Mitochondrial theory of ageing on:
[Wikipedia]
[Google]
[Amazon]
The mitochondrial theory of ageing has two varieties: free radical and non-free radical. The first is one of the variants of the free radical theory of ageing. It was formulated by J. Miquel and colleagues in 1980 and was developed in the works of Linnane and coworkers (1989). The second was proposed by A. N. Lobachev in 1978.
The mitochondrial free radical theory of ageing (MFRTA) proposes that free radicals produced by mitochondrial activity damage cellular components, leading to ageing.
 Mitochondria are
Mitochondria are 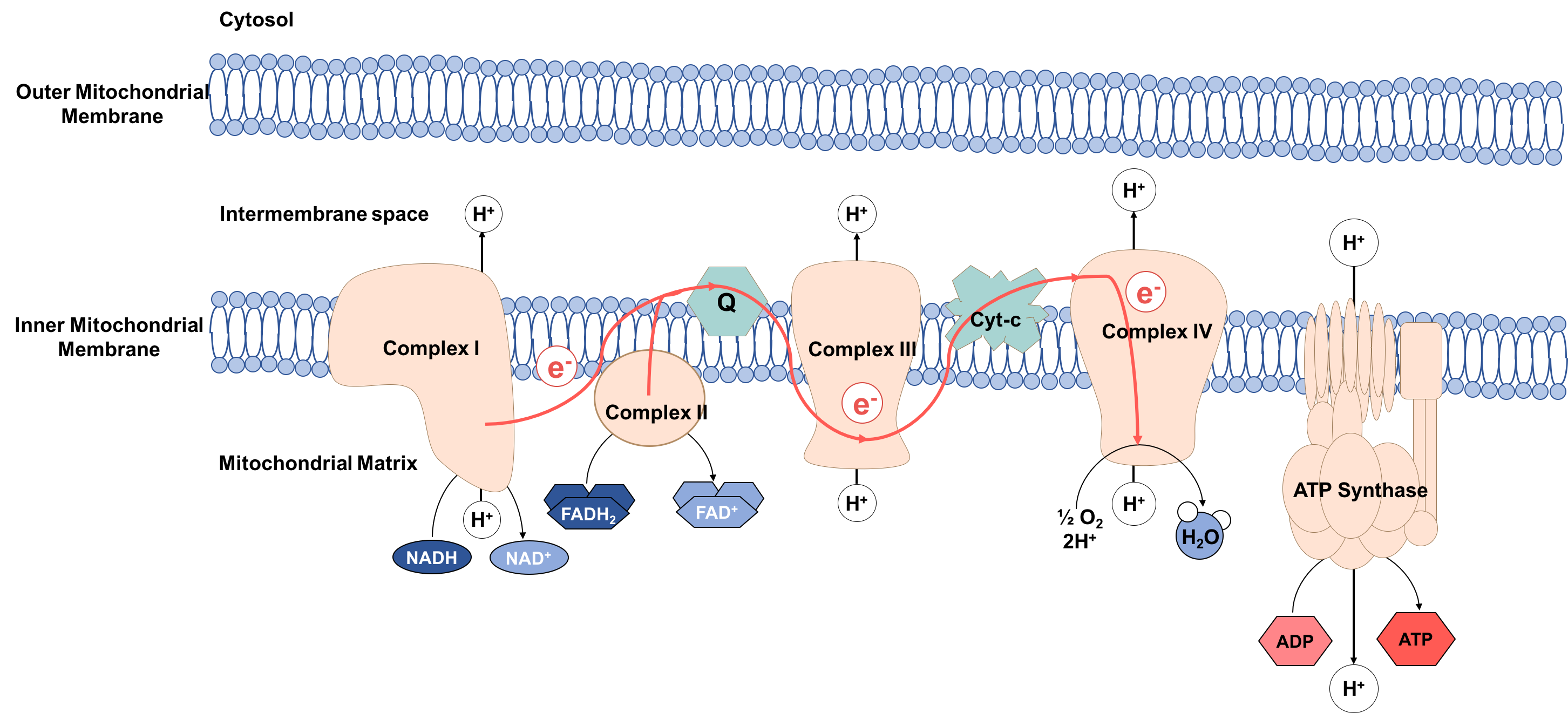 In the 1950s
In the 1950s
 Mitochondria are thought to be organelles that developed from endocytosed
Mitochondria are thought to be organelles that developed from endocytosed
 ROS are highly
ROS are highly
 Mitochondria are
Mitochondria are cell
Cell most often refers to:
* Cell (biology), the functional basic unit of life
Cell may also refer to:
Locations
* Monastic cell, a small room, hut, or cave in which a religious recluse lives, alternatively the small precursor of a monastery ...
organelles which function to provide the cell with energy by producing ATP (adenosine triphosphate). During ATP production electrons
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no ...
can escape the mitochondrion and react with water, producing reactive oxygen species
In chemistry, reactive oxygen species (ROS) are highly reactive chemicals formed from diatomic oxygen (). Examples of ROS include peroxides, superoxide, hydroxyl radical, singlet oxygen, and alpha-oxygen.
The reduction of molecular oxygen () p ...
, ROS for short. ROS can damage macromolecules
A macromolecule is a very large molecule important to biophysical processes, such as a protein or nucleic acid. It is composed of thousands of covalently bonded atoms. Many macromolecules are polymers of smaller molecules called monomers. The ...
, including lipids
Lipids are a broad group of naturally-occurring molecules which includes fats, waxes, sterols, fat-soluble vitamins (such as vitamins A, D, E and K), monoglycerides, diglycerides, phospholipids, and others. The functions of lipids include ...
, proteins and DNA, which is thought to facilitate the process of ageing.
 In the 1950s
In the 1950s Denham Harman
Denham Harman (February 14, 1916 – November 25, 2014) was an American medical academic who latterly served as professor emeritus at the University of Nebraska Medical Center. Harman is known as the "father of the free radical theory of aging". ...
proposed the free radical theory of ageing, which he later expanded to the MFRTA.
When studying the mutations in antioxidants
Antioxidants are compounds that inhibit oxidation, a chemical reaction that can produce free radicals. This can lead to polymerization and other chain reactions. They are frequently added to industrial products, such as fuels and lubricants, ...
, which remove ROS, results were inconsistent. However, it has been observed that overexpression of antioxidant enzymes in yeast
Yeasts are eukaryotic, single-celled microorganisms classified as members of the fungus kingdom. The first yeast originated hundreds of millions of years ago, and at least 1,500 species are currently recognized. They are estimated to constit ...
, worms Worms may refer to:
*Worm, an invertebrate animal with a tube-like body and no limbs
Places
*Worms, Germany
Worms () is a city in Rhineland-Palatinate, Germany, situated on the Upper Rhine about south-southwest of Frankfurt am Main. It had ...
, flies
Flies are insects of the order Diptera, the name being derived from the Greek δι- ''di-'' "two", and πτερόν ''pteron'' "wing". Insects of this order use only a single pair of wings to fly, the hindwings having evolved into advanced m ...
and mice were shown to increase lifespan.
Molecular basis
 Mitochondria are thought to be organelles that developed from endocytosed
Mitochondria are thought to be organelles that developed from endocytosed bacteria
Bacteria (; singular: bacterium) are ubiquitous, mostly free-living organisms often consisting of one Cell (biology), biological cell. They constitute a large domain (biology), domain of prokaryotic microorganisms. Typically a few micrometr ...
which learned to coexist
Coexist may refer to:
* ''Coexist'' (album), a 2012 album by The xx
* Coexist (song), 2019 song by Coldrain
* Coexist Foundation, a charitable organization based in London, England
* Coexist (image)
The Coexist image (often styled as "CoeXisT ...
inside ancient
Ancient history is a time period from the beginning of writing and recorded human history to as far as late antiquity. The span of recorded history is roughly 5,000 years, beginning with the Sumerian cuneiform script. Ancient history cov ...
cells. These bacteria maintained their own DNA, the mitochondrial DNA (mtDNA), which codes for components of the electron transport chain (ETC). The ETC is found in the inner mitochondrial membrane
The inner mitochondrial membrane (IMM) is the mitochondrial membrane which separates the mitochondrial matrix from the intermembrane space.
Structure
The structure of the inner mitochondrial membrane is extensively folded and compartmentalized. ...
and functions to produce energy
In physics, energy (from Ancient Greek: ἐνέργεια, ''enérgeia'', “activity”) is the quantitative property that is transferred to a body or to a physical system, recognizable in the performance of work and in the form of hea ...
in the form of ATP molecules. The process is called oxidative phosphorylation, because ATP is produced from ADP in a series of redox reactions. Electrons are transferred through the ETC from NADH
Nicotinamide adenine dinucleotide (NAD) is a coenzyme central to metabolism. Found in all living cells, NAD is called a dinucleotide because it consists of two nucleotides joined through their phosphate groups. One nucleotide contains an aden ...
and FADH2 to oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as ...
, reducing oxygen to water.
ROS
 ROS are highly
ROS are highly reactive
Reactive may refer to:
*Generally, capable of having a reaction (disambiguation)
*An adjective abbreviation denoting a bowling ball coverstock made of reactive resin
*Reactivity (chemistry)
*Reactive mind
*Reactive programming
See also
*Reactanc ...
, oxygen-containing chemical species
A chemical species is a chemical substance or ensemble composed of chemically identical molecular entities that can explore the same set of molecular energy levels on a characteristic or delineated time scale. These energy levels determine the wa ...
, which include superoxide
In chemistry, a superoxide is a compound that contains the superoxide ion, which has the chemical formula . The systematic name of the anion is dioxide(1−). The reactive oxygen ion superoxide is particularly important as the product of t ...
, hydrogen peroxide
Hydrogen peroxide is a chemical compound with the formula . In its pure form, it is a very pale blue liquid that is slightly more viscous than water. It is used as an oxidizer, bleaching agent, and antiseptic, usually as a dilute solution (3%� ...
and hydroxyl radical
The hydroxyl radical is the diatomic molecule . The hydroxyl radical is very stable as a dilute gas, but it decays very rapidly in the condensed phase. It is pervasive in some situations. Most notably the hydroxyl radicals are produced from the ...
.
If the complexes of the ETC do not function properly, electrons can leak and react with water, forming ROS. Normally leakage is low and ROS is kept at physiological levels, fulfilling roles in signaling and homeostasis
In biology, homeostasis (British also homoeostasis) (/hɒmɪə(ʊ)ˈsteɪsɪs/) is the state of steady internal, physical, and chemical conditions maintained by living systems. This is the condition of optimal functioning for the organism and ...
. In fact, their presence at low levels lead to increased life span, by activating transcription factors
In molecular biology, a transcription factor (TF) (or sequence-specific DNA-binding factor) is a protein that controls the rate of transcription of genetic information from DNA to messenger RNA, by binding to a specific DNA sequence. The fun ...
and metabolic pathways
In biochemistry, a metabolic pathway is a linked series of chemical reactions occurring within a cell. The reactants, products, and intermediates of an enzymatic reaction are known as metabolites, which are modified by a sequence of chemical re ...
involved in longevity. At increased levels ROS cause oxidative damage
Oxidative stress reflects an imbalance between the systemic manifestation of reactive oxygen species and a biological system's ability to readily detoxify the reactive intermediates or to repair the resulting damage. Disturbances in the normal r ...
by oxidizing macromolecules, such as lipids, proteins and DNA. This oxidative damage to macromolecules is thought to be the cause of ageing. Mitochondrial DNA is especially susceptible to oxidative damage, due to its proximity to the site of production of these species. Damaging of mitochondrial DNA causes mutations, leading to production of ETC complexes, which don’t function properly, increasing ROS production, increasing oxidative damage to macromolecules.
UPRmt
The mitochondrial unfolded protein response (UPRmt) is turned on in response to mitochondrial stress. Mitochondrial stress occurs when theproton gradient
An electrochemical gradient is a gradient of electrochemical potential, usually for an ion that can move across a membrane. The gradient consists of two parts, the chemical gradient, or difference in solute concentration across a membrane, and th ...
across the inner mitochondrial membrane is dissipated, mtDNA is mutated, and/or ROS accumulates, which can lead to misfolding
Protein folding is the physical process by which a protein chain is Translation (biology), translated to its native protein tertiary structure, three-dimensional structure, typically a "folded" Protein structure, conformation by which the prote ...
and reduced function of mitochondrial proteins. Stress is sensed by the nucleus
Nucleus ( : nuclei) is a Latin word for the seed inside a fruit. It most often refers to:
*Atomic nucleus, the very dense central region of an atom
* Cell nucleus, a central organelle of a eukaryotic cell, containing most of the cell's DNA
Nucl ...
, where chaperones and proteases
A protease (also called a peptidase, proteinase, or proteolytic enzyme) is an enzyme that catalyzes (increases reaction rate or "speeds up") proteolysis, breaking down proteins into smaller polypeptides or single amino acids, and spurring the for ...
are upregulated
In the biological context of organisms' production of gene products, downregulation is the process by which a cell decreases the quantity of a cellular component, such as RNA or protein, in response to an external stimulus. The complementary pro ...
, which can correct folding
Fold, folding or foldable may refer to:
Arts, entertainment, and media
* ''Fold'' (album), the debut release by Australian rock band Epicure
* Fold (poker), in the game of poker, to discard one's hand and forfeit interest in the current pot
*Abov ...
or remove damaged proteins, respectively. Decrease in protease levels are associated with ageing, as mitochondrial stress will remain, maintaining high ROS levels. Such mitochondrial stress and dysfunction has been linked to various age-associated diseases, including cardiovascular diseases
Cardiovascular disease (CVD) is a class of diseases that involve the heart or blood vessels. CVD includes coronary artery diseases (CAD) such as angina and myocardial infarction (commonly known as a heart attack). Other CVDs include stroke, ...
, and type-2 diabetes
Type 2 diabetes, formerly known as adult-onset diabetes, is a form of diabetes mellitus that is characterized by high blood sugar, insulin resistance, and relative lack of insulin. Common symptoms include increased thirst, frequent urinatio ...
.
Mitochondrial metabolites
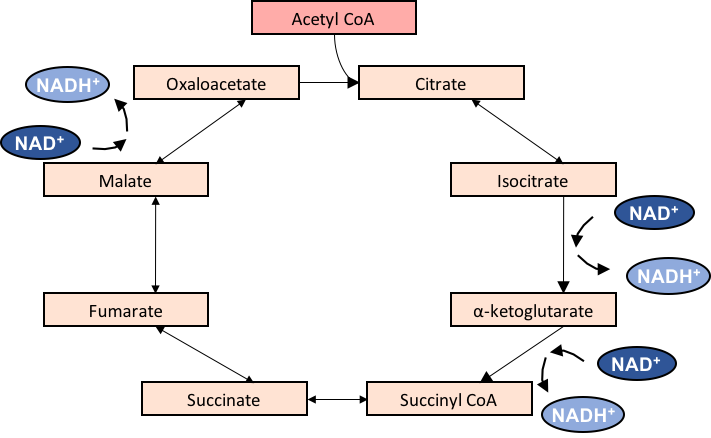
As themitochondrial matrix
In the mitochondrion, the matrix is the space within the inner membrane. The word "matrix" stems from the fact that this space is viscous, compared to the relatively aqueous cytoplasm. The mitochondrial matrix contains the mitochondrial DNA, ribo ...
is where the TCA cycle
The citric acid cycle (CAC)—also known as the Krebs cycle or the TCA cycle (tricarboxylic acid cycle)—is a series of chemical reactions to release stored energy through the oxidation of acetyl-CoA derived from carbohydrates, fats, and protein ...
takes place, different metabolites
In biochemistry, a metabolite is an intermediate or end product of metabolism.
The term is usually used for small molecules. Metabolites have various functions, including fuel, structure, signaling, stimulatory and inhibitory effects on enzymes, ...
are commonly confined to the mitochondria. Upon ageing, mitochondrial function declines, allowing escape of these metabolites, which can induce epigenetic changes, associated with ageing.

Acetyl-coenzyme A
Acetyl-CoA (acetyl coenzyme A) is a molecule that participates in many biochemical reactions in protein, carbohydrate and lipid metabolism. Its main function is to deliver the acetyl group to the citric acid cycle (Krebs cycle) to be oxidized for ...
(Acetyl-CoA) enters the TCA cycle in the mitochondrial matrix, and is oxidized
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a ...
in the process of energy production. Upon escaping the mitochondria and entering the nucleus, it can act as a substrate for histone acetylation
Histone acetyltransferases (HATs) are enzymes that acetylate conserved lysine amino acids on histone proteins by transferring an acetyl group from acetyl-CoA to form ε-''N''-acetyllysine. DNA is wrapped around histones, and, by transferring an ...
. Histone acetylation is an epigenetic modification, which leads to gene activation
Regulation of gene expression, or gene regulation, includes a wide range of mechanisms that are used by cells to increase or decrease the production of specific gene products (protein or RNA). Sophisticated programs of gene expression are wi ...
.
At a young age, acetyl-CoA levels are higher in the nucleus and cytosol
The cytosol, also known as cytoplasmic matrix or groundplasm, is one of the liquids found inside cells ( intracellular fluid (ICF)). It is separated into compartments by membranes. For example, the mitochondrial matrix separates the mitochondri ...
, and its transport to the nucleus can extend lifespan in worms.
Nicotinamide Adenine Dinucleotide (NAD+) is produced in the mitochondria and upon escaping to the nucleus, can act as a substrate for sirtuins. Sirtuins are family of proteins, known to play a role in longevity. Cellular NAD+ levels have been shown to decrease with age.
DAMPs
Damage-associated molecular patterns (DAMPs) are molecules that are released during cell stress. Mitochondrial DNA is a DAMP, which only becomes available during mitochondrial damage. Blood mitochondrial DNA levels become elevated with age, contributing to inflamm-ageing, a chronic state of inflammation characteristic of advanced age.Mitochondrial-derived peptides
Mitochondrial DNA has been known to encode 13 proteins. Recently, other short protein coding sequences have been identified, and their products are referred to as mitochondria-derived peptides. The mitochondrial-derived peptide,humanin
Humanin is a micropeptide encoded in the mitochondrial genome by the 16S ribosomal RNA gene, MT-RNR2. Its structure contains a three-turn α-helix, and no symmetry.
In ''in vitro'' and animal models, it appears to have cytoprotective effects. ...
has been shown to protect against Alzheimer’s disease
Alzheimer's disease (AD) is a neurodegenerative disease that usually starts slowly and progressively worsens. It is the cause of 60–70% of cases of dementia. The most common early symptom is difficulty in remembering recent events. As t ...
, which is considered an age-associated disease.
MOTS-c has been shown to prevent age-associated insulin resistance, the main cause of type 2 diabetes.
Humanin and MOTS-c levels have been shown to decline with age, and their activity seems to increase longevity.
Mitochondrial membrane
Almaida-Pagan and coworkers found that mitochondrialmembrane
A membrane is a selective barrier; it allows some things to pass through but stops others. Such things may be molecules, ions, or other small particles. Membranes can be generally classified into synthetic membranes and biological membranes. ...
lipid composition changes with age, when studying Turquoise killifish. The proportion of monosaturated fatty acids
In chemistry, particularly in biochemistry, a fatty acid is a carboxylic acid with an aliphatic chain, which is either saturated and unsaturated compounds#Organic chemistry, saturated or unsaturated. Most naturally occurring fatty acids have an B ...
decreased with age, and the proportion of polysaturated
''Polysaturated'' is the debut album released by New Zealand hip-hop group, Nesian Mystik, in 2002. The album entered the New Zealand charts at number one, and was certified gold by the end of the first week of sales.
The song "For the People" w ...
fatty acids increased. The overall phospholipid content also decreased with age.
History
In 1956Denham Harman
Denham Harman (February 14, 1916 – November 25, 2014) was an American medical academic who latterly served as professor emeritus at the University of Nebraska Medical Center. Harman is known as the "father of the free radical theory of aging". ...
first postulated the free radical theory of ageing, which he later modified to the mitochondrial free radical theory of ageing (MFRTA). He found ROS as the main cause of damage to macromolecules, known as “ageing”. He later modified his theory because he found that mitochondria were producing and being damaged by ROS, leading him to the conclusion that mitochondria determine ageing. In 1972, he published his theory in the Journal of the American Geriatrics Society.
Evidence
It has been observed that with age, mitochondrial function declines and mitochondrial DNA mutation increases in tissue cells in an age-dependent manner. This leads to increase in ROS production and potential decrease in the cell’s ability to remove ROS. Most long-living animals have been shown to be more resistant to oxidative damage and have lower ROS production, linking ROS levels to lifespan. Overexpression ofantioxidants
Antioxidants are compounds that inhibit oxidation, a chemical reaction that can produce free radicals. This can lead to polymerization and other chain reactions. They are frequently added to industrial products, such as fuels and lubricants, ...
, which function to remove ROS has also been shown to increase lifespan.
Bioinformatics analysis showed that amino acid composition of mitochondrial proteins correlate with longevity (long-living species are depleted in cysteine and methionine), linking mitochondria to the process of ageing.
By studying expression of certain genes in ''C. elegans
''Caenorhabditis elegans'' () is a free-living transparent nematode about 1 mm in length that lives in temperate soil environments. It is the type species of its genus. The name is a blend of the Greek ''caeno-'' (recent), ''rhabditis'' (r ...
'', ''Drosophila
''Drosophila'' () is a genus of flies, belonging to the family Drosophilidae, whose members are often called "small fruit flies" or (less frequently) pomace flies, vinegar flies, or wine flies, a reference to the characteristic of many speci ...
'', and mice it was found that disruption of ETC complexes can extend life – linking mitochondrial function to the process of ageing.
Evidence supporting the theory started to crumble in the early 2000s.
Mice with reduced expression of the mitochondrial antioxidant, SOD2, accumulated oxidative damage and developed cancer
Cancer is a group of diseases involving abnormal cell growth with the potential to invade or spread to other parts of the body. These contrast with benign tumors, which do not spread. Possible signs and symptoms include a lump, abnormal b ...
, but did not live longer than normal life. Overexpression of antioxidants reduced cellular stress, but did not increase mouse life span. The naked mole-rat
The naked mole-rat (''Heterocephalus glaber''), also known as the sand puppy, is a burrowing rodent native to the Horn of Africa and parts of Kenya, notably in Somali regions. It is closely related to the blesmols and is the only species in the ...
, which lives 10-times longer than normal mice, has been shown to have higher levels of oxidative damage.
See also
* Ageing *Calorie Restriction
Calorie restriction (caloric restriction or energy restriction) is a dietary regimen that reduces intake of energy from caloric foods & beverages without incurring malnutrition. "Reduce" can be defined relative to the subject's previous intake b ...
*Denham Harman
Denham Harman (February 14, 1916 – November 25, 2014) was an American medical academic who latterly served as professor emeritus at the University of Nebraska Medical Center. Harman is known as the "father of the free radical theory of aging". ...
* Free radical theory of ageing
* Gerontology
References
{{reflist Ageing