Sigmatropic reaction on:
[Wikipedia]
[Google]
[Amazon]
A sigmatropic reaction in
 A convenient means of determining the order of a given sigmatropic rearrangement is to number the atoms of the bond being broken as atom 1, and then count the atoms in each direction from the broken bond to the atoms that form the new σ-bond in the product, numbering consecutively. The numbers that correspond to the atoms forming the new bond are then separated by a comma and placed within brackets to create the sigmatropic reaction order descriptor.
:
A convenient means of determining the order of a given sigmatropic rearrangement is to number the atoms of the bond being broken as atom 1, and then count the atoms in each direction from the broken bond to the atoms that form the new σ-bond in the product, numbering consecutively. The numbers that correspond to the atoms forming the new bond are then separated by a comma and placed within brackets to create the sigmatropic reaction order descriptor.
: In the case of hydrogen atom migrations, a similar technique may be applied. When determining the order of a sigmatropic shift involving a hydrogen atom migration it is critical to count across all atoms involved in the reaction rather than only across the closest atoms. For example, the following hydrogen atom migration is of order ,5 attained by counting counterclockwise through the π system, rather than the ,3order designation through the ring CH2 group that would mistakenly result if counted clockwise.
:
In the case of hydrogen atom migrations, a similar technique may be applied. When determining the order of a sigmatropic shift involving a hydrogen atom migration it is critical to count across all atoms involved in the reaction rather than only across the closest atoms. For example, the following hydrogen atom migration is of order ,5 attained by counting counterclockwise through the π system, rather than the ,3order designation through the ring CH2 group that would mistakenly result if counted clockwise.
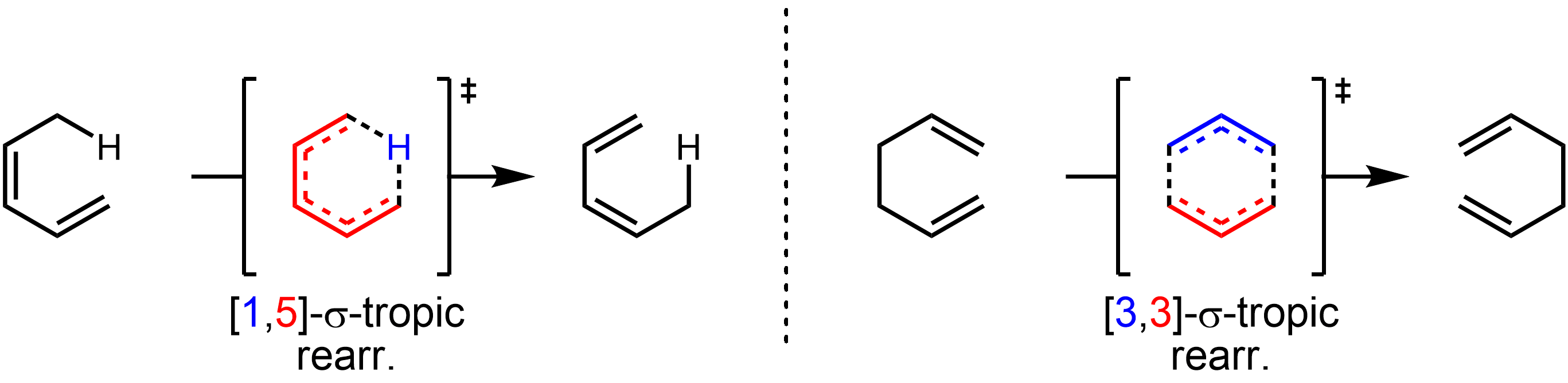
: As a general approach, one can simply draw the transition state of the reaction. For a sigmatropic reaction, the transition state will consist of two fragments, joined together by the forming and breaking σ-bonds. The sigmatropic reaction is named as a 'i'',''j''sigmatropic rearrangement (''i'' ≤ ''j'') if these two fragments consist of ''i'' and ''j'' atoms. This is illustrated below, with the relevant fragments shown in color.
:
As a general approach, one can simply draw the transition state of the reaction. For a sigmatropic reaction, the transition state will consist of two fragments, joined together by the forming and breaking σ-bonds. The sigmatropic reaction is named as a 'i'',''j''sigmatropic rearrangement (''i'' ≤ ''j'') if these two fragments consist of ''i'' and ''j'' atoms. This is illustrated below, with the relevant fragments shown in color.
:
 In cases of
In cases of 

 Photoirradiation would require an antarafacial shift of hydrogen. Although rare, there are examples where antarafacial shifts are favored:
:
Photoirradiation would require an antarafacial shift of hydrogen. Although rare, there are examples where antarafacial shifts are favored:
: In contrast to hydrogen ,5shifts, there have never been any observed ,5alkyl shifts in an open-chain compound. Several studies have, however, been done to determine rate preferences for ,5alkyl shifts in cyclic systems:
In contrast to hydrogen ,5shifts, there have never been any observed ,5alkyl shifts in an open-chain compound. Several studies have, however, been done to determine rate preferences for ,5alkyl shifts in cyclic systems:  This same mechanistic process is seen below, without the final electrocyclic ring-closing reaction, in the interconversion of lumisterol to vitamin D2.
This same mechanistic process is seen below, without the final electrocyclic ring-closing reaction, in the interconversion of lumisterol to vitamin D2.
 Bicyclic nonatrienes also undergo ,7shifts in a so-called walk rearrangement, which is the shift of
Bicyclic nonatrienes also undergo ,7shifts in a so-called walk rearrangement, which is the shift of 

 When both the ortho positions on the
When both the ortho positions on the 



 The choice of acid catalyst is very important. Brønsted acids such as
The choice of acid catalyst is very important. Brønsted acids such as


 Proceeding through a ,5shift, the walk rearrangement of norcaradienes is expected to proceed suprafacially with a retention of stereochemistry. Experimental observations, however, show that the 1,5-shifts of norcaradienes proceed antarafacially. Theoretical calculations found the ,5shift to be a
Proceeding through a ,5shift, the walk rearrangement of norcaradienes is expected to proceed suprafacially with a retention of stereochemistry. Experimental observations, however, show that the 1,5-shifts of norcaradienes proceed antarafacially. Theoretical calculations found the ,5shift to be a
organic chemistry
Organic chemistry is a subdiscipline within chemistry involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms.Clayden, J.; ...
is a pericyclic reaction
In organic chemistry, a pericyclic reaction is the type of organic reaction wherein the transition state of the molecule has a cyclic geometry, the reaction progresses in a concerted fashion, and the bond orbitals involved in the reaction overlap ...
wherein the net result is one σ-bond is changed to another σ-bond in an uncatalyzed intramolecular reaction Intramolecular in chemistry describes a process or characteristic limited within the structure of a single molecule, a property or phenomenon limited to the extent of a single molecule.
Examples
* intramolecular hydride transfer (transfer of a hy ...
. The name ''sigmatropic'' is the result of a compounding
In the field of pharmacy, compounding (performed in compounding pharmacies) is preparation of a custom formulation of a medication to fit a unique need of a patient that cannot be met with commercially available products. This may be done for me ...
of the long-established sigma designation from single carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent
In chemistry, the valence (US spelling) or valency (British spelling) of an element is the measure of its combining capacity with o ...
–carbon bonds and the Greek word ''tropos'', meaning turn. In this type of rearrangement reaction
In organic chemistry, a rearrangement reaction is a broad class of organic reactions where the carbon skeleton of a molecule is rearranged to give a structural isomer of the original molecule. Often a substituent moves from one atom to another ...
, a substituent
A substituent is one or a group of atoms that replaces (one or more) atoms, thereby becoming a moiety in the resultant (new) molecule. (In organic chemistry and biochemistry, the terms ''substituent'' and ''functional group'', as well as ''side ...
moves from one part of a π-bonded system to another part in an intramolecular reaction with simultaneous rearrangement of the π system. True sigmatropic reactions are usually uncatalyzed, although Lewis acid
A Lewis acid (named for the American physical chemist Gilbert N. Lewis) is a chemical species that contains an empty orbital which is capable of accepting an electron pair from a Lewis base to form a Lewis adduct. A Lewis base, then, is any sp ...
catalysis
Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recyc ...
is possible. Sigmatropic reactions often have transition-metal catalysts that form intermediates in analogous reactions. The most well-known of the sigmatropic rearrangements are the ,3Cope rearrangement
The Cope rearrangement is an extensively studied organic reaction involving the ,3sigmatropic rearrangement of 1,5-dienes. It was developed by Arthur C. Cope and Elizabeth Hardy. For example, 3-methyl-hexa-1,5-diene heated to 300 °C yield ...
, Claisen rearrangement
The Claisen rearrangement is a powerful carbon–carbon bond-forming chemical reaction discovered by Rainer Ludwig Claisen. The heating of an allyl vinyl ether will initiate a ,3sigmatropic rearrangement to give a γ,δ-unsaturated carbonyl, d ...
, Carroll rearrangement, and the Fischer indole synthesis
The Fischer indole synthesis is a chemical reaction that produces the aromatic heterocycle indole from a (substituted) phenylhydrazine and an aldehyde or ketone under acidic conditions. The reaction was discovered in 1883 by Emil Fischer. Today ...
.
Overview of sigmatropic shifts
Woodward–Hoffman sigmatropic shift nomenclature
Sigmatropic rearrangements are concisely described by an order term ,j'', which is defined as themigration
Migration, migratory, or migrate may refer to: Human migration
* Human migration, physical movement by humans from one region to another
** International migration, when peoples cross state boundaries and stay in the host state for some minimum le ...
of a σ-bond adjacent to one or more π systems to a new position (i−1) and (j−1) atom
Every atom is composed of a nucleus and one or more electrons bound to the nucleus. The nucleus is made of one or more protons and a number of neutrons. Only the most common variety of hydrogen has no neutrons.
Every solid, liquid, gas, and ...
s removed from the original location of the σ-bond. When the sum of i and j is an even number, this is an indication of the involvement of a neutral, all C atom chain. An odd number is an indication of the involvement of a charged C atom or of a heteroatom lone pair replacing a CC double bond. Thus, ,5'' and ,3'' shifts become ,4'' and ,3'' shifts with heteroatoms, while preserving symmetry considerations. Hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, an ...
s are omitted in the third example for clarity.
: A convenient means of determining the order of a given sigmatropic rearrangement is to number the atoms of the bond being broken as atom 1, and then count the atoms in each direction from the broken bond to the atoms that form the new σ-bond in the product, numbering consecutively. The numbers that correspond to the atoms forming the new bond are then separated by a comma and placed within brackets to create the sigmatropic reaction order descriptor.
:
A convenient means of determining the order of a given sigmatropic rearrangement is to number the atoms of the bond being broken as atom 1, and then count the atoms in each direction from the broken bond to the atoms that form the new σ-bond in the product, numbering consecutively. The numbers that correspond to the atoms forming the new bond are then separated by a comma and placed within brackets to create the sigmatropic reaction order descriptor.
: In the case of hydrogen atom migrations, a similar technique may be applied. When determining the order of a sigmatropic shift involving a hydrogen atom migration it is critical to count across all atoms involved in the reaction rather than only across the closest atoms. For example, the following hydrogen atom migration is of order ,5 attained by counting counterclockwise through the π system, rather than the ,3order designation through the ring CH2 group that would mistakenly result if counted clockwise.
:
In the case of hydrogen atom migrations, a similar technique may be applied. When determining the order of a sigmatropic shift involving a hydrogen atom migration it is critical to count across all atoms involved in the reaction rather than only across the closest atoms. For example, the following hydrogen atom migration is of order ,5 attained by counting counterclockwise through the π system, rather than the ,3order designation through the ring CH2 group that would mistakenly result if counted clockwise.
: As a general approach, one can simply draw the transition state of the reaction. For a sigmatropic reaction, the transition state will consist of two fragments, joined together by the forming and breaking σ-bonds. The sigmatropic reaction is named as a 'i'',''j''sigmatropic rearrangement (''i'' ≤ ''j'') if these two fragments consist of ''i'' and ''j'' atoms. This is illustrated below, with the relevant fragments shown in color.
:
As a general approach, one can simply draw the transition state of the reaction. For a sigmatropic reaction, the transition state will consist of two fragments, joined together by the forming and breaking σ-bonds. The sigmatropic reaction is named as a 'i'',''j''sigmatropic rearrangement (''i'' ≤ ''j'') if these two fragments consist of ''i'' and ''j'' atoms. This is illustrated below, with the relevant fragments shown in color.
:
Suprafacial and antarafacial shifts
In principle, all sigmatropic shifts can occur with either a retention or inversion of the geometry of the migrating group, depending upon whether the original bonding lobe of the migrating atom or its other lobe is used to form the new bond. : In cases of
In cases of stereochemical
Stereochemistry, a subdiscipline of chemistry, involves the study of the relative spatial arrangement of atoms that form the structure of molecules and their manipulation. The study of stereochemistry focuses on the relationships between stereois ...
retention, the migrating group translates without rotation into the bonding position, while in the case of stereochemical inversion the migrating group both rotates and translates to reach its bonded conformation.
However, another stereochemical transition effect equally capable of producing inversion or retention products is whether the migrating group remains on the original face of the π system after rebonding or instead transfers to the opposite face of the π system. If the migrating group remains on the same face of the π system, the shift is known as suprafacial Antarafacial ( Woodward-Hoffmann symbol a) and suprafacial (s) are two topological concepts in organic chemistry describing the relationship between two simultaneous chemical bond making and/or bond breaking processes in or around a reaction center ...
, while if the migrating group transfers to the opposite face is called an antarafacial shift, Woodward, R.B.; Hoffmann, R. The Conservation of Orbital Symmetry. Verlag Chemie Academic Press. 2004. . which are impossible for transformations that occur within small- or medium-sized rings.
:
Classes of sigmatropic rearrangements
,3shifts
Thermal hydride shifts
In athermal
A thermal column (or thermal) is a rising mass of buoyant air, a convective current in the atmosphere, that transfers heat energy vertically. Thermals are created by the uneven heating of Earth's surface from solar radiation, and are an example ...
,3hydride
In chemistry, a hydride is formally the anion of hydrogen( H−). The term is applied loosely. At one extreme, all compounds containing covalently bound H atoms are called hydrides: water (H2O) is a hydride of oxygen, ammonia is a hydride of ...
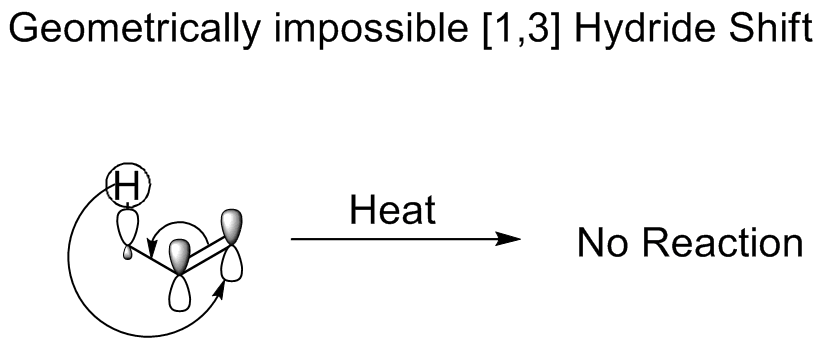
shift, a hydride moves three atoms. The Woodward–Hoffmann rules The Woodward–Hoffmann rules (or the pericyclic selection rules), devised by Robert Burns Woodward and Roald Hoffmann, are a set of rules used to rationalize or predict certain aspects of the stereochemistry and activation energy of pericyclic reac ...
dictate that it would proceed in an antarafacial shift. Although such a shift is symmetry allowed, the Mobius
Moebius, Möbius or Mobius may refer to:
People
* August Ferdinand Möbius (1790–1868), German mathematician and astronomer
* Theodor Möbius (1821–1890), German philologist
* Karl Möbius (1825–1908), German zoologist and ecologist
* Pau ...
topology
In mathematics, topology (from the Greek language, Greek words , and ) is concerned with the properties of a mathematical object, geometric object that are preserved under Continuous function, continuous Deformation theory, deformations, such ...
required in the transition state
In chemistry, the transition state of a chemical reaction is a particular configuration along the reaction coordinate. It is defined as the state corresponding to the highest potential energy along this reaction coordinate. It is often marked wi ...
prohibits such a shift because it is geometrically impossible, which accounts for the fact that enol
In organic chemistry, alkenols (shortened to enols) are a type of reactive structure or intermediate in organic chemistry that is represented as an alkene ( olefin) with a hydroxyl group attached to one end of the alkene double bond (). The te ...
s do not isomerize without an acid
In computer science, ACID ( atomicity, consistency, isolation, durability) is a set of properties of database transactions intended to guarantee data validity despite errors, power failures, and other mishaps. In the context of databases, a sequ ...
or base catalyst
Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recyc ...
.Miller, Bernard. ''Advanced Organic Chemistry''. 2nd Ed. Upper Saddle River: Pearson Prentice Hall. 2004.
:
Thermal alkyl shifts
Thermal alkyl ,3shifts, similar to ,3hydride shifts, must proceed antarafacially. Here the geometry of the transition state is prohibitive, but analkyl group
In organic chemistry, an alkyl group is an alkane missing one hydrogen.
The term ''alkyl'' is intentionally unspecific to include many possible substitutions.
An acyclic alkyl has the general formula of . A cycloalkyl is derived from a cycloalk ...
, due to the nature of its orbitals, can invert its geometry, form a new bond with the back lobe of its sp3 orbital, and therefore proceed via a suprafacial shift. These reactions are still not common in open-chain compound
In chemistry, an open-chain compound (also spelled as open chain compound) or acyclic compound (Greek prefix "α", ''without'' and "κύκλος", ''cycle'') is a compound with a linear structure, rather than a cyclic one.
An open-chain compound ha ...
s because of the highly ordered nature of the transition state, which is more readily achieved in cyclic
Cycle, cycles, or cyclic may refer to:
Anthropology and social sciences
* Cyclic history, a theory of history
* Cyclical theory, a theory of American political history associated with Arthur Schlesinger, Sr.
* Social cycle, various cycles in s ...
molecules.
:
Photochemical ,3shifts
Photochemical ,3shifts should proceed through suprafacial shifts; however, most are non-concerted because they proceed through atriplet state
In quantum mechanics, a triplet is a quantum state of a system with a spin of quantum number =1, such that there are three allowed values of the spin component, = −1, 0, and +1.
Spin, in the context of quantum mechanics, is not a mechanical ...
(i.e., have a diradical
In chemistry, a diradical is a molecular species with two electrons occupying molecular orbitals (MOs) which are degenerate. The term "diradical" is mainly used to describe organic compounds, where most diradicals are extremely reactive and in ...
mechanism, to which the Woodward-Hoffmann rules do not apply).
,5shifts
A ,5shift involves the shift of 1substituent
A substituent is one or a group of atoms that replaces (one or more) atoms, thereby becoming a moiety in the resultant (new) molecule. (In organic chemistry and biochemistry, the terms ''substituent'' and ''functional group'', as well as ''side ...
(hydride, alkyl, or aryl
In organic chemistry, an aryl is any functional group or substituent derived from an aromatic ring, usually an aromatic hydrocarbon, such as phenyl and naphthyl. "Aryl" is used for the sake of abbreviation or generalization, and "Ar" is used as ...
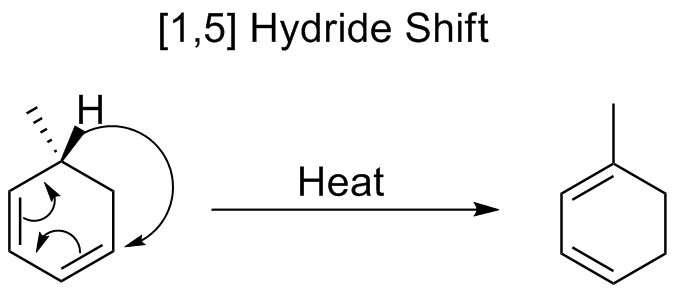
) down 5 atoms of a π system. Hydrogen has been shown to shift in both cyclic and open-chain compounds at temperatures at or above 200 ˚C. These reactions are predicted to proceed suprafacially, via a Hückel-topology transition state.
: Photoirradiation would require an antarafacial shift of hydrogen. Although rare, there are examples where antarafacial shifts are favored:
:
Photoirradiation would require an antarafacial shift of hydrogen. Although rare, there are examples where antarafacial shifts are favored:
: In contrast to hydrogen ,5shifts, there have never been any observed ,5alkyl shifts in an open-chain compound. Several studies have, however, been done to determine rate preferences for ,5alkyl shifts in cyclic systems:
In contrast to hydrogen ,5shifts, there have never been any observed ,5alkyl shifts in an open-chain compound. Several studies have, however, been done to determine rate preferences for ,5alkyl shifts in cyclic systems: carbonyl
In organic chemistry, a carbonyl group is a functional group composed of a carbon atom double-bonded to an oxygen atom: C=O. It is common to several classes of organic compounds, as part of many larger functional groups. A compound containing a ...
and carboxyl
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is or , with R referring to the alkyl, alkenyl, aryl, or other group. Carboxylic ...
> hydride> phenyl
In organic chemistry, the phenyl group, or phenyl ring, is a cyclic group of atoms with the formula C6 H5, and is often represented by the symbol Ph. Phenyl group is closely related to benzene and can be viewed as a benzene ring, minus a hydrogen ...
and vinyl
Vinyl may refer to:
Chemistry
* Polyvinyl chloride (PVC), a particular vinyl polymer
* Vinyl cation, a type of carbocation
* Vinyl group, a broad class of organic molecules in chemistry
* Vinyl polymer, a group of polymers derived from vinyl m ...
>> alkyl.
Alkyl groups undergo ,5shifts very poorly, usually requiring high temperatures, however, for cyclohexadiene Cyclohexadiene may refer to:
* 1,3-Cyclohexadiene,
* 1,4-Cyclohexadiene,
See also
* Benzene or its theoretical isomer ''1,3,5-Cyclohexatriene''
* Cyclohexene
Cyclohexene is a hydrocarbon with the formula C6H10. This cycloalkene is a colorle ...
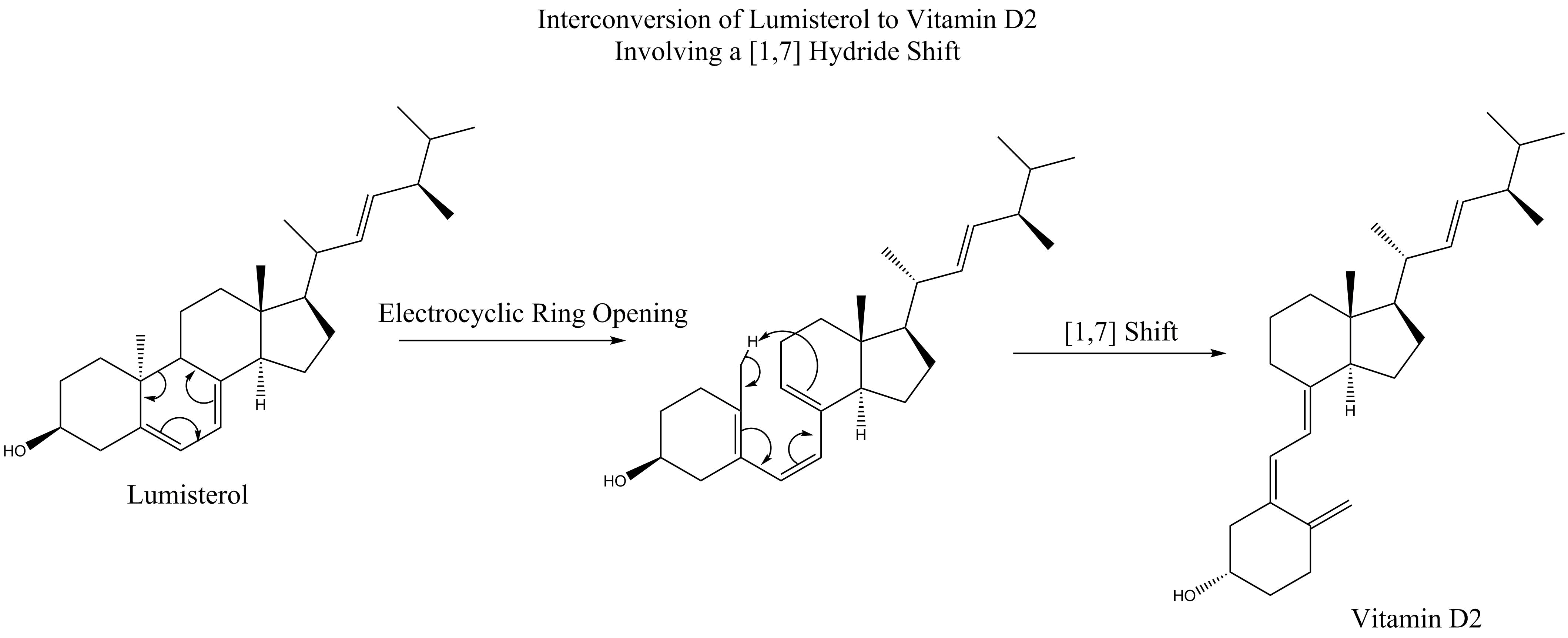
, the temperature for alkyl shifts isn't much higher than that for carbonyls, the best migratory group. A study showed that this is because alkyl shifts on cyclohexadienes proceed through a different mechanism. First the ring opens, followed by a ,7shift, and then the ring reforms electrocyclically:
: This same mechanistic process is seen below, without the final electrocyclic ring-closing reaction, in the interconversion of lumisterol to vitamin D2.
This same mechanistic process is seen below, without the final electrocyclic ring-closing reaction, in the interconversion of lumisterol to vitamin D2.
,7shifts
,7sigmatropic shifts are predicted by the Woodward–Hoffmann rules to proceed in an antarafacial fashion, via a Mobius topology transition state. An antarafacial ,7'' shift is observed in the conversion oflumisterol
Lumisterol is a compound that is part of the vitamin D family of steroid compounds. It is the (9β,10α) stereoisomer of ergosterol
Ergosterol (ergosta-5,7,22-trien-3β-ol) is a sterol found in cell membranes of fungi and protozoa, serving man ...
to vitamin D2, where following an electrocyclic ring opening to previtamin D2, a methyl
In organic chemistry, a methyl group is an alkyl derived from methane, containing one carbon atom bonded to three hydrogen atoms, having chemical formula . In formulas, the group is often abbreviated as Me. This hydrocarbon group occurs in many ...
hydrogen shifts.
: Bicyclic nonatrienes also undergo ,7shifts in a so-called walk rearrangement, which is the shift of
Bicyclic nonatrienes also undergo ,7shifts in a so-called walk rearrangement, which is the shift of divalent
In chemistry, the valence (US spelling) or valency (British spelling) of an element is the measure of its combining capacity with other atoms when it forms chemical compounds or molecules.
Description
The combining capacity, or affinity of an ...
group, as part of a three-membered ring, in a bicyclic molecule
In chemistry, a bicyclic molecule () is a molecule that features two joined rings. Bicyclic structures occur widely, for example in many biologically important molecules like α-thujene and camphor. A bicyclic compound can be carbocyclic (all ...
.
:
,3shifts
,3sigmatropic shifts are well studied sigmatropic rearrangements. The Woodward–Hoffman rules predict that these six-electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no kn ...
reactions would proceed suprafacially, via a Hückel topology transition state.
Claisen rearrangement
Discovered in 1912 byRainer Ludwig Claisen
Rainer Ludwig Claisen (; 14 January 1851 – 5 January 1930) was a German chemist best known for his work with condensations of carbonyls and sigmatropic rearrangements. He was born in Cologne as the son of a jurist and studied chemistry at the u ...
, the Claisen rearrangement is the first recorded example of a ,3sigmatropic rearrangement. This rearrangement is a useful carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent
In chemistry, the valence (US spelling) or valency (British spelling) of an element is the measure of its combining capacity with o ...
-carbon bond
Bond or bonds may refer to:
Common meanings
* Bond (finance), a type of debt security
* Bail bond, a commercial third-party guarantor of surety bonds in the United States
* Chemical bond, the attraction of atoms, ions or molecules to form chemica ...
-forming reaction
Reaction may refer to a process or to a response to an action, event, or exposure:
Physics and chemistry
*Chemical reaction
*Nuclear reaction
*Reaction (physics), as defined by Newton's third law
*Chain reaction (disambiguation).
Biology and me ...
. An example of Claisen rearrangement is the ,3rearrangement of an allyl
In organic chemistry, an allyl group is a substituent with the structural formula , where R is the rest of the molecule. It consists of a methylene bridge () attached to a vinyl group (). The name is derived from the scientific name for garlic, ...
vinyl ether
In organic chemistry, ethers are a class of compounds that contain an ether group—an oxygen atom connected to two alkyl or aryl groups. They have the general formula , where R and R′ represent the alkyl or aryl groups. Ethers can again be c ...
, which upon heating yields a γ,δ-unsaturated carbonyl. The formation of a carbonyl group makes this reaction, unlike other sigmatropic rearrangements, inherently irreversible.
:
=Aromatic Claisen rearrangement
= The ortho-Claisen rearrangement involves the ,3shift of an allyl phenyl ether to an intermediate which quicklytautomer
Tautomers () are structural isomers (constitutional isomers) of chemical compounds that readily interconvert.
The chemical reaction interconverting the two is called tautomerization. This conversion commonly results from the relocation of a hydr ...
izes to an ortho-substituted phenol
Phenol (also called carbolic acid) is an aromatic organic compound with the molecular formula . It is a white crystalline solid that is volatile. The molecule consists of a phenyl group () bonded to a hydroxy group (). Mildly acidic, it req ...
.
: When both the ortho positions on the
When both the ortho positions on the benzene
Benzene is an organic chemical compound with the molecular formula C6H6. The benzene molecule is composed of six carbon atoms joined in a planar ring with one hydrogen atom attached to each. Because it contains only carbon and hydrogen atoms, ...
ring are blocked, a second ,3rearrangement will occur. This para-Claisen rearrangement ends with the tautomerization to a tri-substituted phenol.
:
Cope rearrangement
The Cope rearrangement is an extensively studiedorganic reaction
Organic reactions are chemical reactions involving organic compounds. The basic organic chemistry reaction types are addition reactions, elimination reactions, substitution reactions, pericyclic reactions, rearrangement reactions, Mechanistic Organ ...
involving the ,3sigmatropic rearrangement of 1,5-dienes. It was developed by Arthur C. Cope
Arthur C. Cope (June 27, 1909 – June 4, 1966) was an American organic chemist and member of the United States National Academy of Sciences. He is credited with the development of several important chemical reactions which bear his name includin ...
. For example, 3,4-dimethyl-1,5-hexadiene heated to 300 °C yields 2,6-octadiene.
:
=Oxy-Cope rearrangement
= In the oxy-Cope rearrangement, ahydroxyl
In chemistry, a hydroxy or hydroxyl group is a functional group with the chemical formula and composed of one oxygen atom covalently bonded to one hydrogen atom. In organic chemistry, alcohols and carboxylic acids contain one or more hydroxy ...
group is added at C3 forming an enal or enone after keto-enol tautomerism
In organic chemistry, alkenols (shortened to enols) are a type of reactive structure or intermediate in organic chemistry that is represented as an alkene ( olefin) with a hydroxyl group attached to one end of the alkene double bond (). The te ...
of the intermediate enol:
:
Carroll rearrangement
The Carroll rearrangement is arearrangement reaction
In organic chemistry, a rearrangement reaction is a broad class of organic reactions where the carbon skeleton of a molecule is rearranged to give a structural isomer of the original molecule. Often a substituent moves from one atom to another ...
in organic chemistry
Organic chemistry is a subdiscipline within chemistry involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms.Clayden, J.; ...
and involves the transformation of a β- keto allyl
In organic chemistry, an allyl group is a substituent with the structural formula , where R is the rest of the molecule. It consists of a methylene bridge () attached to a vinyl group (). The name is derived from the scientific name for garlic, ...
ester
In chemistry, an ester is a compound derived from an oxoacid (organic or inorganic) in which at least one hydroxyl group () is replaced by an alkoxy group (), as in the substitution reaction of a carboxylic acid and an alcohol. Glycerides ar ...
into a α-allyl-β-ketocarboxylic acid. This organic reaction is accompanied by decarboxylation
Decarboxylation is a chemical reaction that removes a carboxyl group and releases carbon dioxide (CO2). Usually, decarboxylation refers to a reaction of carboxylic acids, removing a carbon atom from a carbon chain. The reverse process, which is t ...
and the final product is a γ,δ-allylketone. The Carroll rearrangement is an adaptation of the Claisen rearrangement
The Claisen rearrangement is a powerful carbon–carbon bond-forming chemical reaction discovered by Rainer Ludwig Claisen. The heating of an allyl vinyl ether will initiate a ,3sigmatropic rearrangement to give a γ,δ-unsaturated carbonyl, d ...
and effectively a decarboxylative allylation.
:
Fischer indole synthesis
The Fischer indole synthesis is achemical reaction
A chemical reaction is a process that leads to the IUPAC nomenclature for organic transformations, chemical transformation of one set of chemical substances to another. Classically, chemical reactions encompass changes that only involve the pos ...
that produces the aromatic heterocycle
A heterocyclic compound or ring structure is a cyclic compound that has atoms of at least two different chemical element, elements as members of its ring(s). Heterocyclic chemistry is the branch of organic chemistry dealing with the synthesis ...
indole
Indole is an aromatic heterocyclic organic compound with the formula C8 H7 N. It has a bicyclic structure, consisting of a six-membered benzene ring fused to a five-membered pyrrole ring. Indole is widely distributed in the natural environmen ...
from a (substituted) phenylhydrazine
Phenylhydrazine is the chemical compound with the formula . It is often abbreviated as . It is also found in edible mushrooms.
Properties
Phenylhydrazine forms monoclinic prisms that melt to an oil around room temperature which may turn yellow ...
and an aldehyde
In organic chemistry, an aldehyde () is an organic compound containing a functional group with the structure . The functional group itself (without the "R" side chain) can be referred to as an aldehyde but can also be classified as a formyl grou ...
or ketone
In organic chemistry, a ketone is a functional group with the structure R–C(=O)–R', where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group –C(=O)– (which contains a carbon-oxygen double bo ...
under acid
In computer science, ACID ( atomicity, consistency, isolation, durability) is a set of properties of database transactions intended to guarantee data validity despite errors, power failures, and other mishaps. In the context of databases, a sequ ...
ic conditions. The reaction was discovered in 1883 by Hermann Emil Fischer
Hermann Emil Louis Fischer (; 9 October 1852 – 15 July 1919) was a German chemist and 1902 recipient of the Nobel Prize in Chemistry. He discovered the Fischer esterification. He also developed the Fischer projection, a symbolic way of dra ...
.
: The choice of acid catalyst is very important. Brønsted acids such as
The choice of acid catalyst is very important. Brønsted acids such as HCl HCL may refer to:
Science and medicine
* Hairy cell leukemia, an uncommon and slowly progressing B cell leukemia
* Harvard Cyclotron Laboratory, from 1961 to 2002, a proton accelerator used for research and development
* Hollow-cathode lamp, a spe ...
, H2SO4, polyphosphoric acid
A phosphoric acid, in the general sense, is a phosphorus oxoacid in which each phosphorus (P) atom is in the oxidation state +5, and is bonded to four oxygen (O) atoms, one of them through a double bond, arranged as the corners of a tetrahedron. ...
and p-toluenesulfonic acid
''p''-Toluenesulfonic acid (PTSA or ''p''TsOH) or tosylic acid (TsOH) is an organic compound with the formula CH3 C6H4 SO3H. It is a white extremely hygroscopic solid that is soluble in water, alcohols, and other polar organic solvents. The CH3C ...
have been used successfully. Lewis acid
A Lewis acid (named for the American physical chemist Gilbert N. Lewis) is a chemical species that contains an empty orbital which is capable of accepting an electron pair from a Lewis base to form a Lewis adduct. A Lewis base, then, is any sp ...
s such as boron trifluoride
Boron trifluoride is the inorganic compound with the formula BF3. This pungent, colourless, and toxic gas forms white fumes in moist air. It is a useful Lewis acid and a versatile building block for other boron compounds.
Structure and bondin ...
, zinc chloride
Zinc chloride is the name of inorganic chemical compounds with the formula ZnCl2 and its hydrates. Zinc chlorides, of which nine crystalline forms are known, are colorless or white, and are highly soluble in water. This salt is hygroscopic and ev ...
, iron(III) chloride
Iron(III) chloride is the inorganic compound with the formula . Also called ferric chloride, it is a common compound of iron in the +3 oxidation state. The anhydrous compound is a crystalline solid with a melting point of 307.6 °C. The col ...
, and aluminium chloride
Aluminium chloride, also known as aluminium trichloride, is an inorganic compound with the formula . It forms hexahydrate with the formula , containing six water molecules of hydration. Both are colourless crystals, but samples are often contam ...
are also useful catalysts.
Several reviews have been published.
,5Shifts
Similar to ,3shifts, the Woodward-Hoffman rules predict that ,5sigmatropic shifts would proceed suprafacially, Hückel topology transition state. These reactions are rarer than ,3sigmatropic shifts, but this is mainly a function of the fact that molecules that can undergo ,5shifts are rarer than molecules that can undergo ,3shifts. :
,3shifts
An example of a 2,3-sigmatropic rearrangement is the2,3-Wittig rearrangement
The ,3Wittig rearrangement is the transformation of an allylic ether into a homoallylic Alcohol (chemistry), alcohol via a concerted, pericyclic reaction, pericyclic process. Because the reaction is concerted, it exhibits a high degree of ster ...
:
:
Walk rearrangements
The migration of a divalent group, such as O, S, N–R, or C–R2, which is part of a three-membered ring in a bicyclic molecule, is commonly referred to as a walk rearrangement. This can be formally characterized according to the Woodward-Hofmann rules as being a (1, n) sigmatropic shift. An example of such a rearrangement is the shift of substituents on tropilidenes (1,3,5-cycloheptatrienes). When heated, the pi-system goes through an electrocyclic ring closing to form bicycle ,1,0eptadiene (norcaradiene). Thereafter follows a ,5alkyl shift and an electrocyclic ring opening. : Proceeding through a ,5shift, the walk rearrangement of norcaradienes is expected to proceed suprafacially with a retention of stereochemistry. Experimental observations, however, show that the 1,5-shifts of norcaradienes proceed antarafacially. Theoretical calculations found the ,5shift to be a
Proceeding through a ,5shift, the walk rearrangement of norcaradienes is expected to proceed suprafacially with a retention of stereochemistry. Experimental observations, however, show that the 1,5-shifts of norcaradienes proceed antarafacially. Theoretical calculations found the ,5shift to be a diradical
In chemistry, a diradical is a molecular species with two electrons occupying molecular orbitals (MOs) which are degenerate. The term "diradical" is mainly used to describe organic compounds, where most diradicals are extremely reactive and in ...
process, but without involving any diradical minima
In mathematical analysis, the maxima and minima (the respective plurals of maximum and minimum) of a function, known collectively as extrema (the plural of extremum), are the largest and smallest value of the function, either within a given ran ...
on the potential energy surface
A potential energy surface (PES) describes the energy of a system, especially a collection of atoms, in terms of certain parameters, normally the positions of the atoms. The surface might define the energy as a function of one or more coordinates; ...
.Kless, A.; Nendel, M.; Wilsey, S.; Houk, K. N. ''J. Am. Chem. Soc.
The ''Journal of the American Chemical Society'' is a weekly peer-reviewed scientific journal that was established in 1879 by the American Chemical Society. The journal has absorbed two other publications in its history, the ''Journal of Analytic ...
'', 1999, ''121'', 4524.
See also
* 2,3-sigmatropic rearrangement *NIH shift An NIH shift is a chemical rearrangement where a hydrogen atom on an aromatic ring undergoes an intramolecular migration primarily during a hydroxylation reaction. This process is also known as a 1,2-hydride shift. These shifts are often studied ...
*Frontier molecular orbital theory In chemistry, frontier molecular orbital theory is an application of MO theory describing HOMO/LUMO interactions.
History
In 1952, Kenichi Fukui published a paper in the ''Journal of Chemical Physics'' titled "A molecular theory of reactivity i ...
References
{{Reflist Reaction mechanisms Rearrangement reactions