Polymorphism (materials Science) on:
[Wikipedia]
[Google]
[Amazon]
In crystallography, polymorphism is the phenomenon where a compound or element can crystallize into more than one crystal structure.
The preceding definition has evolved over many years and is still under discussion today. Discussion of the defining characteristics of polymorphism involves distinguishing among types of transitions and structural changes occurring in polymorphism versus those in other phenomena.
ART) summarized in the Journal of Materials Science 2011 Nevertheless, there are known systems, such as metacetamol, where only narrow cooling rate favors obtaining metastable form II. Polymorphs have disparate stabilities. Some convert rapidly at room (or any) temperature. Most polymorphs of organic molecules only differ by a few kJ/mol in lattice energy. Approximately 50% of known polymorph pairs differ by less than 2 kJ/mol and stability differences of more than 10 kJ/mol are rare. Polymorph stability may change upon temperature or pressure. Importantly, structural and thermodynamic stability are different. Thermodynamic stability may be studied using experimental or computational methods. Polymorphism is affected by the details of crystallisation. The solvent in all respects affects the nature of the polymorph, including concentration, other components of the solvent, i.e., species that inhibiting or promote certain growth patterns. A decisive factor is often the temperature of the solvent from which crystallisation is carried out. Metastable polymorphs are not always reproducibly obtained, leading to cases of "
 Now, suppose there are two kinds of crystals, with different energies and , and if they have the same shape as in Figure 2, then the two curves intersect at some . Then the system has three phases:
* . Crystals tend to dissolve. Amorphous phase.
* . Crystals tend to grow as form 1.
* . Crystals tend to grow as form 2.
If the crystal is grown slowly, it could be kinetically stuck in form 1.
Now, suppose there are two kinds of crystals, with different energies and , and if they have the same shape as in Figure 2, then the two curves intersect at some . Then the system has three phases:
* . Crystals tend to dissolve. Amorphous phase.
* . Crystals tend to grow as form 1.
* . Crystals tend to grow as form 2.
If the crystal is grown slowly, it could be kinetically stuck in form 1.
"Small Molecule Crystallization"
(
"SiC and Polytpism"
{{Authority control Mineralogy Gemology Crystallography
Overview
Phase transitions (phase changes) that help describe polymorphism include polymorphic transitions as well as melting and vaporization transitions. According to IUPAC, a polymorphic transition is "A reversible transition of a solid crystalline phase at a certain temperature and pressure (the inversion point) to another phase of the same chemical composition with a different crystal structure." Additionally,Walter McCrone
Walter Cox McCrone (1916-2002) was an American chemist who was considered a leading expert in microscopy. To the general public, however, he was best known for his work on the Shroud of Turin, the Vinland map, and forensic science.
Biograph ...
described the phases in polymorphic matter as "different in crystal structure but identical in the liquid or vapor states." McCrone also defines a polymorph as “a crystalline phase of a given compound resulting from the possibility of at least two different arrangements of the molecules of that compound in the solid state.” These defining facts imply that polymorphism involves changes in physical properties but cannot include chemical change. Some early definitions do not make this distinction.
Eliminating chemical change from those changes permissible during a polymorphic transition delineates polymorphism. For example, isomerization can often lead to polymorphic transitions. However, tautomerism (dynamic isomerization) leads to chemical change, not polymorphism. As well, allotropy of elements and polymorphism have been linked historically. However, allotropes of an element are not always polymorphs. A common example is the allotropes of carbon
Carbon is capable of forming many allotropy, allotropes (structurally different forms of the same element) due to its Valence (chemistry), valency. Well-known forms of carbon include diamond and graphite. In recent decades, many more allotrope ...
, which include graphite, diamond, and londsdaleite. While all three forms are allotropes, graphite is not a polymorph of diamond and londsdaleite. Isomerization and allotropy are only two of the phenomena linked to polymorphism. For additional information about identifying polymorphism and distinguishing it from other phenomena, see the review by Brog et al.
It is also useful to note that materials with two polymorphic phases can be called dimorphic, those with three polymorphic phases, trimorphic, etc.
Polymorphism is of practical relevance to pharmaceuticals, agrochemical
An agrochemical or agrichemical, a contraction of ''agricultural chemical'', is a chemical product used in industrial agriculture. Agrichemical refers to biocides ( pesticides including insecticides, herbicides, fungicides and nematicides) an ...
s, pigments, dyestuffs, food
Food is any substance consumed by an organism for nutritional support. Food is usually of plant, animal, or fungal origin, and contains essential nutrients, such as carbohydrates, fats, proteins, vitamins, or minerals. The substance is inge ...
s, and explosive
An explosive (or explosive material) is a reactive substance that contains a great amount of potential energy that can produce an explosion if released suddenly, usually accompanied by the production of light, heat, sound, and pressure. An expl ...
s.
Detection
Experimental methods
Early records of the discovery of polymorphism credit Eilhard Mitscherlich andJöns Jacob Berzelius
Baron Jöns Jacob Berzelius (; by himself and his contemporaries named only Jacob Berzelius, 20 August 1779 – 7 August 1848) was a Swedish chemist. Berzelius is considered, along with Robert Boyle, John Dalton, and Antoine Lavoisier, to be on ...
for their studies of phosphates and arsenates in the early 1800s. The studies involved measuring the interfacial angles of the crystals to show that chemically identical salts could have two different forms. Mitscherlich originally called this discovery isomorphism. The measurement of crystal density was also used by Wilhelm Ostwald and expressed in Ostwald's Ratio.
The development of the microscope enhanced observations of polymorphism and aided Moritz Ludwig Frankenheim
Moritz Ludwig Frankenheim (29 June 1801 – 14 January 1869) was a Germans, German physicist, geographer, and crystallography, crystallographer.
Life and education
Moritz Ludwig Frankenheim was born in 1801 in Braunschweig, Brunswick. His fam ...
’s studies in the 1830s. He was able to demonstrate methods to induce crystal phase changes and formally summarized his findings on the nature of polymorphism. Soon after, the more sophisticated polarized light microscope came into use, and it provided better visualization of crystalline phases allowing crystallographers to distinguish between different polymorphs. The hot stage was invented and fitted to a polarized light microscope by Otto Lehmann Otto Lehmann may refer to:
* Otto Lehmann (physicist) (1855–1922), German physicist
* Otto Lehmann (movie producer) Otto Lehmann (22 January 1889 in Berlin – 28 April 1968 in Munich ) was a German movie producer.
Career
Lehmann studied to ...
in about 1877. This invention helped crystallographers determine melting points and observe polymorphic transitions.
While the use of hot stage microscopes continued throughout the 1900s, thermal methods also became commonly used to observe the heat flow that occurs during phase changes such as melting and polymorphic transitions. One such technique, differential scanning calorimetry
Differential scanning calorimetry (DSC) is a thermoanalytical technique in which the difference in the amount of heat required to increase the temperature of a sample and reference is measured as a function of temperature. Both the sample and ref ...
(DSC), continues to be used for determining the enthalpy of polymorphic transitions.
In the 20th century, X-ray crystallography became commonly used for studying the crystal structure of polymorphs. Both single crystal x-ray diffraction and powder x-ray diffraction techniques are used to obtain measurements of the crystal unit cell. Each polymorph of a compound has a unique crystal structure. As a result, different polymorphs will produce different x-ray diffraction patterns.
Vibrational spectroscopic methods came into use for investigating polymorphism in the second half of the twentieth century and have become more commonly used as optical, computer, and semiconductor technologies improved. These techniques include infrared (IR) spectroscopy, terahertz spectroscopy Terahertz spectroscopy detects and controls properties of matter with electromagnetic fields that are in the frequency range between a few hundred gigahertz and several terahertz (abbreviated as THz). In many-body systems, several of the relevant ...
and Raman spectroscopy
Raman spectroscopy () (named after Indian physicist C. V. Raman) is a spectroscopic technique typically used to determine vibrational modes of molecules, although rotational and other low-frequency modes of systems may also be observed. Raman sp ...
. Mid-frequency IR and Raman spectroscopies are sensitive to changes in hydrogen bond
In chemistry, a hydrogen bond (or H-bond) is a primarily electrostatic force of attraction between a hydrogen (H) atom which is covalently bound to a more electronegative "donor" atom or group (Dn), and another electronegative atom bearing a ...
ing patterns. Such changes can subsequently be related to structural differences. Additionally, terahertz and low frequency Raman spectroscopies reveal vibrational modes resulting from intermolecular interactions in crystalline solids. Again, these vibrational modes are related to crystal structure and can be used to uncover differences in 3-dimensional structure among polymorphs.
Computational methods
Computational chemistry
Computational chemistry is a branch of chemistry that uses computer simulation to assist in solving chemical problems. It uses methods of theoretical chemistry, incorporated into computer programs, to calculate the structures and properties of m ...
may be used in combination with vibrational spectroscopy techniques to understand the origins of vibrations within crystals. The combination of techniques provides detailed information about crystal structures, similar to what can be achieved with x-ray crystallography. In addition to using computational methods for enhancing the understanding of spectroscopic data, the latest development in identifying polymorphism in crystals is the field of crystal structure prediction. This technique uses computational chemistry to model the formation of crystals and predict the existence of specific polymorphs of a compound before they have been observed experimentally by scientists.
Examples
Many compounds exhibit polymorphism. It has been claimed that "every compound has different polymorphic forms, and that, in general, the number of forms known for a given compound is proportional to the time and money spent in research on that compound."Organic compounds
Benzamide
The phenomenon was discovered in 1832 byFriedrich Wöhler
Friedrich Wöhler () FRS(For) HonFRSE (31 July 180023 September 1882) was a German chemist known for his work in inorganic chemistry, being the first to isolate the chemical elements beryllium and yttrium in pure metallic form. He was the firs ...
and Justus von Liebig
Justus Freiherr von Liebig (12 May 1803 – 20 April 1873) was a German scientist who made major contributions to agricultural and biological chemistry, and is considered one of the principal founders of organic chemistry. As a professor at t ...
. They observed that the silky needles of freshly crystallized benzamide slowly converted to rhombic crystals. Present-day analysis identifies three polymorphs for benzamide: the least stable one, formed by flash cooling is the orthorhombic
In crystallography, the orthorhombic crystal system is one of the 7 crystal systems. Orthorhombic lattices result from stretching a cubic lattice along two of its orthogonal pairs by two different factors, resulting in a rectangular prism with a r ...
form II. This type is followed by the monoclinic form III (observed by Wöhler/Liebig). The most stable form is monoclinic form I. The hydrogen bonding mechanisms are the same for all three phases; however, they differ strongly in their pi-pi interactions.
Maleic acid
In 2006 a new polymorph of maleic acid was discovered, 124 years after the first crystal form was studied. Maleic acid is manufactured on an industrial scale in the chemical industry. It forms salt found in medicine. The new crystal type is produced when a co-crystal of caffeine and maleic acid (2:1) is dissolved inchloroform
Chloroform, or trichloromethane, is an organic compound with chemical formula, formula Carbon, CHydrogen, HChlorine, Cl3 and a common organic solvent. It is a colorless, strong-smelling, dense liquid produced on a large scale as a precursor to ...
and when the solvent is allowed to evaporate slowly. Whereas form I has monoclinic space group ''P''21/''c'', the new form has space group ''Pc''. Both polymorphs consist of sheets of molecules connected through hydrogen bond
In chemistry, a hydrogen bond (or H-bond) is a primarily electrostatic force of attraction between a hydrogen (H) atom which is covalently bound to a more electronegative "donor" atom or group (Dn), and another electronegative atom bearing a ...
ing of the carboxylic acid
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is or , with R referring to the alkyl, alkenyl, aryl, or other group. Carboxylic ...
groups: in form I, the sheets alternate with respect of the net dipole moment, while in form II, the sheets are oriented in the same direction.
1,3,5-Trinitrobenzene
After 125 years of study, 1,3,5-trinitrobenzene yielded a second polymorph. The usual form has the space group ''Pbca'', but in 2004, a second polymorph was obtained in the space group ''Pca''21 when the compound was crystallised in the presence of an additive, trisindane. This experiment shows that additives can induce the appearance of polymorphic forms.Other organic compounds
Acridine has been obtained as eight polymorphs and aripiprazole has nine. The record for the largest number of well-characterised polymorphs is held by a compound known as ROY. Glycine crystallizes as both monoclinic and hexagonal crystals. Polymorphism in organic compounds is often the result of conformational polymorphism.Inorganic matter
Elements
Elements including metals may exhibit polymorphism. Allotropy is the term used when describing elements having different forms and is used commonly in the field of metallurgy. Some (but not all) allotropes are also polymorphs. For example iron has three allotropes that are also polymorphs. Alpha-iron, which exists at room temperature, has a bcc form. Above 910 degrees gamma-iron exists, which has a fcc form. Above 1390 degrees delta-iron exists with a bcc form. Another metallic example is tin, which has two allotropes that are also polymorphs. At room temperature, beta-tin exists as a white tetragonal form. When cooled below 13.2 degrees, alpha-tin forms which is gray in color and has a cubic diamond form. A classic example of a nonmetal that exhibits polymorphism is carbon. Carbon has many allotropes, including graphite, diamond, and londsdaleite. However, these are not all polymorphs of each other. Graphite is not a polymorph of diamond and londsdaleite, since it is chemically distinct, having sp2 hybridized bonding. Diamond, and londsdaleite are chemically identical, both having sp3 hybridized bonding, and they differ only in their crystal structures, making them polymorphs. Additionally, graphite has two polymorphs, a hexagonal (alpha) form and a rhombohedral (beta) form.Binary metal oxides
Polymorphism in binary metal oxides has attracted much attention because these materials are of significant economic value. One set of famous examples have the composition SiO2, which form many polymorphs. Important ones include: α-quartz, β-quartz, tridymite, cristobalite,moganite
Moganite is an oxide mineral with the chemical formula Si O2 (silicon dioxide) that was discovered in 1976. It was initially described as a new form of silica from specimens found in the Barranco de Medio Almud, in the municipality of Mogán on ...
, coesite, and stishovite
Stishovite is an extremely hard, dense tetragonal form (Polymorphism (materials science), polymorph) of silicon dioxide. It is very rare on the Earth's surface; however, it may be a predominant form of silicon dioxide in the Earth, especially in ...
.
"Polymorphism in nanocrystalline binary metal oxides", S. Sood, P.Gouma, Nanomaterials and Energy, 2(NME2), 1-15(2013).
Other inorganic compounds
A classical example of polymorphism is the pair of mineralscalcite
Calcite is a Carbonate minerals, carbonate mineral and the most stable Polymorphism (materials science), polymorph of calcium carbonate (CaCO3). It is a very common mineral, particularly as a component of limestone. Calcite defines hardness 3 on ...
, which is rhombohedral, and aragonite, which is orthorhombic. Both are forms of calcium carbonate. A third form of calcium carbonate is vaterite, which is hexagonal and relatively unstable.
β-HgS precipitates as a black solid when Hg(II) salts are treated with H2S. With gentle heating of the slurry, the black polymorph converts to the red form.
Factors affecting polymorphism
According to Ostwald's rule, usually less stable polymorphs crystallize before the stable form. The concept hinges on the idea that unstable polymorphs more closely resemble the state in solution, and thus are kinetically advantaged. The founding case of fibrous vs rhombic benzamide illustrates the case. Another example is provided by two polymorphs of titanium dioxide.Anatase to Rutile TransformationART) summarized in the Journal of Materials Science 2011 Nevertheless, there are known systems, such as metacetamol, where only narrow cooling rate favors obtaining metastable form II. Polymorphs have disparate stabilities. Some convert rapidly at room (or any) temperature. Most polymorphs of organic molecules only differ by a few kJ/mol in lattice energy. Approximately 50% of known polymorph pairs differ by less than 2 kJ/mol and stability differences of more than 10 kJ/mol are rare. Polymorph stability may change upon temperature or pressure. Importantly, structural and thermodynamic stability are different. Thermodynamic stability may be studied using experimental or computational methods. Polymorphism is affected by the details of crystallisation. The solvent in all respects affects the nature of the polymorph, including concentration, other components of the solvent, i.e., species that inhibiting or promote certain growth patterns. A decisive factor is often the temperature of the solvent from which crystallisation is carried out. Metastable polymorphs are not always reproducibly obtained, leading to cases of "
disappearing polymorphs
In materials science, disappearing polymorphs (or perverse polymorphism) describes a phenomenon in which a seemingly stable crystal structure is suddenly unable to be produced, instead transforming into a polymorph, or differing crystal structur ...
", with usually negative implications on law and business.Crystal Engineering: The Design and Application of Functional Solids, Volume 539, Kenneth Richard Seddon, Michael Zaworotk 1999
In pharmaceuticals
Legal aspects
Drugs
A drug is any chemical substance that causes a change in an organism's physiology or psychology when consumed. Drugs are typically distinguished from food and substances that provide nutritional support. Consumption of drugs can be via inhalat ...
receive regulatory approval
Regulation is the management of complex systems according to a set of rules and trends. In systems theory, these types of rules exist in various fields of biology and society, but the term has slightly different meanings according to context. For ...
and are granted patents for only a single polymorph.
In a classic patent dispute, the GlaxoSmithKline
GSK plc, formerly GlaxoSmithKline plc, is a British multinational pharmaceutical and biotechnology company with global headquarters in London, England. Established in 2000 by a merger of Glaxo Wellcome and SmithKline Beecham. GSK is the ten ...
defended its patent for the Type II polymorph of the active ingredient in Zantac against competitors while that of the Type I polymorph had already expired.
Polymorphism in drugs can also have direct medical implications since dissolution rates depend on the polymorph. Polymorphic purity of drug samples can be checked using techniques such as powder X-ray diffraction, IR/Raman
spectroscopy, and utilizing the differences in their optical properties in some cases.
Case studies
The known cases up to 2015 are discussed in a review article by Bučar, Lancaster, and Bernstein. Dibenzoxazepines Multidisciplinary studies involving experimental and computational approaches were applied to pharmaceutical molecules to facilitate the comparison of their solid-state structures. Specifically, this study has focused on exploring how changes in molecular structure affect the molecular conformation, packing motifs, interactions in the resultant crystal lattices and the extent of solid-state diversity of these compounds. The results highlight the value of crystal structure prediction studies and PIXEL calculations in the interpretation of the observed solid-state behaviour and quantifying the intermolecular interactions in the packed structures and identifying the key stabilising interactions. An experimental screen yielded 4 physical forms for clozapine as compared to 60 distinct physical forms for olanzapine. The experimental screening results of clozapine are consistent with its crystal energy landscape which confirms that no alternate packing arrangement is thermodynamically competitive to the experimentally obtained structure. Whilst in case of olanzapine, crystal energy landscape highlights that the extensive experimental screening has probably not found all possible polymorphs of olanzapine, and further solid form diversity could be targeted with a better understanding of the role of kinetics in its crystallisation. CSP studies were able to offer an explanation for the absence of the centrosymmetric dimer in anhydrous clozapine. PIXEL calculations on all the crystal structures of clozapine revealed that similar to olanzapine, the intermolecular interaction energy in each structure is also dominated by the Ed. Despite the molecular structure similarity between amoxapine and loxapine (molecules in group 2), the crystal packing observed in polymorphs of loxa differs significantly from the amoxapine. A combined experimental and computational study demonstrated that the methyl group in loxapine has a significant influence in increasing the range of accessible solid forms and favouring various alternate packing arrangements. CSP studies have again helped in explaining the observed solid-state diversity of loxapine and amoxapine. PIXEL calculations showed that in absence of strong H-bonds, weak H-bonds such as C–H...O, C–H...N and dispersion interactions play a key role in stabilising the crystal lattice of both the molecules. Efficient crystal packing of amoxapine seems to be contributing towards its monomorphic behaviour as compared to the comparatively less efficient packing of loxapine molecules in both polymorphs. The combination of experimental and computational approaches has provided a deeper understanding of the factors influencing the solid-state structure and diversity in these compounds. Hirshfeld surfaces using Crystal Explorer represent another way of exploring packing modes and intermolecular interactions in molecular crystals. The influence of changes in the small substituents on shape and electron distribution can also be investigated by mapping the total electron density on the electrostatic potential for molecules in the gas phase. This allows straightforward visualisation and comparison of overall shape, electron-rich and electron-deficient regions within molecules. The shape of these molecules can be further investigated to study its influence on diverse solid-state diversity. Posaconazole The original formulations of posaconazole on the market licensed as Noxafil were formulated utilising form I of posaconazole. The discovery of polymorphs of posaconazole increased rapidly and resulted in much research in crystallography of posaconazole. A methanol solvate and a 1,4-dioxane co-crystal were added to the Cambridge Structural Database (CSD).Ritonavir
The antiviral drug ritonavir exists as two polymorphs, which differ greatly in efficacy. Such issues were solved by reformulating the medicine into gelcaps and tablets, rather than the original capsules.Aspirin
There was only one proven polymorph Form I of aspirin, though the existence of another polymorph was debated since the 1960s, and one report from 1981 reported that when crystallized in the presence of aspirin ''anhydride'', the diffractogram of aspirin has weak additional peaks. Though at the time it was dismissed as mere impurity, it was, in retrospect, Form II aspirin. Form II was reported in 2005, found after attempted co-crystallization of aspirin and levetiracetam from hotacetonitrile
Acetonitrile, often abbreviated MeCN (methyl cyanide), is the chemical compound with the formula and structure . This colourless liquid is the simplest organic nitrile (hydrogen cyanide is a simpler nitrile, but the cyanide anion is not clas ...
.
In form I, pairs of aspirin molecules form centrosymmetric dimers through the acetyl
In organic chemistry, acetyl is a functional group with the chemical formula and the structure . It is sometimes represented by the symbol Ac (not to be confused with the element actinium). In IUPAC nomenclature, acetyl is called ethanoyl, ...
groups with the (acidic) methyl
In organic chemistry, a methyl group is an alkyl derived from methane, containing one carbon atom bonded to three hydrogen atoms, having chemical formula . In formulas, the group is often abbreviated as Me. This hydrocarbon group occurs in many ...
proton to carbonyl hydrogen bond
In chemistry, a hydrogen bond (or H-bond) is a primarily electrostatic force of attraction between a hydrogen (H) atom which is covalently bound to a more electronegative "donor" atom or group (Dn), and another electronegative atom bearing a ...
s. In form II, each aspirin molecule forms the same hydrogen bonds, but with two neighbouring molecules instead of one. With respect to the hydrogen bonds formed by the carboxylic acid
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is or , with R referring to the alkyl, alkenyl, aryl, or other group. Carboxylic ...
groups, both polymorphs form identical dimer structures. The aspirin polymorphs contain identical 2-dimensional sections and are therefore more precisely described as polytypes.
Pure Form II aspirin could be prepared by seeding the batch with aspirin anhydrate in 15% weight.
Paracetamol
Paracetamol
Paracetamol, also known as acetaminophen, is a medication used to treat fever and mild to moderate pain. Common brand names include Tylenol and Panadol.
At a standard dose, paracetamol only slightly decreases body temperature; it is inferior ...
powder has poor compression properties, which poses difficulty in making tablets. A second polymorph was found with more suitable compressive properties.
Cortisone acetate
Cortisone acetate exists in at least five different polymorphs, four of which are unstable in water and change to a stable form.Carbamazepine
Carbamazepine, estrogen, paroxetine, andchloramphenicol
Chloramphenicol is an antibiotic useful for the treatment of a number of bacterial infections. This includes use as an eye ointment to treat conjunctivitis. By mouth or by injection into a vein, it is used to treat meningitis, plague, cholera, a ...
also show polymorphism.
Pyrazinamide
Pyrazinamide has at least 4 polymorphs. All of them transforms to stable α form at room temperature upon storage or mechanical treatment. Recent studies prove that α form is thermodynamically stable at room temperature.Polytypism
Polytypes are a special case of polymorphs, where multiple close-packed crystal structures differ in one dimension only. Polytypes have identical close-packed planes, but differ in the stacking sequence in the third dimension perpendicular to these planes. Silicon carbide (SiC) has more than 170 known polytypes, although most are rare. All the polytypes of SiC have virtually the same density and Gibbs free energy. The most common SiC polytypes are shown in Table 1. Table 1: Some polytypes of SiC. A second group of materials with different polytypes are thetransition metal dichalcogenides :image:Cadmium sulfide.jpg, 220px, Cadmium sulfide, a prototypical metal chalcogenide, is used as a yellow pigment.
A chalcogenide is a chemical compound consisting of at least one chalcogen anion and at least one more electropositive element. Altho ...
, layered materials such as molybdenum disulfide (MoS2). For these materials the polytypes have more distinct effects on material properties, e.g. for MoS2, the 1T polytype is metallic in character, while the 2H form is more semiconducting.
Another example is tantalum disulfide, where the common 1T as well as 2H polytypes occur, but also more complex 'mixed coordination' types such as 4Hb and 6R, where the trigonal prismatic and the octahedral geometry layers are mixed. Here, the 1T polytype exhibits a charge density wave, with distinct influence on the conductivity as a function of temperature, while the 2H polytype exhibits superconductivity
Superconductivity is a set of physical properties observed in certain materials where electrical resistance vanishes and magnetic flux fields are expelled from the material. Any material exhibiting these properties is a superconductor. Unlike ...
.
ZnS and CdI2 are also polytypical. It has been suggested that this type of polymorphism is due to kinetics where screw dislocations rapidly reproduce partly disordered sequences in a periodic fashion.
Theory
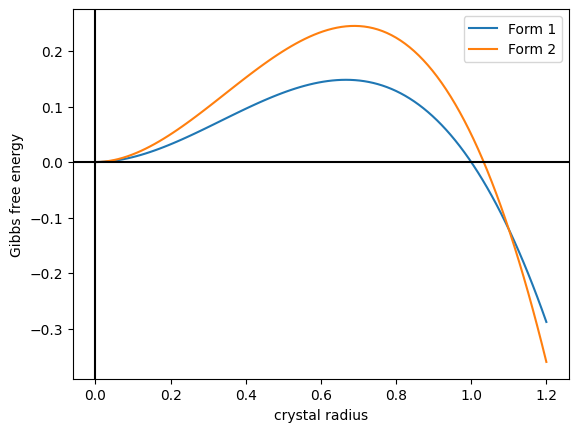
]In terms of thermodynamics, two types of polymorphic behaviour are recognized. For a monotropic system, plots of the free energies of the various polymorphs against temperature do not cross before all polymorphs melt. As a result, any transition from one polymorph to another below the melting point will be irreversible. For an enantiotropic system, a plot of the free energy against temperature shows a crossing point before the various melting points. It may also be possible to convert interchangeably between the two polymorphs by heating or cooling, or through physical contact with a lower energy polymorph. A simple model of polymorphism is to model the Gibbs free energy of a ball-shaped crystal as . Here, the first term is the surface energy, and the second term is the volume energy. Both parameters . The function rises to a maximum before dropping, crossing zero at . In order to crystallize, a ball of crystal much overcome the energetic barrier to the part of the energy landscape. Now, suppose there are two kinds of crystals, with different energies and , and if they have the same shape as in Figure 2, then the two curves intersect at some . Then the system has three phases:
* . Crystals tend to dissolve. Amorphous phase.
* . Crystals tend to grow as form 1.
* . Crystals tend to grow as form 2.
If the crystal is grown slowly, it could be kinetically stuck in form 1.
Now, suppose there are two kinds of crystals, with different energies and , and if they have the same shape as in Figure 2, then the two curves intersect at some . Then the system has three phases:
* . Crystals tend to dissolve. Amorphous phase.
* . Crystals tend to grow as form 1.
* . Crystals tend to grow as form 2.
If the crystal is grown slowly, it could be kinetically stuck in form 1.
See also
* Allotropy * Isomorphism (crystallography) * Dimorphism (Wiktionary) * PolyamorphismReferences
External links
"Small Molecule Crystallization"
(
PDF
Portable Document Format (PDF), standardized as ISO 32000, is a file format developed by Adobe in 1992 to present documents, including text formatting and images, in a manner independent of application software, hardware, and operating systems. ...
) at Illinois Institute of Technology website
"SiC and Polytpism"
{{Authority control Mineralogy Gemology Crystallography