Organoaluminium Compounds on:
[Wikipedia]
[Google]
[Amazon]
 Organoaluminium chemistry is the study of compounds containing bonds between
Organoaluminium chemistry is the study of compounds containing bonds between

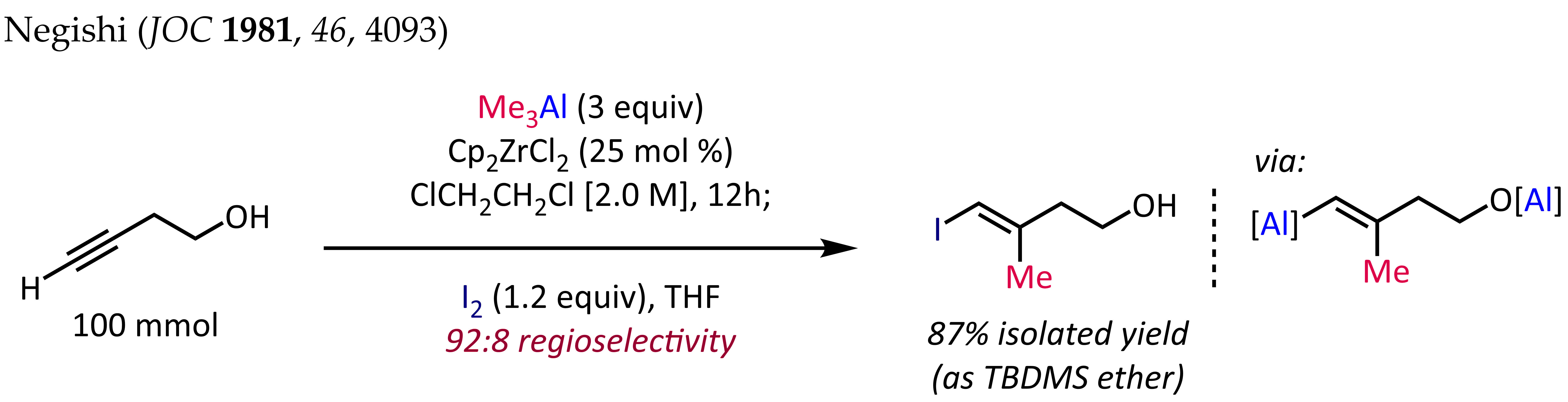 For terminal alkynes, the reaction generally proceeds with good regioselectivity (>90:10 rr) and complete ''syn'' selectivity, even in the presence of propargylic or homopropargylic heteroatom substituents. Unfortunately, extension of the zirconocene-catalyzed methylalumination to alkylalumination with higher alkyls results in lower yields and poor regioselectivities.
For terminal alkynes, the reaction generally proceeds with good regioselectivity (>90:10 rr) and complete ''syn'' selectivity, even in the presence of propargylic or homopropargylic heteroatom substituents. Unfortunately, extension of the zirconocene-catalyzed methylalumination to alkylalumination with higher alkyls results in lower yields and poor regioselectivities.
 Organoaluminium chemistry is the study of compounds containing bonds between
Organoaluminium chemistry is the study of compounds containing bonds between carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent
In chemistry, the valence (US spelling) or valency (British spelling) of an element is the measure of its combining capacity with o ...
and aluminium
Aluminium (aluminum in American and Canadian English) is a chemical element with the symbol Al and atomic number 13. Aluminium has a density lower than those of other common metals, at approximately one third that of steel. I ...
. It is one of the major themes within organometallic chemistry
Organometallic chemistry is the study of organometallic compounds, chemical compounds containing at least one chemical bond between a carbon atom of an organic molecule and a metal, including alkali, alkaline earth, and transition metals, and so ...
. Illustrative organoaluminium compounds are the dimer trimethylaluminium
Trimethylaluminium is one of the simplest examples of an organoaluminium compound. Despite its name it has the formula Al2( CH3)6 (abbreviated as Al2Me6 or TMA), as it exists as a dimer. This colorless liquid is pyrophoric. It is an industriall ...
, the monomer triisobutylaluminium
Triisobutylaluminium (TiBA) is an organoaluminium compound with the formula Al(CH2CH(CH3)2)3. This colorless pyrophoricity, pyrophoric liquid is mainly used to make linear primary alcohol (chemistry), alcohols and α-olefins.Michael J. Krause, Fr ...
, and the titanium-aluminium compound called Tebbe's reagent
Tebbe's reagent is the organometallic compound with the formula (C5H5)2TiCH2ClAl(CH3)2. It is used in the methylenation of carbonyl compounds, that is it converts organic compounds containing the R2C=O group into the related R2C=CH2 derivative. It ...
. The behavior of organoaluminium compounds can be understood in terms of the polarity of the C−Al bond and the high Lewis acidity
A Lewis acid (named for the American physical chemist Gilbert N. Lewis) is a chemical species that contains an empty orbital which is capable of accepting an electron pair from a Lewis base to form a Lewis adduct. A Lewis base, then, is any sp ...
of the three-coordinated species. Industrially, these compounds are mainly used for the production of polyolefin
A polyolefin is a type of polymer with the general formula (CH2CHR)n where R is an alkyl group. They are usually derived from a small set of simple olefins (alkenes). Dominant in a commercial sense are polyethylene and polypropylene. More speciali ...
s.
History
The first organoaluminium compound (C2H5)3Al2I3 was discovered in 1859. Organoaluminium compounds were, however, little known until the 1950s whenKarl Ziegler
Karl Waldemar Ziegler (26 November 1898 – 12 August 1973) was a German chemist who won the Nobel Prize in Chemistry in 1963, with Giulio Natta, for work on polymers. The Nobel Committee recognized his "excellent work on organometallic compounds ...
and colleagues discovered the direct synthesis of trialkylaluminium compounds and applied these compounds to catalytic olefin polymerization
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
. This line of research ultimately resulted in the Nobel Prize to Ziegler.
Structure and bonding
Aluminium(III) compounds
Organoaluminium compounds generally feature three- and four-coordinate Al centers, although highercoordination number
In chemistry, crystallography, and materials science, the coordination number, also called ligancy, of a central atom in a molecule or crystal is the number of atoms, molecules or ions bonded to it. The ion/molecule/atom surrounding the central i ...
s are observed with inorganic ligands such as fluoride
Fluoride (). According to this source, is a possible pronunciation in British English. is an inorganic, monatomic anion of fluorine, with the chemical formula (also written ), whose salts are typically white or colorless. Fluoride salts typ ...
. In accord with the usual trends, four-coordinate Al prefers to be tetrahedral. In contrast to boron, aluminium is a larger atom and easily accommodates four carbon ligands. The triorganoaluminium compounds are thus usually dimeric with a pair of bridging alkyl ligands, e.g., Al2(C2H5)4(μ-C2H5)2. Thus, despite its common name of triethylaluminium, this compound contains two aluminium centres, and six ethyl group
In organic chemistry, an ethyl group (abbr. Et) is an alkyl substituent with the formula , derived from ethane (). ''Ethyl'' is used in the International Union of Pure and Applied Chemistry's nomenclature of organic chemistry for a saturated ...
s. When the organoaluminium compound contain hydride
In chemistry, a hydride is formally the anion of hydrogen( H−). The term is applied loosely. At one extreme, all compounds containing covalently bound H atoms are called hydrides: water (H2O) is a hydride of oxygen, ammonia is a hydride of ...
or halide
In chemistry, a halide (rarely halogenide) is a binary chemical compound, of which one part is a halogen atom and the other part is an element or radical that is less electronegative (or more electropositive) than the halogen, to make a fluor ...
, these smaller ligands tend to occupy the bridging sites. Three coordination occurs when the R groups is bulky, e.g. Al(Mes)3 (Mes = 2,4,6-Me3C6H2 or mesityl) or isobutyl.

Ligand exchange in trialkylaluminium compounds
The trialkylaluminium dimers often participate in dynamic equilibria, resulting in the interchange of bridging and terminal ligands as well as ligand exchange between dimers. Even in noncoordinating solvents, Al-Me exchange is fast, as confirmed by proton NMR spectroscopy. For example, at −25 °C the 1H NMR spectrum of Me6Al2 comprises two signals in 1:2 ratio, as expected from the solid state structure. At 20 °C, only one signal is observed because exchange of terminal and bridging methyl groups is too fast to be resolved by NMR. The high Lewis acidity of the monomeric species is related to the size of the Al(III) center and its tendency to achieve an octet configuration.Low oxidation state organoaluminium compounds
The first organoaluminium compound with an Al-Al bond was reported in 1988 as (((Me3Si)2CH)2Al)2 (a dialane). They are typically prepared reduction of the dialkylaluminium chlorides by metallic potassium: :(R2AlCl)2 + 2 K → R2Al-AlR2 + 2 KCl Another notable group of alanes are tetraalanes containing four Al(I) centres. These compounds adopt a tetrahedrane core, as illustrated by ( Cp*Al)4 and ((Me3Si3C)Al)4. The cluster i-Bu)12">isobutyl.html" ;"title="l12(isobutyl">i-Bu)12sup>2− was obtained from related investigations on the reduction of organoaluminium compounds. This dianion adopts an icosahedral structure reminiscent of dodecaborate ([B12H12]2−). Its formal oxidation state is less than one.Preparation
From alkyl halides and aluminium
Industrially, simple aluminium alkyls of the type Al2R6 (R = Me, Et) are prepared in a two-step process beginning with the alkylation of aluminium powder: :2 Al + 3 CH3CH2Cl → (CH3CH2)3Al2Cl3 The reaction resembles the synthesisGrignard reagent
A Grignard reagent or Grignard compound is a chemical compound with the general formula , where X is a halogen and R is an organic group, normally an alkyl or aryl. Two typical examples are methylmagnesium chloride and phenylmagnesium bromide ...
s. The product, (CH3CH2)3Al2Cl3, is called ethylaluminium sesquichloride
Ethylaluminium sesquichloride, also called EASC, is an industrially important organoaluminium compound used primarily as a precursor to triethylaluminium and as a catalyst component in Ziegler–Natta type systems for olefin and diene polymeriza ...
. The term sesquichloride refers to the fact that, on average, the Cl:Al ratio is 1.5. These sesquichlorides can be converted to the triorganoaluminium derivatives by reduction:
:2 (CH3CH2)3Al2Cl3 + 6 Na → (CH3CH2)6Al2 + 2 Al + 6 NaCl
This method is used for production of trimethylaluminium and triethylaluminium.
Hydroalumination
Aluminium powder reacts directly with certain terminal alkenes in the presence of hydrogen. The process entails two steps, the first producing dialkylaluminium hydrides. Such reactions are typically conducted at elevated temperatures and require activation by trialkylaluminium reagents: :6 Al + 3 H2 + 12 CH2=CHR → 2 Al(CH2CHR)2sub>3 For nonbulky R groups, the organoaluminium hydrides are typically trimeric. In a subsequent step, these hydrides are treated with more alkene to effect hydroalumiunation: :2 Al(CH2CHR)2sub>3 + 3 CH2=CHR → 3 l2(CH2CHR)3 Diisobutylaluminium hydride, which is dimeric, is prepared by hydride elimination from triisobutylaluminium: :2 ''i''-Bu3Al → (''i''-Bu2AlH)2 + 2 isobutene">(CH3)2C=CH2Carboalumination
Organoaluminum compounds can react with alkenes and alkynes, resulting in the net addition of one organyl group and the metal fragment across the multiple bond (carboalumination). This process can proceed in a purely thermal manner or in the presence of a transition metal catalyst. For the uncatalyzed process, monoaddition is only possible when the alkene is substituted. For ethylene, carboalumination leads to a Poisson distribution of higher alkylaluminum species. The reaction is regioselective for 1-alkenes. The so-called ZACA reaction first reported by Ei-ichi Negishi is an example of an asymmetric carboalumination of alkenes catalyzed by a chiral zirconocene catalyst. The methylalumination of alkynes in the presence of Cp2ZrCl2 is employed for the synthesis of stereodefined trisubstituted olefin fragments, a common substructure in terpene and polyketide natural products. The synthesis of (''E'')-4-iodo-3-methylbut-3-en-1-ol shown below is a typical application of this reaction: For terminal alkynes, the reaction generally proceeds with good regioselectivity (>90:10 rr) and complete ''syn'' selectivity, even in the presence of propargylic or homopropargylic heteroatom substituents. Unfortunately, extension of the zirconocene-catalyzed methylalumination to alkylalumination with higher alkyls results in lower yields and poor regioselectivities.
For terminal alkynes, the reaction generally proceeds with good regioselectivity (>90:10 rr) and complete ''syn'' selectivity, even in the presence of propargylic or homopropargylic heteroatom substituents. Unfortunately, extension of the zirconocene-catalyzed methylalumination to alkylalumination with higher alkyls results in lower yields and poor regioselectivities.
Laboratory preparations
Although the simple members are commercially available at low cost, many methods have been developed for their synthesis in the laboratory, including metathesis ortransmetalation Transmetalation (alt. spelling: transmetallation) is a type of organometallic reaction that involves the transfer of ligands from one metal to another. It has the general form:
:M1–R + M2–R′ → M1–R′ + M2–R
where R and R′ can be, but ...
.
*Metathesis of aluminium trichloride with RLi or RMgX gives the trialkyl:
:AlCl3 + 3 BuLi → Bu3Al + 3 LiCl
*Transmetalation:
:2 Al + 3 HgPh2 → 2 AlPh3 + 3 Hg
Reactions
The high reactivity of organoaluminium compounds toward electrophiles is attributed to the charge separation betweenaluminium
Aluminium (aluminum in American and Canadian English) is a chemical element with the symbol Al and atomic number 13. Aluminium has a density lower than those of other common metals, at approximately one third that of steel. I ...
and carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent
In chemistry, the valence (US spelling) or valency (British spelling) of an element is the measure of its combining capacity with o ...
atom.
Lewis acidity
Organoaluminium compounds are hard acids and readily form adducts with bases such as pyridine, THF and tertiary amines. These adducts are tetrahedral at Al.Electrophiles
The Al–C bond is polarized such that the carbon is highly basic. Acids react to give alkanes. For example, alcohols givealkoxide
In chemistry, an alkoxide is the conjugate base of an alcohol and therefore consists of an organic group bonded to a negatively charged oxygen atom. They are written as , where R is the organic substituent. Alkoxides are strong bases and, whe ...
s:
:AlR'3 + ROH → 1/n (R'2Al−OR)n + R'H
A wide variety of acids can be employed beyond the simple mineral acids. Amines give amido derivatives. With carbon dioxide, trialkylaluminium compounds give the dialkylaluminium carboxylate, and subsequently alkyl aluminium dicarboxylates:
:AlR3 + CO2 → R2AlO2CR
:R2AlO2CR + CO2 → RAl(O2CR)2
The conversion is reminiscent of the carbonation of Grignard reagent
A Grignard reagent or Grignard compound is a chemical compound with the general formula , where X is a halogen and R is an organic group, normally an alkyl or aryl. Two typical examples are methylmagnesium chloride and phenylmagnesium bromide ...
s.
Similarly, the reaction between trialkylaluminum compounds and carbon dioxide has been used to synthesise alcohols, olefins, or ketones.
With oxygen one obtains the corresponding alkoxides, which can be hydrolysed to the alcohols:
:AlR3 + 3/2 O2 → Al(OR)3
A structurally characterized organo aluminum peroxide is l(R)-O-O-CMe3 =CH(SiMe3)2
The reaction between pure trialalkylaluminum compounds and water, alcohol
Alcohol most commonly refers to:
* Alcohol (chemistry), an organic compound in which a hydroxyl group is bound to a carbon atom
* Alcohol (drug), an intoxicant found in alcoholic drinks
Alcohol may also refer to:
Chemicals
* Ethanol, one of sev ...
s, phenols, amines, carbon dioxide, sulfur oxides, nitrogen oxide Nitrogen oxide may refer to a binary compound of oxygen and nitrogen, or a mixture of such compounds:
Charge-neutral
*Nitric oxide (NO), nitrogen(II) oxide, or nitrogen monoxide
*Nitrogen dioxide (), nitrogen(IV) oxide
* Nitrogen trioxide (), or n ...
s, halogen
The halogens () are a group in the periodic table consisting of five or six chemically related elements: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts). In the modern IUPAC nomenclature, this group is ...
s, and halogenated hydrocarbons can be violent.
Applications
Organoaluminium compounds are widely used in the production of alkenes, alcohols, and polymers. Some relevant processes include the Ziegler Process for the production of alcohols from ethylene. Several technologies exist for the oligomerization of ethylene to give alpha-olefins. Organoaluminium compounds are used as catalysts for alkene polymerization to polyolefins, for example the catalyst methylaluminoxane.References
{{Authority control