pyramidane on:
[Wikipedia]
[Google]
[Amazon]
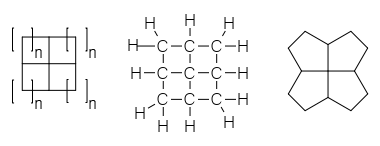 A fenestrane in
A fenestrane in

 Structures within this class of chemicals can be named according to the number of atoms in each ring in addition to the
Structures within this class of chemicals can be named according to the number of atoms in each ring in addition to the  In addition to the ring sizes, fenestranes can have various combinations of cis and trans geometry at each ring fusion. These details are denoted by "''c''" and "''t''" prefixes to the structure name, listed in the same order as the ring-sizes. For example, ''c'',''t'',''c'',''c''- .5.5.5enestrane has a trans configuration at one of the cyclopentane/cyclopentane fusions, but cis configuration at the other cyclopentane/cyclopentane fusion and at both butanepentane/cyclopentane fusions.
In an extreme case the central carbon atom, which would ordinarily have
In addition to the ring sizes, fenestranes can have various combinations of cis and trans geometry at each ring fusion. These details are denoted by "''c''" and "''t''" prefixes to the structure name, listed in the same order as the ring-sizes. For example, ''c'',''t'',''c'',''c''- .5.5.5enestrane has a trans configuration at one of the cyclopentane/cyclopentane fusions, but cis configuration at the other cyclopentane/cyclopentane fusion and at both butanepentane/cyclopentane fusions.
In an extreme case the central carbon atom, which would ordinarily have 
 In the
In the 
 A fenestrane in
A fenestrane in organic chemistry
Organic chemistry is a subdiscipline within chemistry involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms.Clayden, ...
is a type of chemical compound
A chemical compound is a chemical substance composed of many identical molecules (or molecular entities) containing atoms from more than one chemical element held together by chemical bonds. A molecule consisting of atoms of only one element ...
with a central quaternary carbon
A quaternary carbon is a carbon atom bound to four other carbon atoms. For this reason, quaternary carbon atoms are found only in hydrocarbons having at least five carbon atoms. Quaternary carbon atoms can occur in branched alkanes, but not in li ...
atom which serves as a common vertex for four fused carbocycle
In organic chemistry, an alicyclic compound contains one or more all-carbon rings which may be either saturated or unsaturated, but do not have aromatic character. Alicyclic compounds may have one or more aliphatic side chains attached.
The s ...
s. They can be regarded as spiro compound
In organic chemistry, spiro compounds are compounds that have at least two molecular rings with only one common atom. The simplest spiro compounds are bicyclic (having just two rings), or have a bicyclic portion as part of the larger ring sy ...
s twice over. Because of their inherent strain
Strain may refer to:
Science and technology
* Strain (biology), variants of plants, viruses or bacteria; or an inbred animal used for experimental purposes
* Strain (chemistry), a chemical stress of a molecule
* Strain (injury), an injury to a mu ...
and instability, fenestranes are of theoretical interest to chemists. The name—proposed in 1972 by Vlasios Georgian and Martin Saltzman—is derived from the Latin
Latin (, or , ) is a classical language belonging to the Italic branch of the Indo-European languages. Latin was originally a dialect spoken in the lower Tiber area (then known as Latium) around present-day Rome, but through the power of the ...
word for window
A window is an opening in a wall, door, roof, or vehicle that allows the exchange of light and may also allow the passage of sound and sometimes air. Modern windows are usually glazed or covered in some other transparent or translucent mat ...
, ''fenestra''. Georgian had intended that "fenestrane" solely referred to .4.4.4enestrane, whose skeletal structure looks like windows, and Kenneth B. Wiberg called that specific structure "windowpane". The term ''fenestrane'' has since become generalized to refer to the whole class of molecules that have various other ring-sizes. Georgian recommended ''rosettane'' for the class, based on the structural appearance as a rosette of flowers.
Nomenclature and structure

 Structures within this class of chemicals can be named according to the number of atoms in each ring in addition to the
Structures within this class of chemicals can be named according to the number of atoms in each ring in addition to the systematic nomenclature
A chemical nomenclature is a set of rules to generate systematic names for chemical compounds. The nomenclature used most frequently worldwide is the one created and developed by the International Union of Pure and Applied Chemistry (IUPAC).
The ...
of IUPAC naming rules. The smallest member of the family, consisting of four fused cyclopropane rings, is .3.3.3enestrane, which has systematic name tetracyclo .1.0.01,3.02,5entane and is also called pyramidane. The next symmetric member, .4.4.4enestrane, has four cyclobutane
Cyclobutane is a cycloalkane and organic compound with the formula (CH2)4. Cyclobutane is a colourless gas and commercially available as a liquefied gas. Derivatives of cyclobutane are called cyclobutanes. Cyclobutane itself is of no commercia ...
rings fused, and has systematic name tetracyclo .3.1.03,9.07,9onane. The rings need not all be the same size as each other, so .4.4.5enestrane has three cyclobutane rings and one cyclopentane
Cyclopentane (also called C pentane) is a highly flammable alicyclic hydrocarbon with chemical formula C5H10 and CAS number 287-92-3, consisting of a ring of five carbon atoms each bonded with two hydrogen atoms above and below the plane. It occu ...
ring. Other structural modifications vary the name as usual in systematic nomenclature, so a .6.4.6enestradiene has two cyclobutane rings and two cyclohexane rings in an alternating pattern and two alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
units in the ring structure.
 In addition to the ring sizes, fenestranes can have various combinations of cis and trans geometry at each ring fusion. These details are denoted by "''c''" and "''t''" prefixes to the structure name, listed in the same order as the ring-sizes. For example, ''c'',''t'',''c'',''c''- .5.5.5enestrane has a trans configuration at one of the cyclopentane/cyclopentane fusions, but cis configuration at the other cyclopentane/cyclopentane fusion and at both butanepentane/cyclopentane fusions.
In an extreme case the central carbon atom, which would ordinarily have
In addition to the ring sizes, fenestranes can have various combinations of cis and trans geometry at each ring fusion. These details are denoted by "''c''" and "''t''" prefixes to the structure name, listed in the same order as the ring-sizes. For example, ''c'',''t'',''c'',''c''- .5.5.5enestrane has a trans configuration at one of the cyclopentane/cyclopentane fusions, but cis configuration at the other cyclopentane/cyclopentane fusion and at both butanepentane/cyclopentane fusions.
In an extreme case the central carbon atom, which would ordinarily have tetrahedral molecular geometry
In a tetrahedral molecular geometry, a central atom is located at the center with four substituents that are located at the corners of a tetrahedron. The bond angles are arccosine, cos−1(−) = 109.4712206...° ≈ 109.5° when all four substit ...
for its four bonds gets completely flattened. In the molecular orbital
In chemistry, a molecular orbital is a mathematical function describing the location and wave-like behavior of an electron in a molecule. This function can be used to calculate chemical and physical properties such as the probability of findin ...
picture for the resulting square planar
The square planar molecular geometry in chemistry describes the stereochemistry (spatial arrangement of atoms) that is adopted by certain chemical compounds. As the name suggests, molecules of this geometry have their atoms positioned at the corne ...
geometry of methane
Methane ( , ) is a chemical compound with the chemical formula (one carbon atom bonded to four hydrogen atoms). It is a group-14 hydride, the simplest alkane, and the main constituent of natural gas. The relative abundance of methane on Ea ...
, two of a total of three sp2-hybridized carbon atomic orbitals form regular bonds with two of the hydrogen atoms as in a planar alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
. The third sp2 orbital interacts in a three-center two-electron bond
A three-center two-electron (3c–2e) bond is an electron-deficient chemical bond where three atoms share two electrons. The combination of three atomic orbitals form three molecular orbitals: one bonding, one ''non''-bonding, and one ''anti''-b ...
with the two remaining hydrogen atoms utilizing only the hydrogen electrons. Two additional carbon valence electron
In chemistry and physics, a valence electron is an electron in the outer shell associated with an atom, and that can participate in the formation of a chemical bond if the outer shell is not closed. In a single covalent bond, a shared pair form ...
s are situated in a p orbital perpendicular to the plane of the molecule. The four C–H bonds are equal due to resonance
Resonance describes the phenomenon of increased amplitude that occurs when the frequency of an applied Periodic function, periodic force (or a Fourier analysis, Fourier component of it) is equal or close to a natural frequency of the system ...
. In silico calculations show that it takes 95 to 250 kcal/mol (400 to 1,050 kJ/mol) for this process.
One of the most highly strained fenestranes to have been isolated is a .4.4.5enestrane with bond angle
Bond or bonds may refer to:
Common meanings
* Bond (finance), a type of debt security
* Bail bond, a commercial third-party guarantor of surety bonds in the United States
* Chemical bond, the attraction of atoms, ions or molecules to form chemical ...
s at the central carbon atom of around 130° (based on X-ray crystallography
X-ray crystallography is the experimental science determining the atomic and molecular structure of a crystal, in which the crystalline structure causes a beam of incident X-rays to diffract into many specific directions. By measuring the angles ...
), as compared to the 109.45° standard for tetrahedral atoms. The carbon–carbon bond-lengths deviate from those of normal alkanes as well. Whereas the C–C bond in ethane
Ethane ( , ) is an organic chemical compound with chemical formula . At standard temperature and pressure, ethane is a colorless, odorless gas. Like many hydrocarbons, ethane is isolated on an industrial scale from natural gas and as a petroc ...
is 155 pm, in this fenestrane, the bonds extending from the central carbon atom are shortened to 149 pm while those at the perimeter are lengthened to 159 pm.
A diterpene
Diterpenes are a class of chemical compounds composed of four isoprene units, often with the molecular formula C20H32. They are biosynthesized by plants, animals and fungi via the HMG-CoA reductase pathway, with geranylgeranyl pyrophosphate being ...
called laurenene containing a .5.5.7enestrane ring system was the first natural fenestrane to be discovered. The first fenestrane ever synthesized was a .5.5.6enestrane:

Pyramidanes
Pyramidane ( .3.3.3enestrane) is the smallest possible fenestrane, and has never been synthesised. If the central carbon were to be tetrahedral, it would have the form ofspiropentadiene
Spiropentadiene, or bowtiediene, is a hydrocarbon with formula C5H4. The simplest spiro-connected cycloalkene, it is very unstable—decomposing even below −100 °C—due to its high bond strain and does not occur in nature. I ...
, but with additional bonds between the two cyclopropyl rings rather than double-bonds within them. The analogous germa- and stannapyramidanes, with trimethylsilyl
A trimethylsilyl group (abbreviated TMS) is a functional group in organic chemistry. This group consists of three methyl groups bonded to a silicon atom minus;Si(CH3)3 which is in turn bonded to the rest of a molecule. This structural group is ch ...
groups bonded to the corners, Ge 4(SiMe3)4and Sn 4(SiMe3)4on the other hand have been synthesised. These adopt a square pyramidal
In molecular geometry, square pyramidal geometry describes the shape of certain Chemical compound, compounds with the formula where L is a ligand. If the ligand atoms were connected, the resulting shape would be that of a Square pyramid, pyram ...
geometry analogous to the trigonal pyramid of tetrahedrane
Tetrahedrane is a hypothetical platonic hydrocarbon with chemical formula and a tetrahedral structure. The molecule would be subject to considerable angle strain and has not been synthesized as of 2021. However, a number of derivatives have be ...
, with the germanium or tin
Tin is a chemical element with the symbol Sn (from la, stannum) and atomic number 50. Tin is a silvery-coloured metal.
Tin is soft enough to be cut with little force and a bar of tin can be bent by hand with little effort. When bent, t ...
atom at the vertex. That atom has an inverted tetrahedral geometry
In a tetrahedral molecular geometry, a central atom is located at the center with four substituents that are located at the corners of a tetrahedron. The bond angles are cos−1(−) = 109.4712206...° ≈ 109.5° when all four substituents are ...
. According to nuclear magnetic resonance
Nuclear magnetic resonance (NMR) is a physical phenomenon in which nuclei in a strong constant magnetic field are perturbed by a weak oscillating magnetic field (in the near field) and respond by producing an electromagnetic signal with a ...
analysis, the four carbons of the base of the pyramid behave as an aromatic ring
In chemistry, aromaticity is a chemical property of cyclic ( ring-shaped), ''typically'' planar (flat) molecular structures with pi bonds in resonance (those containing delocalized electrons) that gives increased stability compared to satur ...
.
Synthetic approaches
In one study, a .5.5.5enestrane was synthesized with one carbon atom replaced by nitrogen because aza- compounds and their salts are more likely to form crystalline compounds suitable for X-ray analysis than low-molecular-weight alkanes. In step 1 thealkyl halide
The haloalkanes (also known as halogenoalkanes or alkyl halides) are alkanes containing one or more halogen substituents. They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalkanes are widely us ...
1-iodo-3-butene 1 is converted to a cyanozinc cuprate
Cuprate loosely refers to a material that can be viewed as containing anionic copper complexes. Examples include tetrachloridocuprate ( uCl4sup>2−), the superconductor YBa2Cu3O7, and the organocuprates (e.g., dimethylcuprate u(CH3)2sup>� ...
2 (by transmetalation of the organozinc
Organozinc compounds in organic chemistry contain carbon (C) to zinc (Zn) chemical bonds. Organozinc chemistry is the science of organozinc compounds describing their physical properties, synthesis and reactions.The Chemistry of Organozinc Compoun ...
iodide with copper cyanide) which reacts in the next step with 1-nitrocyclopentene 3 in a nucleophilic addition whereby the nitronate
A nitronate (IUPAC: azinate) in organic chemistry is a functional group with the general structure . It is the anion of nitronic acid (sometimes also called an aci, or an azinic acid), a tautomeric form of a nitro compound. Just as ketones and al ...
4 is captured by phenylselenenyl bromide to the selenium intermediate 5. Hydrogen peroxide
Hydrogen peroxide is a chemical compound with the formula . In its pure form, it is a very pale blue liquid that is slightly more viscous than water. It is used as an oxidizer, bleaching agent, and antiseptic, usually as a dilute solution (3%� ...
oxidation
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a ...
of 5 yields the nitroalkene 6 as a mixture of ''syn'' and ''anti'' isomers. A +2 ycloaddition with ''n''-butylenol ether
In organic chemistry an enol ether is an alkene with an alkoxy substituent. The general structure is R2C=CR-OR where R = H, alkyl or aryl. A common subfamily of enol ethers are vinyl ethers, with the formula ROCH=CH2. Important enol ethers includ ...
in presence of trimethylaluminium
Trimethylaluminium is one of the simplest examples of an organoaluminium compound. Despite its name it has the formula Al2( CH3)6 (abbreviated as Al2Me6 or TMA), as it exists as a dimer. This colorless liquid is pyrophoric. It is an industriall ...
gives the nitronate
A nitronate (IUPAC: azinate) in organic chemistry is a functional group with the general structure . It is the anion of nitronic acid (sometimes also called an aci, or an azinic acid), a tautomeric form of a nitro compound. Just as ketones and al ...
7 and a second +2ycloaddition by heating in presence of potassium carbonate
Potassium carbonate is the inorganic compound with the formula K2 CO3. It is a white salt, which is soluble in water. It is deliquescent, often appearing as a damp or wet solid. Potassium carbonate is mainly used in the production of soap and gl ...
gives the nitroso
In organic chemistry, nitroso refers to a functional group in which the nitric oxide () group is attached to an organic moiety (chemistry), moiety. As such, various nitroso groups can be categorized as ''C''-nitroso compounds (e.g., nitrosoalkane ...
acetal
In organic chemistry, an acetal is a functional group with the connectivity . Here, the R groups can be organic fragments (a carbon atom, with arbitrary other atoms attached to that) or hydrogen, while the R' groups must be organic fragments n ...
8. Hydrogenation
Hydrogenation is a chemical reaction between molecular hydrogen (H2) and another compound or element, usually in the presence of a catalyst such as nickel, palladium or platinum. The process is commonly employed to reduce or saturate organ ...
with Raney nickel gives the diol 9 which on a double Mitsunobu reaction
The Mitsunobu reaction is an organic reaction that converts an alcohol into a variety of functional groups, such as an ester, using triphenylphosphine and an azodicarboxylate such as diethyl azodicarboxylate (DEAD) or diisopropyl azodicarboxylat ...
(with an amine proton donor) gives the azafenestrane 10 as the borane
Trihydridoboron, also known as borane or borine, is an unstable and highly reactive molecule with the chemical formula . The preparation of borane carbonyl, BH3(CO), played an important role in exploring the chemistry of boranes, as it indicated ...
salt.
 In the
In the borane
Trihydridoboron, also known as borane or borine, is an unstable and highly reactive molecule with the chemical formula . The preparation of borane carbonyl, BH3(CO), played an important role in exploring the chemistry of boranes, as it indicated ...
salt the N–C–C bond angle
Bond or bonds may refer to:
Common meanings
* Bond (finance), a type of debt security
* Bail bond, a commercial third-party guarantor of surety bonds in the United States
* Chemical bond, the attraction of atoms, ions or molecules to form chemical ...
is 126°.
One study describes an unusual 8π disrotatory
An electrocyclic reaction can either be classified as conrotatory or disrotatory based on the rotation at each end of the molecule. In conrotatory mode, both atomic orbitals of the end groups turn in the same direction (such as both atomic orbital ...
– 6π conrotatory
An electrocyclic reaction can either be classified as conrotatory or disrotatory based on the rotation at each end of the molecule. In conrotatory mode, both atomic orbitals of the end groups turn in the same direction (such as both atomic orbital ...
electrocyclic In organic chemistry, an electrocyclic reaction is a type of pericyclic rearrangement where the net result is one pi bond being converted into one sigma bond or vice versa. These reactions are usually categorized by the following criteria:
* React ...
cascade reaction aiming to minimise the number of steps required to synthesise a fenestrane.Reagents: P-2 Ni (Ni(OAc)2·4H2O) / hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic ...
gas. Reaction initiated by organic reduction
Organic reductions or organic oxidations or organic redox reactions are redox reactions that take place with organic compounds. In organic chemistry oxidations and reductions are different from ordinary redox reactions, because many reactions car ...
of alkyne to alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
See also
*Acepentalene
Acepentalene is a tricyclic anti-aromatic compound. Its molecular formula is . It consists of three five-membered rings fused across three of the five carbon atoms. The central carbon atom in acepentalene is part of all three rings. There are for ...
* Triquinacene
References
{{Reflist Polycyclic nonaromatic hydrocarbons