Six-vertex Model on:
[Wikipedia]
[Google]
[Amazon]
In statistical mechanics, the ice-type models or six-vertex models are a family of
 The energy of a state is understood to be a function of the configurations at each vertex. For square lattices, one assumes that the total energy is given by
:
for some constants , where here denotes the number of vertices with the th configuration from the above figure. The value is the energy associated with vertex configuration number .
One aims to calculate the partition function of an ice-type model, which is given by the formula
:
where the sum is taken over all states of the model, is the energy of the state, is the
The energy of a state is understood to be a function of the configurations at each vertex. For square lattices, one assumes that the total energy is given by
:
for some constants , where here denotes the number of vertices with the th configuration from the above figure. The value is the energy associated with vertex configuration number .
One aims to calculate the partition function of an ice-type model, which is given by the formula
:
where the sum is taken over all states of the model, is the energy of the state, is the
vertex model A vertex model is a type of statistical mechanics model in which the Boltzmann weights are associated with a vertex in the model (representing an atom or particle). This contrasts with a nearest-neighbour model, such as the Ising model, in which t ...
s for crystal lattice
In geometry and crystallography, a Bravais lattice, named after , is an infinite array of discrete points generated by a set of discrete translation operations described in three dimensional space by
: \mathbf = n_1 \mathbf_1 + n_2 \mathbf_2 + n ...
s with hydrogen bonds. The first such model was introduced by Linus Pauling
Linus Carl Pauling (; February 28, 1901August 19, 1994) was an American chemist, biochemist, chemical engineer, peace activist, author, and educator. He published more than 1,200 papers and books, of which about 850 dealt with scientific top ...
in 1935 to account for the residual entropy
Residual entropy is the difference in entropy between a non-equilibrium state and crystal state of a substance close to absolute zero. This term is used in condensed matter physics to describe the entropy at zero kelvin of a glass or plastic crys ...
of water ice.
Variants have been proposed as models of certain ferroelectric
Ferroelectricity is a characteristic of certain materials that have a spontaneous electric polarization that can be reversed by the application of an external electric field. All ferroelectrics are also piezoelectric and pyroelectric, with the ad ...
and antiferroelectric Antiferroelectricity is a physical property of certain materials. It is closely related to ferroelectricity; the relation between antiferroelectricity and ferroelectricity is analogous to the relation between antiferromagnetism and ferromagnetism ...
crystals.
In 1967, Elliott H. Lieb
Elliott Hershel Lieb (born July 31, 1932) is an American mathematical physicist and professor of mathematics and physics at Princeton University who specializes in statistical mechanics, condensed matter theory, and functional analysis.
Lieb ...
found the exact solution to a two-dimensional ice model known as "square ice".
The exact solution in three dimensions is only known for a special "frozen" state.
Description
An ice-type model is a lattice model defined on a lattice ofcoordination number
In chemistry, crystallography, and materials science, the coordination number, also called ligancy, of a central atom in a molecule or crystal is the number of atoms, molecules or ions bonded to it. The ion/molecule/atom surrounding the central i ...
4. That is, each vertex of the lattice is connected by an edge to four "nearest neighbours". A state of the model consists of an arrow on each edge of the lattice, such that the number of arrows pointing inwards at each vertex is 2. This restriction on the arrow configurations is known as the ice rule. In graph theoretic terms, the states are Eulerian orientations of an underlying 4-regular
The term regular can mean normal or in accordance with rules. It may refer to:
People
* Moses Regular (born 1971), America football player
Arts, entertainment, and media Music
* "Regular" (Badfinger song)
* Regular tunings of stringed instrum ...
undirected graph. The partition function also counts the number of nowhere-zero 3-flows.
For two-dimensional models, the lattice is taken to be the square lattice. For more realistic models, one can use a three-dimensional lattice appropriate to the material being considered; for example, the hexagonal ice lattice is used to analyse ice.
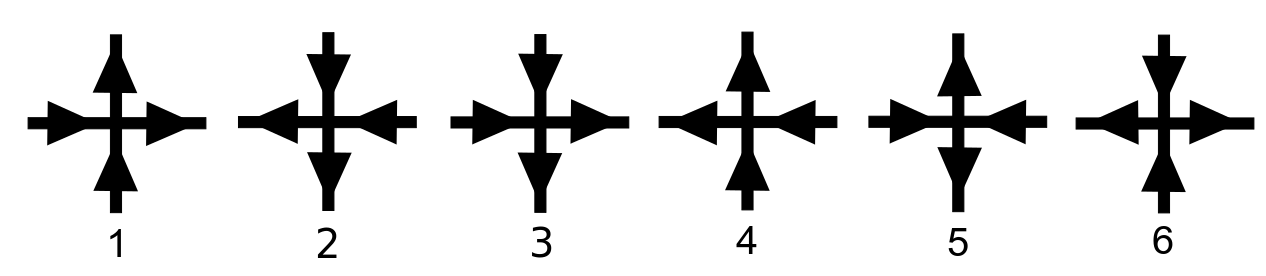
At any vertex, there are six configurations of the arrows which satisfy the ice rule (justifying the name "six-vertex model"). The valid configurations for the (two-dimensional) square lattice are the following:
: The energy of a state is understood to be a function of the configurations at each vertex. For square lattices, one assumes that the total energy is given by
:
for some constants , where here denotes the number of vertices with the th configuration from the above figure. The value is the energy associated with vertex configuration number .
One aims to calculate the partition function of an ice-type model, which is given by the formula
:
where the sum is taken over all states of the model, is the energy of the state, is the
The energy of a state is understood to be a function of the configurations at each vertex. For square lattices, one assumes that the total energy is given by
:
for some constants , where here denotes the number of vertices with the th configuration from the above figure. The value is the energy associated with vertex configuration number .
One aims to calculate the partition function of an ice-type model, which is given by the formula
:
where the sum is taken over all states of the model, is the energy of the state, is the Boltzmann constant
The Boltzmann constant ( or ) is the proportionality factor that relates the average relative kinetic energy of particles in a gas with the thermodynamic temperature of the gas. It occurs in the definitions of the kelvin and the gas consta ...
, and is the system's temperature.
Typically, one is interested in the thermodynamic limit
In statistical mechanics, the thermodynamic limit or macroscopic limit, of a system is the limit for a large number of particles (e.g., atoms or molecules) where the volume is taken to grow in proportion with the number of particles.S.J. Blunde ...
in which the number of vertices approaches infinity. In that case, one instead evaluates the free energy per vertex in the limit as , where is given by
:
Equivalently, one evaluates the partition function per vertex in the thermodynamic limit, where
:
The values and are related by
:
Physical justification
Several real crystals with hydrogen bonds satisfy the ice model, including ice and potassium dihydrogen phosphate (KDP). Indeed, such crystals motivated the study of ice-type models. In ice, each oxygen atom is connected by a bond to four other oxygens, and each bond contains one hydrogen atom between the terminal oxygens. The hydrogen occupies one of two symmetrically located positions, neither of which is in the middle of the bond. Pauling argued that the allowed configuration of hydrogen atoms is such that there are always exactly two hydrogens close to each oxygen, thus making the local environment imitate that of a water molecule, . Thus, if we take the oxygen atoms as the lattice vertices and the hydrogen bonds as the lattice edges, and if we draw an arrow on a bond which points to the side of the bond on which the hydrogen atom sits, then ice satisfies the ice model. Similar reasoning applies to show that KDP also satisfies the ice model. In recent years, ice-type models have been explored as descriptions of pyrochlorespin ice
A spin ice is a magnetic substance that does not have a single minimal-energy state. It has magnetic moments (i.e. "spin") as elementary degrees of freedom which are subject to frustrated interactions. By their nature, these interactions preven ...
and artificial spin ice systems, in which geometrical frustration In condensed matter physics, the term geometrical frustration (or in short: frustration) refers to a phenomenon where atoms tend to stick to non-trivial positions or where, on a regular crystal lattice, conflicting inter-atomic forces (each one fav ...
in the interactions between bistable magnetic moment
In electromagnetism, the magnetic moment is the magnetic strength and orientation of a magnet or other object that produces a magnetic field. Examples of objects that have magnetic moments include loops of electric current (such as electromagnets ...
s ("spins") leads to "ice-rule" spin configurations being favoured. Recently such analogies have been extended to explore the circumstances under which spin-ice systems may be accurately described by the Rys F-model.
Specific choices of vertex energies
On the square lattice, the energies associated with vertex configurations 1-6 determine the relative probabilities of states, and thus can influence the macroscopic behaviour of the system. The following are common choices for these vertex energies.The ice model
When modeling ice, one takes , as all permissible vertex configurations are understood to be equally likely. In this case, the partition function equals the total number of valid states. This model is known as the ice model (as opposed to an ''ice-type'' model).The KDP model of a ferroelectric
Slater argued that KDP could be represented by an ice-type model with energies : For this model (called the KDP model), the most likely state (the least-energy state) has all horizontal arrows pointing in the same direction, and likewise for all vertical arrows. Such a state is aferroelectric
Ferroelectricity is a characteristic of certain materials that have a spontaneous electric polarization that can be reversed by the application of an external electric field. All ferroelectrics are also piezoelectric and pyroelectric, with the ad ...
state, in which all hydrogen atoms have a preference for one fixed side of their bonds.
Rys ''F'' model of an antiferroelectric
The Rys model is obtained by setting : The least-energy state for this model is dominated by vertex configurations 5 and 6. For such a state, adjacent horizontal bonds necessarily have arrows in opposite directions and similarly for vertical bonds, so this state is anantiferroelectric Antiferroelectricity is a physical property of certain materials. It is closely related to ferroelectricity; the relation between antiferroelectricity and ferroelectricity is analogous to the relation between antiferromagnetism and ferromagnetism ...
state.
The zero field assumption
If there is no ambient electric field, then the total energy of a state should remain unchanged under a charge reversal, i.e. under flipping all arrows. Thus one may assume without loss of generality that : This assumption is known as the zero field assumption, and holds for the ice model, the KDP model, and the Rys ''F'' model.History
The ice rule was introduced by Linus Pauling in 1935 to account for theresidual entropy
Residual entropy is the difference in entropy between a non-equilibrium state and crystal state of a substance close to absolute zero. This term is used in condensed matter physics to describe the entropy at zero kelvin of a glass or plastic crys ...
of ice that had been measured by William F. Giauque and J. W. Stout. The residual entropy, , of ice is given by the formula
:
where is the Boltzmann constant
The Boltzmann constant ( or ) is the proportionality factor that relates the average relative kinetic energy of particles in a gas with the thermodynamic temperature of the gas. It occurs in the definitions of the kelvin and the gas consta ...
, is the number of oxygen atoms in the piece of ice, which is always taken to be large (the thermodynamic limit
In statistical mechanics, the thermodynamic limit or macroscopic limit, of a system is the limit for a large number of particles (e.g., atoms or molecules) where the volume is taken to grow in proportion with the number of particles.S.J. Blunde ...
) and is the number of configurations of the hydrogen atoms according to Pauling's ice rule. Without the ice rule we would have since the number of hydrogen atoms is and each hydrogen has two possible locations. Pauling estimated that the ice rule reduces this to , a number that would agree extremely well with the Giauque-Stout measurement of . It can be said that Pauling's calculation of for ice is one of the simplest, yet most accurate applications of statistical mechanics to real substances ever made. The question that remained was whether, given the model, Pauling's calculation of , which was very approximate, would be sustained by a rigorous calculation. This became a significant problem in combinatorics
Combinatorics is an area of mathematics primarily concerned with counting, both as a means and an end in obtaining results, and certain properties of finite structures. It is closely related to many other areas of mathematics and has many a ...
.
Both the three-dimensional and two-dimensional models were computed numerically by John F. Nagle in 1966 who found that in three-dimensions and in two-dimensions. Both are amazingly close to Pauling's rough calculation, 1.5.
In 1967, Lieb found the exact solution of three two-dimensional ice-type models: the ice model, the Rys model,
and the KDP model.
The solution for the ice model gave the exact value of in two-dimensions as
:
which is known as Lieb's square ice constant.
Later in 1967, Bill Sutherland generalised Lieb's solution of the three specific ice-type models to a general exact solution for square-lattice ice-type models satisfying the zero field assumption.
Still later in 1967, C. P. Yang
generalised Sutherland's solution to an exact solution for square-lattice ice-type models in a horizontal electric field.
In 1969, John Nagle derived the exact solution for a three-dimensional version of the KDP model, for a specific range of temperatures. For such temperatures, the model is "frozen" in the sense that (in the thermodynamic limit) the energy per vertex and entropy per vertex are both zero. This is the only known exact solution for a three-dimensional ice-type model.
Relation to eight-vertex model
Theeight-vertex model
In statistical mechanics, the eight-vertex model is a generalisation of the ice-type (six-vertex) models; it was discussed by Sutherland, and Fan & Wu, and solved by Baxter in the zero-field case.
Description
As with the ice-type models, the e ...
, which has also been exactly solved, is a generalisation of the (square-lattice) six-vertex model: to recover the six-vertex model from the eight-vertex model, set the energies for vertex configurations 7 and 8 to infinity. Six-vertex models have been solved in some cases for which the eight-vertex model has not; for example, Nagle's solution for the three-dimensional KDP model and Yang's solution of the six-vertex model in a horizontal field.
Boundary conditions
This ice model provide an important 'counterexample' in statistical mechanics: the bulk free energy in thethermodynamic limit
In statistical mechanics, the thermodynamic limit or macroscopic limit, of a system is the limit for a large number of particles (e.g., atoms or molecules) where the volume is taken to grow in proportion with the number of particles.S.J. Blunde ...
depends on boundary conditions. The model was analytically solved for periodic boundary conditions, anti-periodic, ferromagnetic and domain wall boundary conditions. The six vertex model with domain wall boundary conditions on a square lattice has specific significance in combinatorics, it helps to enumerate alternating sign matrices. In this case the partition function can be represented as a determinant of a matrix (whose dimension is equal to the size of the lattice), but in other cases the enumeration of does not come out in such a simple closed form.
Clearly, the largest is given by ''free'' boundary conditions (no constraint at all on the configurations on the boundary), but the same occurs, in the thermodynamic limit, for periodic boundary conditions,
as used originally to derive .
3-colorings of a lattice
The number of states of an ice type model on the internal edges of a finite simply connected union of squares of a lattice is equal to one third of the number of ways to 3-color the squares, with no two adjacent squares having the same color. This correspondence between states is due to Andrew Lenard and is given as follows. If a square has color ''i'' = 0, 1, or 2, then the arrow on the edge to an adjacent square goes left or right (according to an observer in the square) depending on whether the color in the adjacent square is ''i''+1 or ''i''−1 mod 3. There are 3 possible ways to color a fixed initial square, and once this initial color is chosen this gives a 1:1 correspondence between colorings and arrangements of arrows satisfying the ice-type condition.See also
*Eight-vertex model
In statistical mechanics, the eight-vertex model is a generalisation of the ice-type (six-vertex) models; it was discussed by Sutherland, and Fan & Wu, and solved by Baxter in the zero-field case.
Description
As with the ice-type models, the e ...
Notes
Further reading
* *{{Citation , author-link=Rodney J. Baxter , last1=Baxter , first1=Rodney J. , title=Exactly solved models in statistical mechanics , url=http://physics.anu.edu.au/theophys/_files/Exactly.pdf , publisher=Academic Press Inc. arcourt Brace Jovanovich Publishers, location=London , isbn=978-0-12-083180-7 , mr=690578 , year=1982 , access-date=2012-08-12 , archive-date=2021-04-14 , archive-url=https://web.archive.org/web/20210414063635/https://physics.anu.edu.au/theophys/_files/Exactly.pdf , url-status=dead Exactly solvable models Statistical mechanics Lattice models Ice