetism and ligh ...
imagined one container divided into two parts, ''A'' and ''B''. Both parts are filled with the same gas at equal temperatures and placed next to each other, separated by a wall. Observing the
molecule
A molecule is a group of two or more atoms held together by attractive forces known as chemical bonds; depending on context, the term may or may not include ions which satisfy this criterion. In quantum physics, organic chemistry, and bioch ...
s on both sides, an imaginary demon guards a microscopic trapdoor in the wall. When a faster-than-average molecule from ''A'' flies towards the trapdoor, the demon opens it, and the molecule will fly from ''A'' to ''B''. The average speed of the molecules in ''B'' will have increased while in ''A'' they will have slowed down on average. Since average molecular speed corresponds to temperature, the temperature decreases in ''A'' and increases in ''B'', contrary to the second law of thermodynamics.
One response to this question was suggested in 1929 by Leó Szilárd and later by Léon Brillouin. Szilárd pointed out that a real-life Maxwell's demon would need to have some means of measuring molecular speed, and that the act of acquiring information would require an expenditure of energy.
Maxwell's 'demon' repeatedly alters the permeability of the wall between ''A'' and ''B''. It is therefore performing thermodynamic operations on a microscopic scale, not just observing ordinary spontaneous or natural macroscopic thermodynamic processes.
Quotations
See also
*Zeroth law of thermodynamics
*First law of thermodynamics
*Third law of thermodynamics
*Clausius–Duhem inequality
*Fluctuation theorem
*Heat death of the universe
*History of thermodynamics
*Jarzynski equality
*Laws of thermodynamics
*Maximum entropy thermodynamics
*Quantum thermodynamics
*Reflections on the Motive Power of Fire
*Relativistic heat conduction
*Thermal diode
*Thermodynamic equilibrium
References
Sources
*
*Peter Atkins, Atkins, P.W., de Paula, J. (2006). ''Atkins' Physical Chemistry'', eighth edition, W.H. Freeman, New York, .
*Attard, P. (2012). ''Non-equilibrium Thermodynamics and Statistical Mechanics: Foundations and Applications'', Oxford University Press, Oxford UK, .
*Baierlein, R. (1999). ''Thermal Physics'', Cambridge University Press, Cambridge UK, .
*Bailyn, M. (1994). ''A Survey of Thermodynamics'', American Institute of Physics, New York, .
*
*Ludwig Boltzmann, Boltzmann, L. (1896/1964). ''Lectures on Gas Theory'', translated by S.G. Brush, University of California Press, Berkeley.
*Borgnakke, C., Sonntag., R.E. (2009). ''Fundamentals of Thermodynamics'', seventh edition, Wiley, .
*Buchdahl, H.A. (1966). ''The Concepts of Classical Thermodynamics'', Cambridge University Press, Cambridge UK.
*Percy Williams Bridgman, Bridgman, P.W. (1943). ''The Nature of Thermodynamics'', Harvard University Press, Cambridge MA.
*Herbert Callen, Callen, H.B. (1960/1985). ''Thermodynamics and an Introduction to Thermostatistics'', (1st edition 1960) 2nd edition 1985, Wiley, New York, .
*. A translation may be foun
here Also a mostly reliabl
translation is to be foundat Kestin, J. (1976). ''The Second Law of Thermodynamics'', Dowden, Hutchinson & Ross, Stroudsburg PA.
*Nicolas Léonard Sadi Carnot, Carnot, S. (1824/1986)
''Reflections on the motive power of fire'' Manchester University Press, Manchester UK,
Also here.*Sydney Chapman (mathematician), Chapman, S., Thomas George Cowling, Cowling, T.G. (1939/1970). ''The Mathematical Theory of Non-uniform gases. An Account of the Kinetic Theory of Viscosity, Thermal Conduction and Diffusion in Gases'', third edition 1970, Cambridge University Press, London.
* Translated into English:
* Translated into English: Reprinted in:
*Denbigh, K. (1954/1981). ''The Principles of Chemical Equilibrium. With Applications in Chemistry and Chemical Engineering'', fourth edition, Cambridge University Press, Cambridge UK, .
*Eu, B.C. (2002). ''Generalized Thermodynamics. The Thermodynamics of Irreversible Processes and Generalized Hydrodynamics'', Kluwer Academic Publishers, Dordrecht, .
*Josiah Willard Gibbs, Gibbs, J.W. (1876/1878). On the equilibrium of heterogeneous substances, ''Trans. Conn. Acad.'', 3: 108–248, 343–524, reprinted in ''The Collected Works of J. Willard Gibbs, Ph.D, LL. D.'', edited by W.R. Longley, R.G. Van Name, Longmans, Green & Co., New York, 1928, volume 1, pp. 55–353.
*Griem, H.R. (2005). ''Principles of Plasma Spectroscopy (Cambridge Monographs on Plasma Physics)'', Cambridge University Press, New York .
*Glansdorff, P., Prigogine, I. (1971). ''Thermodynamic Theory of Structure, Stability, and Fluctuations'', Wiley-Interscience, London, 1971, .
*
*Greven, A., Keller, G., Warnecke (editors) (2003). ''Entropy'', Princeton University Press, Princeton NJ, .
*Edward A. Guggenheim, Guggenheim, E.A. (1949). 'Statistical basis of thermodynamics', ''Research'', 2: 450–454.
*Edward A. Guggenheim, Guggenheim, E.A. (1967). ''Thermodynamics. An Advanced Treatment for Chemists and Physicists'', fifth revised edition, North Holland, Amsterdam.
*Gyarmati, I. (1967/1970) ''Non-equilibrium Thermodynamics. Field Theory and Variational Principles'', translated by E. Gyarmati and W.F. Heinz, Springer, New York.
*Charles Kittel, Kittel, C., Herbert Kroemer, Kroemer, H. (1969/1980). ''Thermal Physics'', second edition, Freeman, San Francisco CA, .
*Kondepudi, D., Ilya Prigogine, Prigogine, I. (1998). ''Modern Thermodynamics: From Heat Engines to Dissipative Structures'', John Wiley & Sons, Chichester, .
*Lebon, G., Jou, D., Casas-Vázquez, J. (2008). ''Understanding Non-equilibrium Thermodynamics: Foundations, Applications, Frontiers'', Springer-Verlag, Berlin, .
*
*Lieb, E.H., Yngvason, J. (2003). The Entropy of Classical Thermodynamics, pp. 147–195, Chapter 8 of ''Entropy'', Greven, A., Keller, G., Warnecke (editors) (2003).
*
*
*Gottfried Wilhelm Leibniz Prize, Müller, I. (1985). ''Thermodynamics'', Pitman, London, .
*Gottfried Wilhelm Leibniz Prize, Müller, I. (2003). Entropy in Nonequilibrium, pp. 79–109, Chapter 5 of ''Entropy'', Greven, A., Keller, G., Warnecke (editors) (2003).
*Münster, A. (1970), ''Classical Thermodynamics'', translated by E.S. Halberstadt, Wiley–Interscience, London, .
*Brian Pippard, Pippard, A.B. (1957/1966). ''Elements of Classical Thermodynamics for Advanced Students of Physics'', original publication 1957, reprint 1966, Cambridge University Press, Cambridge UK.
*
Planck, M. (1897/1903)
''Treatise on Thermodynamics'', translated by A. Ogg, Longmans Green, London, p. 100.*Max Planck, Planck. M. (1914)
''The Theory of Heat Radiation'' a translation by Masius, M. of the second German edition, P. Blakiston's Son & Co., Philadelphia.
*
Planck, M. (1926). Über die Begründung des zweiten Hauptsatzes der Thermodynamik, ''Sitzungsberichte der Preussischen Akademie der Wissenschaften: Physikalisch-mathematische Klasse'': 453–463.
* Pokrovskii V.N. (2005) Extended thermodynamics in a discrete-system approach, Eur. J. Phys. vol. 26, 769–781.
*
*Quinn, T.J. (1983). ''Temperature'', Academic Press, London, .
*
*Roberts, J.K., Miller, A.R. (1928/1960). ''Heat and Thermodynamics'', (first edition 1928), fifth edition, Blackie & Son Limited, Glasgow.
*Erwin Schrödinger, Schrödinger, E. (1950). Irreversibility, ''Proc. R. Ir. Acad.'', A53: 189–195.
*Dirk ter Haar, ter Haar, D., Harald Wergeland, Wergeland, H. (1966). ''Elements of Thermodynamics'', Addison-Wesley Publishing, Reading MA.
* Also published in
*William Thomson, 1st Baron Kelvin, Thomson, W. (1852). ''On the universal tendency in nature to the dissipation of mechanical energy'' Philosophical Magazine, Ser. 4, p. 304.
*László Tisza, Tisza, L. (1966). ''Generalized Thermodynamics'', M.I.T Press, Cambridge MA.
*Clifford Truesdell, Truesdell, C. (1980). ''The Tragicomical History of Thermodynamics 1822–1854'', Springer, New York, .
*Uffink, J. (2001). Bluff your way in the second law of thermodynamics, ''Stud. Hist. Phil. Mod. Phys.'', 32(3): 305–394.
*Uffink, J. (2003). Irreversibility and the Second Law of Thermodynamics, Chapter 7 of ''Entropy'', Greven, A., Keller, G., Warnecke (editors) (2003), Princeton University Press, Princeton NJ, .
*George Uhlenbeck, Uhlenbeck, G.E., Ford, G.W. (1963). ''Lectures in Statistical Mechanics'', American Mathematical Society, Providence RI.
*Mark Zemansky, Zemansky, M.W. (1968). ''Heat and Thermodynamics. An Intermediate Textbook'', fifth edition, McGraw-Hill Book Company, New York.
Further reading
*Goldstein, Martin, and Inge F., 1993. ''The Refrigerator and the Universe''. Harvard Univ. Press. Chpts. 4–9 contain an introduction to the Second Law, one a bit less technical than this entry.
*Leff, Harvey S., and Rex, Andrew F. (eds.) 2003. ''Maxwell's Demon 2 : Entropy, classical and quantum information, computing''. Bristol UK; Philadelphia PA: Institute of Physics.
*(technical).
*
full text of 1897 ed.html)
*Stephen Jay Kline (1999). ''The Low-Down on Entropy and Interpretive Thermodynamics'', La Cañada, CA: DCW Industries. .
* also a
External links
*Stanford Encyclopedia of Philosophy:
Philosophy of Statistical Mechanics – by Lawrence Sklar.
in the MIT Cours
from Prof. Z. S. Spakovszky
*E.T. Jaynes, 1988,
The evolution of Carnot's principle" in G. J. Erickson and C. R. Smith (eds.)''Maximum-Entropy and Bayesian Methods in Science and Engineering'', Vol,.1: p. 267.
Caratheodory, C., "Examination of the foundations of thermodynamics," trans. by D. H. DelphenichThe Second Law of Thermodynamics BBC Radio 4 discussion with John Gribbin, Peter Atkins & Monica Grady (''In Our Time'', Dec. 16, 2004)
"The Second Law Mystique" Alexey Nikulov and Daniel Sheehan, ''Entropy (journal), Entropy'', 2004
The Journal of the International Society for the History of Philosophy of Science, 2012
{{DEFAULTSORT:Second Law Of Thermodynamics
Equations of physics
Laws of thermodynamics, 2
Non-equilibrium thermodynamics
Philosophy of thermal and statistical physics
 The
The  The first theory of the conversion of heat into mechanical work is due to
The first theory of the conversion of heat into mechanical work is due to  In 1865, the German physicist
In 1865, the German physicist 