Tetraborate on:
[Wikipedia]
[Google]
[Amazon]
 In chemistry, tetraborate or pyroborate is an
In chemistry, tetraborate or pyroborate is an
 In chemistry, tetraborate or pyroborate is an
In chemistry, tetraborate or pyroborate is an anion
An ion () is an atom or molecule with a net electrical charge.
The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by conve ...
(negative ion) with formula ; or a salt
Salt is a mineral composed primarily of sodium chloride (NaCl), a chemical compound belonging to the larger class of salts; salt in the form of a natural crystalline mineral is known as rock salt or halite. Salt is present in vast quanti ...
containing that anion, such as sodium tetraborate
Borax is a salt (chemistry), salt (ionic compound), a hydration (chemistry), hydrated borate of sodium, with chemical formula often written . It is a colorless crystalline solid, that dissolves in water to make a base (chemistry), basic aqueo ...
, . It is one of the boron oxoacid
An oxyacid, oxoacid, or ternary acid is an acid that contains oxygen. Specifically, it is a compound that contains hydrogen, oxygen, and at least one other element, with at least one hydrogen atom bonded to oxygen that can dissociate to produce ...
s, that is, a borate
A borate is any of several boron oxyanions, negative ions consisting of boron and oxygen, such as orthoborate , metaborate , or tetraborate ; or any salt with such anions, such as sodium metaborate, and disodium tetraborate . The name a ...
.
The name is also applied to the hydrated ion as present in borax
Borax is a salt (chemistry), salt (ionic compound), a hydration (chemistry), hydrated borate of sodium, with chemical formula often written . It is a colorless crystalline solid, that dissolves in water to make a base (chemistry), basic aqueo ...
The ion occurs in boric acid
Boric acid, more specifically orthoboric acid, is a compound of boron, oxygen, and hydrogen with formula . It may also be called hydrogen borate or boracic acid. It is usually encountered as colorless crystals or a white powder, that dissolve ...
solutions at neutral pH, being formed by condensation of orthoborate and tetrahydroxyborate anions:
: 2 B(OH)3 + 2 ⇌ + 5 H2O
The tetraborate anion (tetramer
A tetramer () (''tetra-'', "four" + '' -mer'', "parts") is an oligomer formed from four monomers or subunits. The associated property is called ''tetramery''. An example from inorganic chemistry is titanium methoxide with the empirical formula T ...
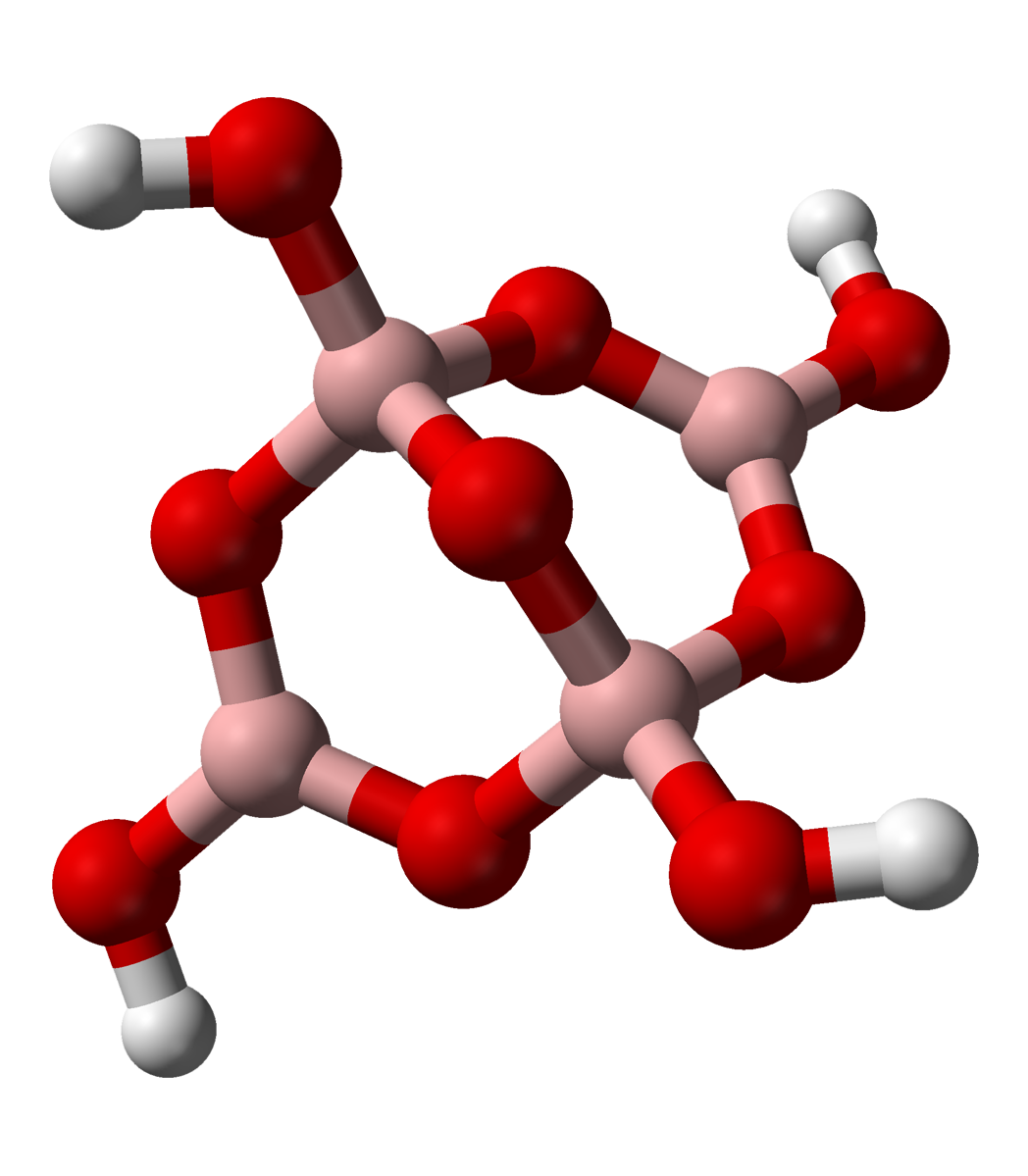
) includes two tetrahedral and two trigonal boron atoms symmetrically assembled in a fused bicyclic structure. The two tetrahedral boron atoms are linked together by a common oxygen atom, and each also bears a negative net charge brought by the supplementary OH− groups laterally attached to them. This intricate molecular anion also exhibits three rings: two fused distorted hexagonal (boroxole) rings and one distorted octagonal ring. Each ring is made of a succession of alternate boron and oxygen atoms. Boroxole rings are a very common structural motif in polyborate ions.
The hydrated tetraborate anion occurs in the mineral borax
Borax is a salt (chemistry), salt (ionic compound), a hydration (chemistry), hydrated borate of sodium, with chemical formula often written . It is a colorless crystalline solid, that dissolves in water to make a base (chemistry), basic aqueo ...
(sodium tetraborate octahydrate) with the formula Na2 4O5(OH)4�8H2O. The borax chemical formula is also commonly written in a more compact notation as Na2B4O7·10H2O. Sodium borate can be obtained in high purity and so can be used to make a standard solution In analytical chemistry, a standard solution is a solution containing a precisely known concentration of an element or a substance. A known mass of solute is dissolved to make a specific volume. It is prepared using a standard substance, such as a ...
in titrimetric analysis..
References
Anions Salts Sodium Borates {{chem-stub