Solenopsin on:
[Wikipedia]
[Google]
[Amazon]
Solenopsin is a lipophilic
alkaloid
Alkaloids are a class of basic
BASIC (Beginners' All-purpose Symbolic Instruction Code) is a family of general-purpose, high-level programming languages designed for ease of use. The original version was created by John G. Kemeny and Th ...
with the molecular formula C17H35N found in the venom of fire ant
Fire ants are several species of ants in the genus ''Solenopsis'', which includes over 200 species. ''Solenopsis'' are stinging ants, and most of their common names reflect this, for example, ginger ants and tropical fire ants. Many of the nam ...
s (''Solenopsis''). It is considered the primary toxin in the venom and may be the component responsible for the cardiorespiratory failure in people who experience excessive fire ant stings.
Structurally solenopsins are a piperidine
Piperidine is an organic compound with the molecular formula (CH2)5NH. This heterocyclic compound, heterocyclic amine consists of a six-membered ring containing five methylene bridges (–CH2–) and one amine bridge (–NH–). It is a colorless ...
ring with a methyl group
In organic chemistry, a methyl group is an alkyl derived from methane, containing one carbon atom bonded to three hydrogen atoms, having chemical formula . In formulas, the group is often abbreviated as Me. This hydrocarbon group occurs in ma ...
substitution at position 2 and a long hydrophobic
In chemistry, hydrophobicity is the physical property of a molecule that is seemingly repelled from a mass of water (known as a hydrophobe). In contrast, hydrophiles are attracted to water.
Hydrophobic molecules tend to be nonpolar and, ...
chain at position 6. They are typically oily at room temperature, water-insoluble, and present an absorbance peak at 232 nanometers. Fire ant venom contains other chemically related piperidines which make purification of solenopsin from ants difficult. Therefore, solenopsin and related compounds have been the target of organic synthesis
Organic synthesis is a special branch of chemical synthesis and is concerned with the intentional construction of organic compounds. Organic molecules are often more complex than inorganic compounds, and their synthesis has developed into one o ...
from which pure compounds can be produced for individual study. Originally synthesized in 1993, several groups have designed novel and creative methods of synthesizing enantiopure solenopsin and other alkaloidal components of ant venom.
Total synthesis
Thetotal synthesis
Total synthesis is the complete chemical synthesis of a complex molecule, often a natural product, from simple, commercially-available precursors. It usually refers to a process not involving the aid of biological processes, which distinguishes i ...
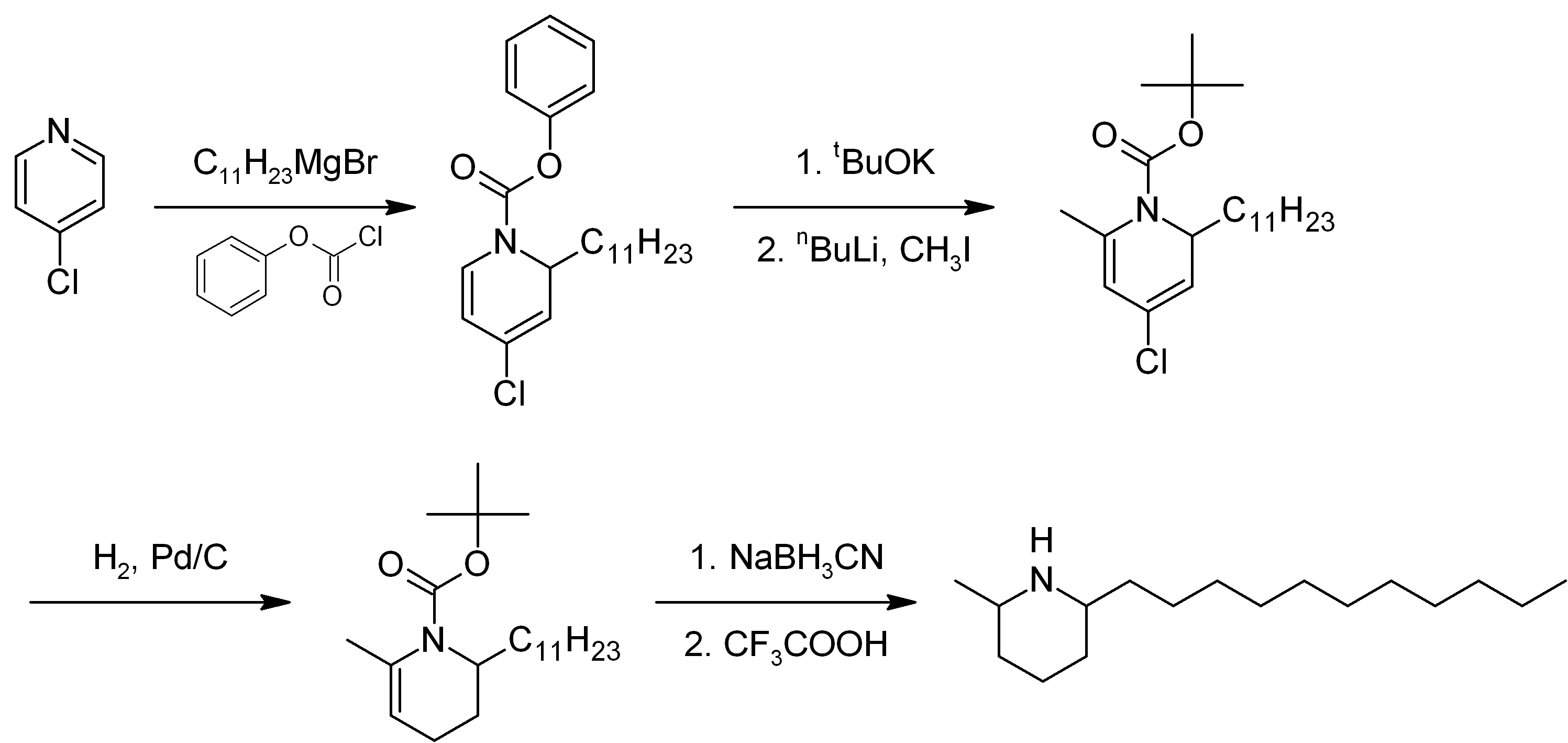
of solenopsin has been described by several methods. A proposed method of synthesis(Figure 1) starts with alkylation of 4-chloropyridine with a Grignard reagent derived from 1-bromoundecane, followed by reaction with phenyl chloroformate to form 4-chloro-1-(phenoxycarbonyl)-2-''n''-undecyl-1,2-dihydropyridine. The phenylcarbamate is converted to the BOC protecting group, and then pyridine is methylated at the 6 position. The pyridine ring is then reduced to a tetrahydropyridine via catalytic hydrogenation
Hydrogenation is a chemical reaction between molecular hydrogen (H2) and another compound or element, usually in the presence of a catalyst such as nickel, palladium or platinum. The process is commonly employed to reduce or saturate org ...
with Pd/C and then further reduced with sodium cyanoborohydride to a piperidine ring. The BOC group is finally removed to yield solenopsin. A number of analogs
Analog or analogue may refer to:
Computing and electronics
* Analog signal, in which information is encoded in a continuous variable
** Analog device, an apparatus that operates on analog signals
*** Analog electronics, circuits which use analog ...
have been synthesized using modifications of this procedure.
A shorter method of synthesis stemming from commercially-available lutidine has been more recently proposed.
:
Biological activities
Solenopsins are described as toxic against vertebrates and invertebrates. For example, the compound known as isosolenopsin A has been demonstrated to have strong insecticidal effects which may play a central role in the biology offire ant
Fire ants are several species of ants in the genus ''Solenopsis'', which includes over 200 species. ''Solenopsis'' are stinging ants, and most of their common names reflect this, for example, ginger ants and tropical fire ants. Many of the nam ...
s.
In addition to its toxicity, solenopsis has a number of other biological activities. It inhibits angiogenesis ''in vitro
''In vitro'' (meaning in glass, or ''in the glass'') studies are performed with microorganisms, cells, or biological molecules outside their normal biological context. Colloquially called "test-tube experiments", these studies in biology and ...
'' via the phosphoinositide 3-kinase
Phosphoinositide 3-kinases (PI3Ks), also called phosphatidylinositol 3-kinases, are a family of enzymes involved in cellular functions such as cell growth, proliferation, differentiation, motility, survival and intracellular trafficking, which i ...
(PI3K) signaling pathway, inhibits neuronal nitric oxide synthase
Nitric oxide synthases () (NOSs) are a family of enzymes catalyzing the production of nitric oxide (NO) from L-arginine. NO is an important cellular signaling molecule. It helps modulate vascular tone, insulin secretion, airway tone, and per ...
(nNOS) in a manner that appears to be non-competitive with L-arginine, and inhibits quorum-sensing signaling in some bacteria. The biological activities of solenopsins have led researchers to propose a number of biotechnological and biomedical applications for these compounds. For instance, mentioned anti-bacterial and interference in quorum-sensing signalling apparently provide solenopsins with considerable anti-biofilm activity, which suggests the potential of analogs as new disinfectants and surface-conditioning agents. Also, solenopsins have been demonstrated to inhibit cell division and viability of Trypanosoma cruzi, the cause of Chagas disease, which suggests these alkaloids as potential chemotherapeutic drugs.
Solenopsin and analogs share structural and biological properties with the sphingolipid
Sphingolipids are a class of lipids containing a backbone of sphingoid bases, a set of aliphatic amino alcohols that includes sphingosine. They were discovered in brain extracts in the 1870s and were named after the mythological sphinx beca ...
ceramide
Ceramides are a family of waxy lipid molecules. A ceramide is composed of N-acetyl sphingosine and a fatty acid. Ceramides are found in high concentrations within the cell membrane
The cell membrane (also known as the plasma membrane (PM) o ...
, a major endogenous regulator of cell signaling
In biology, cell signaling (cell signalling in British English) or cell communication is the ability of a cell to receive, process, and transmit signals with its environment and with itself. Cell signaling is a fundamental property of all cellula ...
, inducing mitophagy Mitophagy is the selective degradation of mitochondria by autophagy. It often occurs to defective mitochondria following damage or stress. The process of mitophagy was first described over a hundred years ago by Margaret Reed Lewis and Warren Har ...
and anti-proliferative effects in different tumor cell lines.
Synthetic analogs
Analog or analogue may refer to:
Computing and electronics
* Analog signal, in which information is encoded in a continuous variable
** Analog device, an apparatus that operates on analog signals
*** Analog electronics, circuits which use analog ...
of solenopsin are being studied for the potential treatment of psoriasis.
References
Further reading
* {{cite journal , doi = 10.1039/NP9971400637 , title = Pyrrole, pyrrolidine pyridine, piperidine, azepine and tropane alkaloids , year = 1997 , author = O'Hagan, David , journal = Natural Product Reports , type = Review , volume = 14 , pages = 637 , issue = 6 Piperidine alkaloids Total synthesis Toxins